Key Takeaways
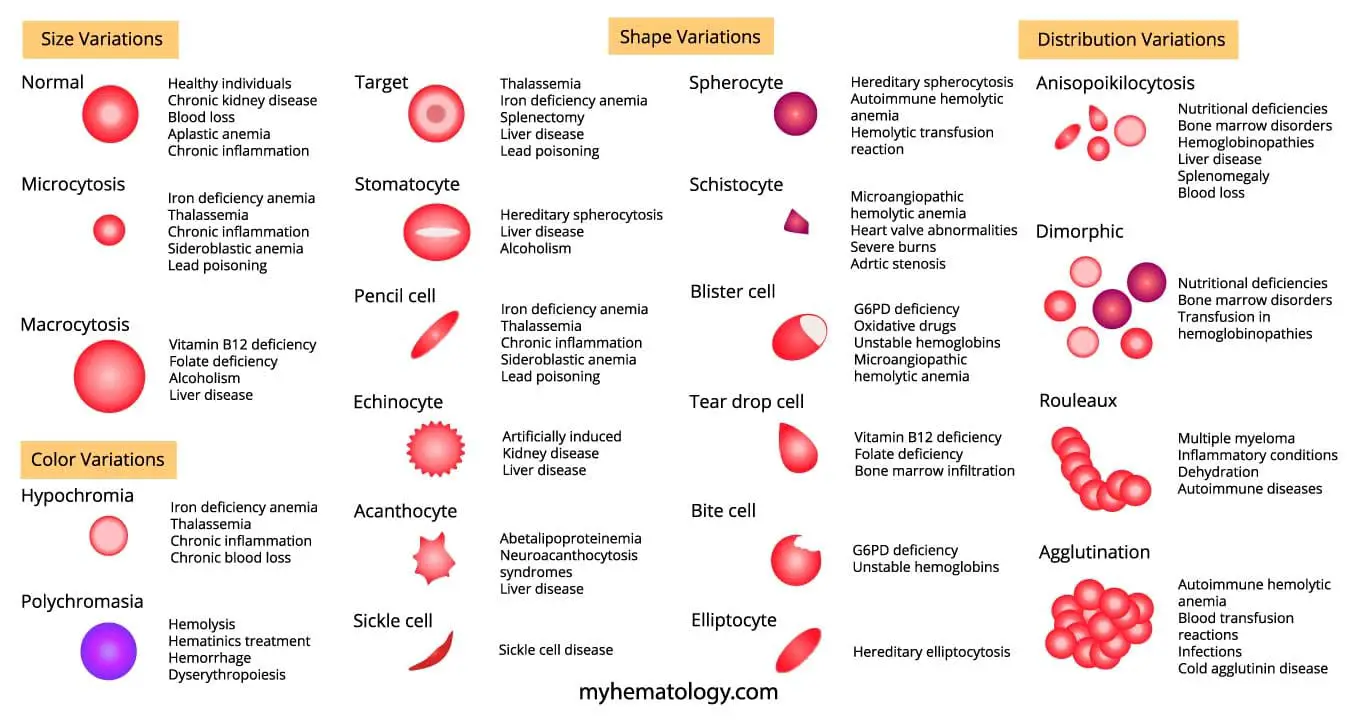
Red blood cell morphology describes the size, shape, and color of red blood cells on a peripheral blood smear, and any deviation from the normal biconcave disc serves as a clinical clue to an underlying disease. Common abnormal red blood cell morphologies and its causes include:
- Microcytosis ▾: Iron deficiency anemia, Thalassemia, Chronic inflammation, Sideroblastic anemia, and Lead poisoning
- Macrocytosis ▾: Vitamin B12 deficiency, Folate deficiency, Alcohol abuse, Liver disease
- Target Cells ▾: Liver Disease, Hemoglobinopathies, Iron Deficiency Anemia, Splenectomy, and Lead Poisoning.
- Pencil Cells ▾: Iron deficiency anemia, Thalassemia, Sideroblastic anemia, and Lead poisoning
- Schistocytes (Fragmented Cells) ▾: Microangiopathic hemolytic anemia (MAHA), Heart valve abnormalities, Severe burns, and Aortic stenosis.
*Click ▾ for more information
Introduction
The biconcave disc shape of a red blood cell is not just a textbook detail. It gives the cell a high surface area to volume ratio, lets it deform through narrow capillaries, and supports efficient oxygen exchange. When that shape fails, the consequences are clinical: shortened red cell survival, hemolysis, and tissue hypoxia. This is why red blood cell morphology is one of the most useful diagnostic tools in hematology.
This guide walks through the size, shape, color, and distribution patterns you will see on a peripheral blood smear, and explains what each pattern points toward.
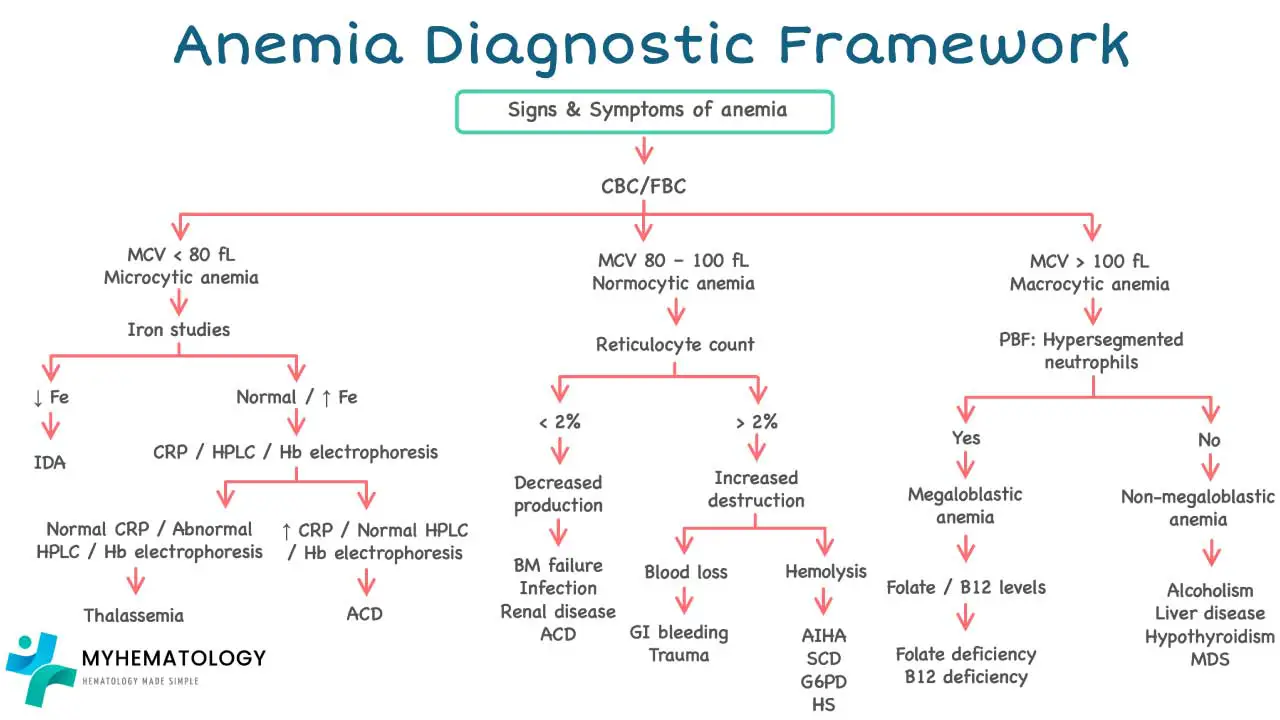
Approach to Anemia: A Brief Overview

Anemia means a deficiency of red blood cells or hemoglobin, the oxygen-carrying protein inside them. The result is reduced oxygen delivery, which produces fatigue, weakness, and shortness of breath. Diagnosis is rarely a single test. It blends history, examination, the complete blood count, and the blood smear.
Complete Blood Count (CBC)
The CBC is the cornerstone. It reports red cell count, hemoglobin, hematocrit, MCV, MCH, MCHC, and RDW (Red Cell Distribution Width). RDW deserves particular attention. It is the laboratory parameter that quantifies anisocytosis — variation in red cell size. A high RDW means a population of mixed sizes, which is common in iron deficiency, mixed nutritional deficiencies, and dimorphic pictures after transfusion [7].
History, Physical Examination, and Additional Tests
A detailed medical history and physical examination can provide valuable clues. These include:
- Dietary habits (pointing towards potential iron or vitamin deficiencies)
- Menstrual history (heavy periods can contribute to iron deficiency)
- Family history (certain anemias are hereditary)
- Presence of chronic illnesses (chronic inflammatory diseases can suppress RBC production)
- Medication use (some medications can cause drug-induced anemia)
Depending on the initial findings, further investigations may be warranted.
- Iron studies: Serum iron, ferritin, transferrin saturation, and total iron binding capacity (TIBC) help differentiate between iron deficiency anemia and other causes.
- Vitamin B12 and folate levels: Assessed to rule out deficiencies that can lead to macrocytic anemia.
- Autoimmune workup: Certain autoimmune conditions can target RBCs, requiring specific tests for confirmation.
- Bone marrow examination: In complex cases, a bone marrow biopsy or aspirate might be necessary to evaluate RBC production.
The Reticulocyte Count: Turning Observation into Function
This is the step many undergraduate texts skip. Morphology alone cannot quantify the marrow's response. Every anemia workup needs a reticulocyte count to separate two very different situations:
- Hyperproliferative anemia (high reticulocyte count) — the marrow is responding well, so the problem is peripheral: hemolysis or acute hemorrhage.
- Hypoproliferative anemia (low reticulocyte count) — the marrow is failing to respond. Think chronic kidney disease, anemia of inflammation, or aplastic anemia.
Polychromasia on a smear is a visual hint of marrow activity. The reticulocyte count is the measurement. Both are useful, and the next section explains the difference in detail.
Diagnosis and Treatment
Putting CBC, history, examination, smear, and reticulocyte count together yields a diagnosis. Treatment then targets the cause: iron supplementation for iron deficiency, B12 or folate replacement for megaloblastic anemia, transfusion or immunosuppression for hemolytic anemias, and management of the underlying disease in chronic conditions.
Key Characteristics Assessed in Red Blood Cell Morphology
The peripheral blood smear is the gold standard for assessing red cell morphology. A laboratory technician or hematopathologist examines a stained smear under high magnification, looking at four things: size, color, shape, and distribution.
Size (Normocytosis, Microcytosis, Macrocytosis)
A healthy red blood cell is 7.2–7.9 µm across — about the size of a small lymphocyte's nucleus. Mean Corpuscular Volume (MCV) is the average cell volume in femtoliters and is the standard way size is reported.
- Normocytic (MCV 80–100 fL): normal size. Healthy red cells fall here, but so do red cells in many hemolytic anemias.
- Microcytic (MCV <80 fL): smaller than normal. Suggests reduced hemoglobin production (iron deficiency, thalassemia, anemia of chronic inflammation).
- Macrocytic (MCV >100 fL): larger than normal. Suggests B12 or folate deficiency, alcohol use, liver disease, or a primary marrow disorder.
Color (Normochromia, Hypochromia, Polychromasia)
Healthy red cells stain pink with a small central pale area (about one-third of the cell's diameter), reflecting the biconcave dip. Mean Corpuscular Hemoglobin (MCH) measures average hemoglobin content per cell.
- Hypochromic cells have a larger central pallor and an MCH below 27 pg, pointing to reduced hemoglobin (classically iron deficiency).
- Polychromatic cells are young red cells that look slightly larger and bluish-tinged on a routine Wright-Giemsa stain because they still contain ribosomal RNA.
A clinically critical point that often gets missed: polychromasia is an indicator of active erythropoiesis (red cell production). If a patient is anemic but there is no polychromasia at all, the marrow is failing to respond.
Polychromasia vs. Reticulocyte — what's the difference? The terms are often used interchangeably, but they're not the same. A reticulocyte is specifically identified using a supravital stain like new methylene blue, which highlights the residual RNA as a network. Polychromasia is what those same young cells look like on a standard Wright-Giemsa stain — bluish and slightly larger. Polychromasia gives you a visual impression. The reticulocyte count gives you a number. You need the number to make a functional diagnosis.
Shape
The biconcave disc is the standard. Departures from it — sickle cells, spherocytes, elliptocytes, schistocytes, and others — point to specific diseases. The catalogue of shapes is the heart of this article and is covered below.
Distribution and the RDW
Normally, red cells are evenly spread across a smear. Two patterns of clumping matter:
- Rouleaux formation — cells stacked face-to-face like coins, driven by elevated plasma proteins (especially fibrinogen or paraproteins).
- Agglutination — irregular dense clumps caused by antibodies binding to the red cell surface.
The RDW complements distribution. A markedly elevated RDW signals mixed populations, which is particularly useful in dimorphic pictures.
Artifact vs. True Pathology
A common beginner mistake is over-reporting shapes that are actually artifacts of slide preparation. The rule of thumb:
- True pathology is scattered. It varies in intensity across the smear — present in some fields, absent in others.
- Artifact is uniform. If every cell in a field shows the same "abnormal" shape, suspect artifact and request a fresh smear.
Stomatocytes are the most common artifact. They are usually meaningless when seen only in the thick part of the smear. They are clinically significant only when found in the feathered edge or in the monolayer.
Common Abnormal Red Blood Cell Morphology Seen in Disorders
Normal Red Blood Cell Morphology
A normal red cell is 7.2–7.9 µm across with a pinkish cytoplasm and a central pallor that takes up about a third of the cell. On a smear, it sits next to a small lymphocyte at roughly the same diameter.
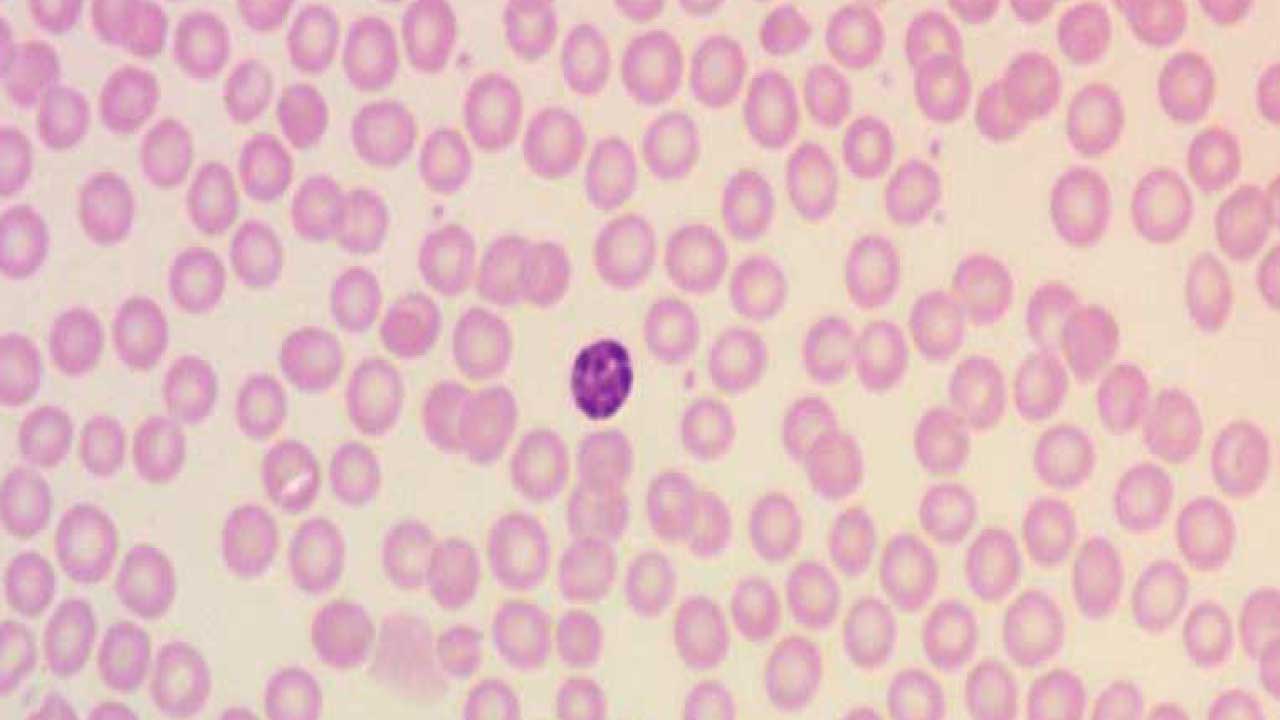
Microcytosis (Microcytes)
Microcytes are smaller than normal, usually below 7.2 µm, and often have an enlarged central pallor when hemoglobin is also reduced.
Causes of Microcytic Anemia
Iron deficiency anemia. The most common cause worldwide. Without enough iron, the marrow produces small, pale cells. Microcytes on a smear should always prompt a search for the source of iron loss — gastrointestinal bleeding, heavy menstruation, or poor intake — and confirmatory iron studies (ferritin, iron, TIBC).
Thalassemia. A genetic disorder of globin chain production. Important teaching point: thalassemia trait carriers are not iron deficient, and iron supplementation is generally contraindicated because it leads to iron overload. Hemoglobin electrophoresis or HPLC confirms the diagnosis.
Chronic inflammation (anemia of inflammation). Long-term inflammation triggers the liver to release hepcidin, a hormone that locks iron inside macrophages and makes it unavailable for new red cell production [3, 4]. These red cells often start as normocytic and progress to a microcytic pattern over time. Treatment focuses on the underlying inflammation, not iron, unless a co-existing iron deficiency is confirmed.
Sideroblastic anemia. A rare group of disorders where iron is present but cannot be incorporated into hemoglobin, accumulating as ring sideroblasts in red cell precursors. Bone marrow biopsy is often needed.
Lead poisoning. Often shows microcytes with basophilic stippling (small dark blue granules). This combination should trigger a blood lead level, especially in children or workers with occupational exposure.
Macrocytosis (Macrocytes)
Macrocytes are red cells larger than normal. The clinical definition is MCV >100 fL. On a smear, the diameter is typically >9 µm. They appear larger than the nucleus of a small lymphocyte.
Macrocytosis is broadly split into megaloblastic and non-megaloblastic causes — and the shape of the macrocyte tells you which one.
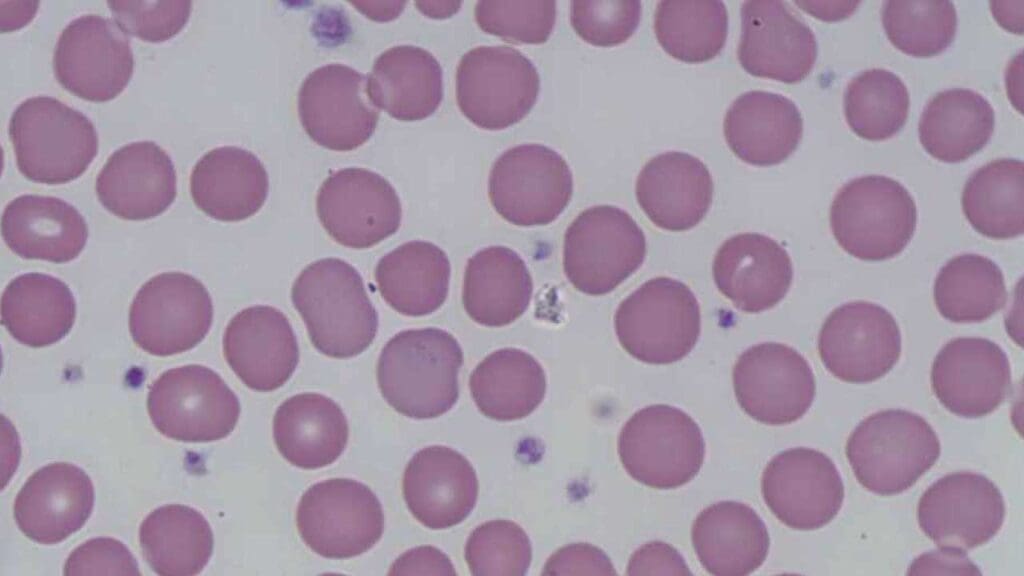
Megaloblastic Macrocytosis (B12 and Folate Deficiency)
Both vitamin B12 and folate are co-factors for DNA synthesis. When they are deficient, the nucleus matures more slowly than the cytoplasm — a process called asynchronous maturation [6].
Morphology: the hallmarks are oval macrocytes (macro-ovalocytes) and hypersegmented neutrophils (white blood cells with six or more nuclear lobes).
- Vitamin B12 deficiency carries the added risk of irreversible neurological damage (peripheral neuropathy, cognitive impairment) if untreated. Recognition of macrocytosis prompts B12 testing and, if confirmed, often lifelong supplementation.
- Folate deficiency produces the same red cell morphology but typically does not cause neurological symptoms. Differentiating from B12 matters because treating with folate alone can mask B12 deficiency and let neurological damage progress.
Non-Megaloblastic Macrocytosis
Here, DNA synthesis is normal — the change is in the red cell membrane. The macrocytes are typically round, not oval.
- Alcohol abuse: a very common cause, even without overt liver disease. Mechanisms include direct marrow toxicity, folate deficiency, and liver effects.
- Liver disease: altered lipid metabolism increases membrane surface area, producing round macrocytes.
- Hypothyroidism: thyroid hormone deficiency alters membrane lipids.
- Certain medications: chemotherapy agents (hydroxyurea, methotrexate, azathioprine) interfere with DNA synthesis; trimethoprim affects folate metabolism.
- Bone marrow disorders, particularly MDS. Persistent macrocytosis after common causes are excluded — especially with other cytopenias or dysplastic features — raises concern for Myelodysplastic Syndrome (MDS). While macrocytosis and dysplasia remain the entry-level clues on the smear, the 2022 WHO and ICC classifications now require genetic testing for definitive diagnosis and risk stratification — for example, identifying SF3B1 mutations rather than relying on ring sideroblasts alone [1, 2].
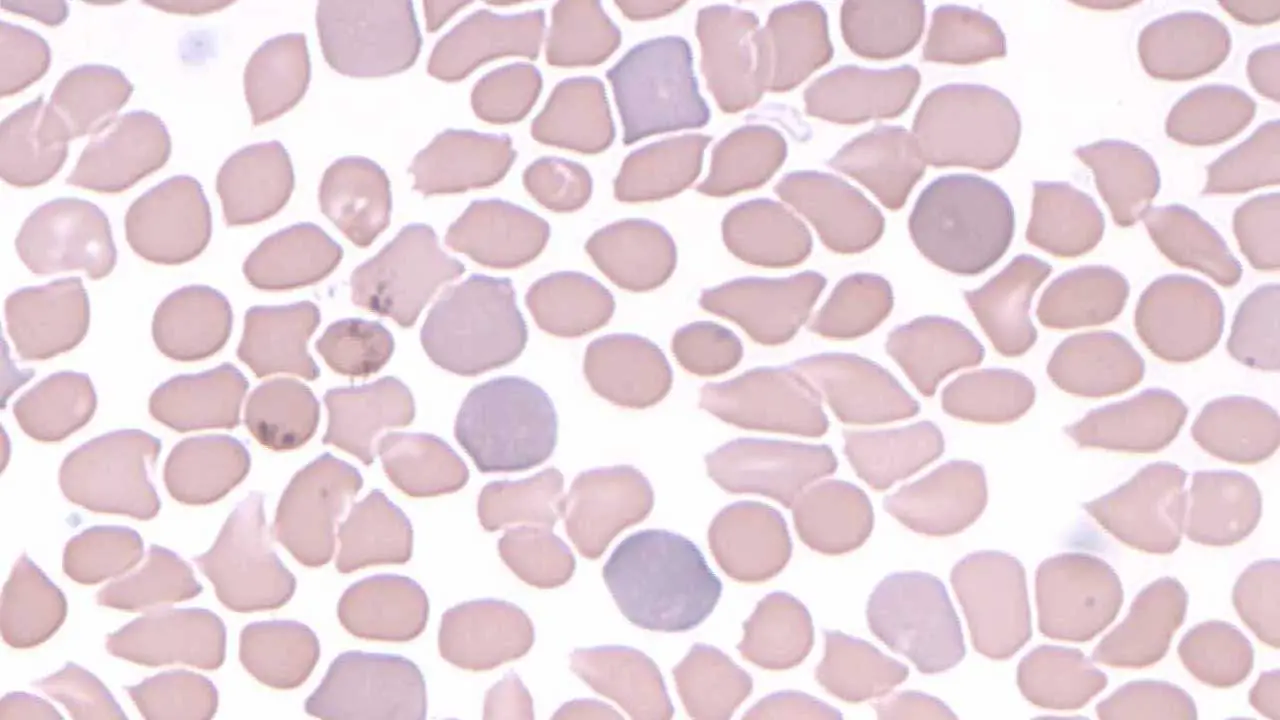
Normochromic Normocytic Red Cells
These cells look normal in size and color, yet a normocytic picture does not exclude anemia. The key here is the reticulocyte count: it tells you whether the marrow is responding.
Causes
Chronic inflammatory disease. Common in rheumatoid arthritis, inflammatory bowel disease, and chronic infection. The reticulocyte count is typically low — a hypoproliferative response driven by hepcidin and reduced erythropoiesis [3, 4].
Chronic kidney disease (CKD). Reduced erythropoietin production by the kidneys leads to a hypoproliferative anemia. Reticulocyte count is low. Treatment uses erythropoiesis-stimulating agents (ESAs) plus iron support.
Acute blood loss. In the first hours to days, remaining red cells are still normocytic. The anemia only becomes microcytic later, as iron stores deplete. Reticulocyte count rises as the marrow responds — a hyperproliferative picture.
Aplastic anemia. Marrow failure produces pancytopenia: low red cells, white cells, and platelets. Reticulocyte count is low. Bone marrow biopsy confirms a hypocellular marrow.
Myelodysplastic Syndromes (MDS). Can present as normocytic or macrocytic anemia. As above, modern diagnosis integrates morphology with genetic testing [1, 2].
Early or mixed nutritional deficiencies. Iron, B12, or folate deficiencies in their early stages can present as normocytic anemia before becoming microcytic or macrocytic.
Hypochromic Red Cells
Hypochromic cells appear pale because they contain less hemoglobin than normal. The central pallor exceeds one-third of the cell's diameter; in severe cases the cell looks almost like a thin ring (a "ghost cell," classically seen in beta-thalassemia major).
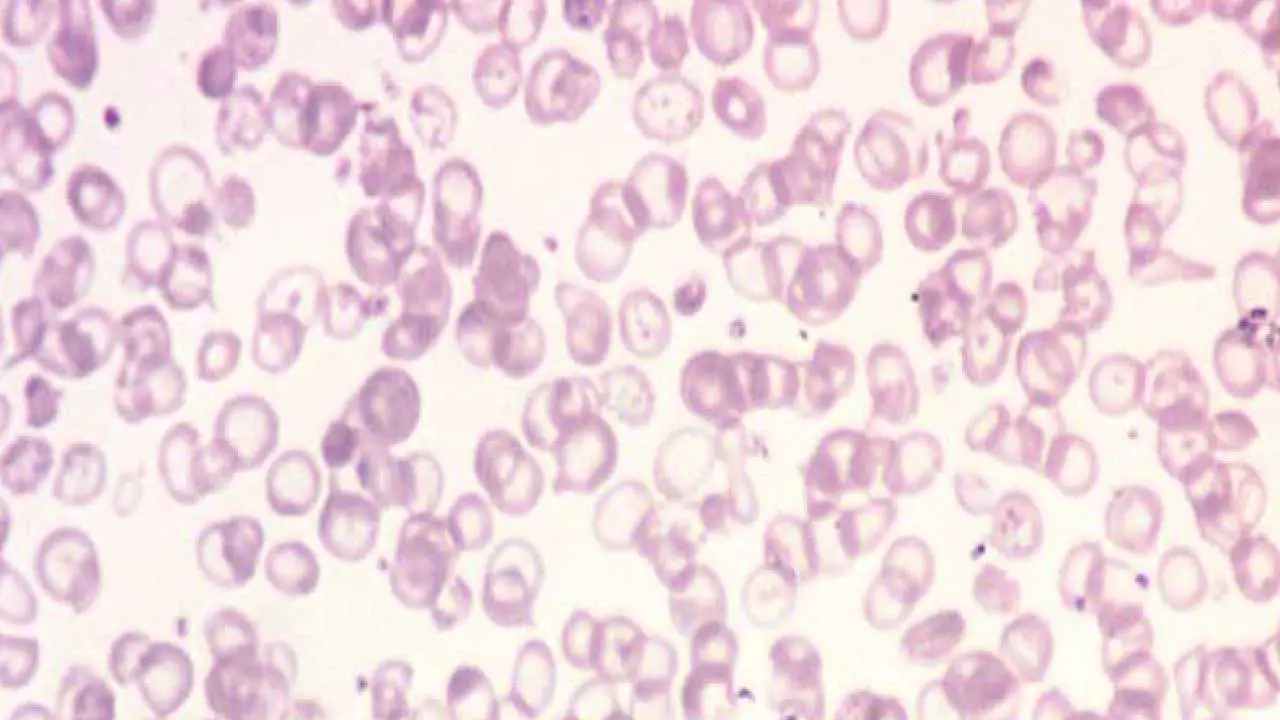
Causes
- Iron deficiency anemia — the most common cause.
- Chronic blood loss — peptic ulcers, heavy menstruation.
- Chronic inflammatory disease — via hepcidin-mediated iron sequestration [3, 4].
- Thalassemia and other inherited disorders affecting hemoglobin synthesis.
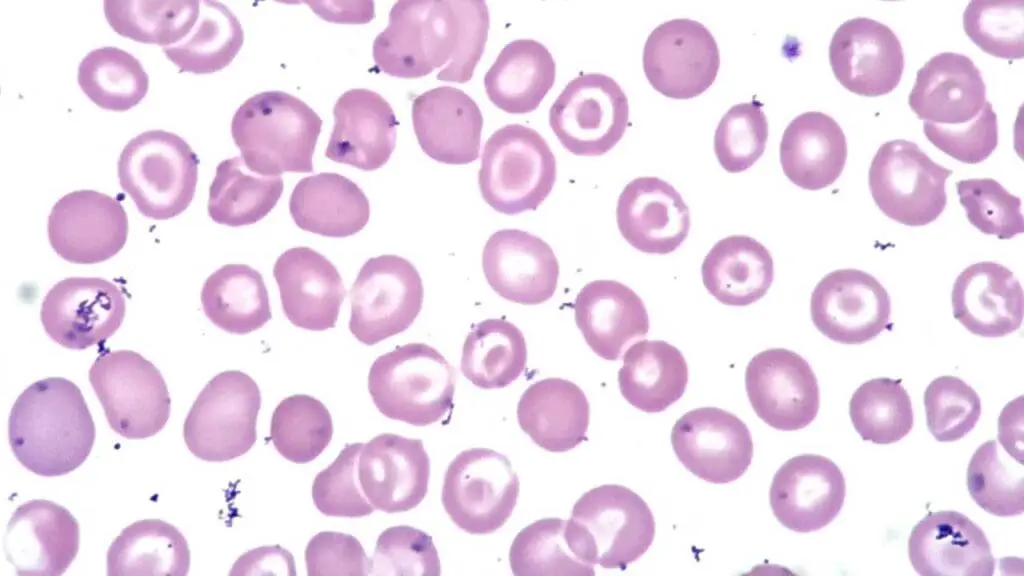
Polychromatic Red Cells (Polychromasia)
Polychromatic cells are young red cells (reticulocytes seen on a routine stain) that appear slightly larger and bluish-tinged due to residual RNA. Less than 1% in the bloodstream is normal.
An increase in polychromasia suggests the marrow is actively producing red cells. This is appropriate after acute blood loss, in hemolytic anemia, and during recovery from anemia treatment. Conversely, the absence of polychromasia in an anemic patient is itself a clue — it suggests a hypoproliferative state.
For the patient, the practical message is this: a smear showing polychromasia in the right clinical context is reassuring. It tells the clinician the marrow is still able to respond.
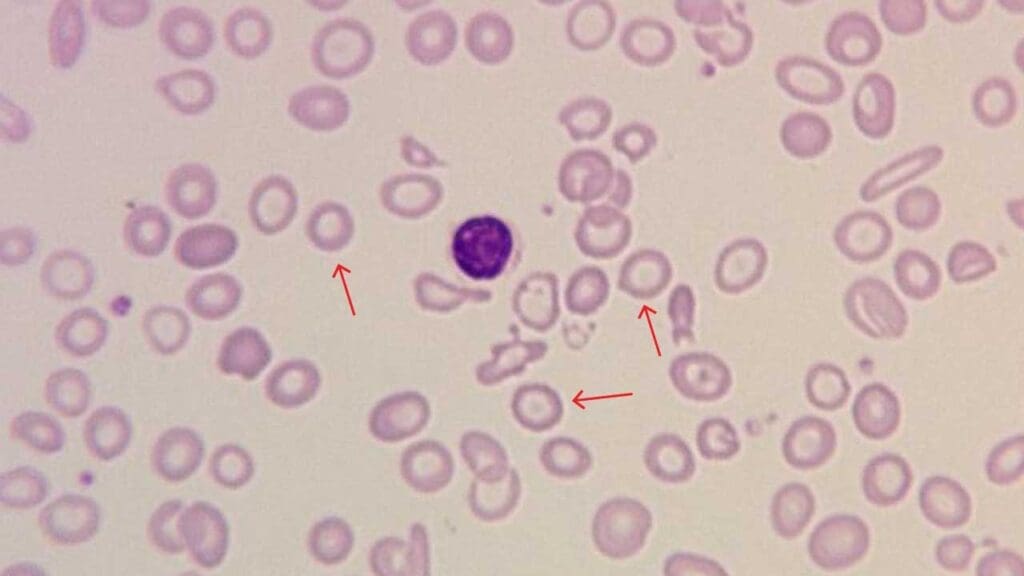
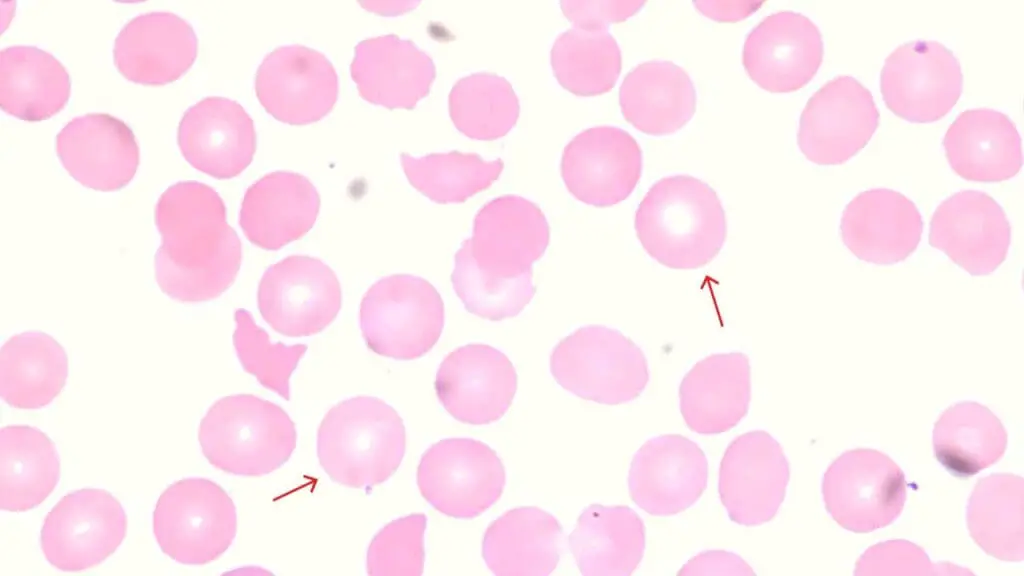
Target Cells
Target cells have a bullseye appearance: a central dense area, a pale ring, and a darker peripheral rim. They form when there is excess membrane relative to cell volume.
Causes: liver disease (especially obstructive jaundice), hemoglobinopathies (especially thalassemia), iron deficiency anemia (mild), post-splenectomy state, and lead poisoning.
Stomatocytes
Stomatocytes show a central slit-like pale area, like a small mouth.
Causes:
- Hereditary stomatocytosis — a rare inherited membrane defect.
- Liver disease, particularly alcoholic liver disease.
- Alcoholism.
- Certain drugs, including phenothiazines and chlorpromazine.
Important caveat
Stomatocytes are the most common artifact on a smear. Seen only in the thick part of the slide, they are usually meaningless. Treat them as significant only when present in the feathered edge or monolayer.
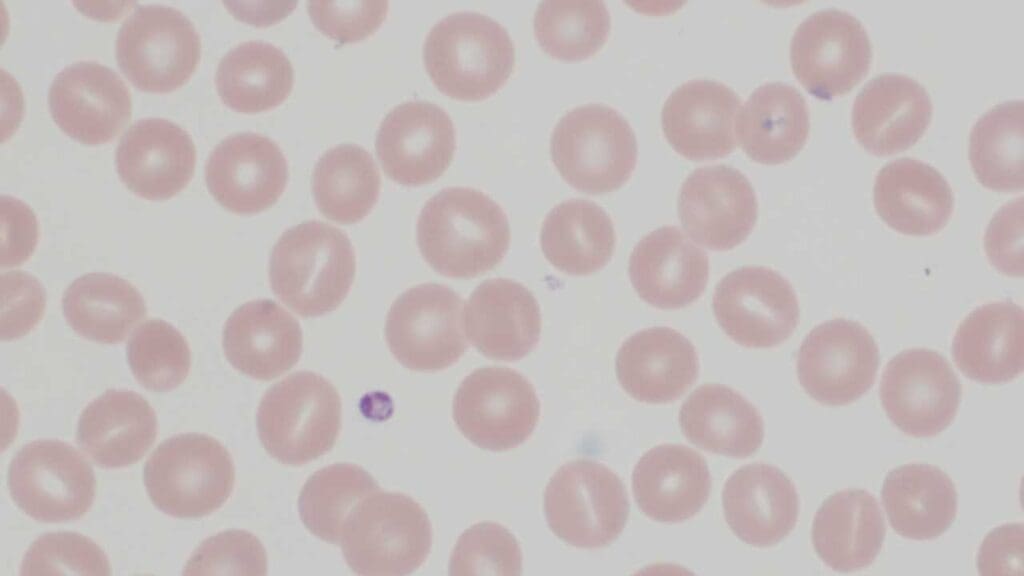
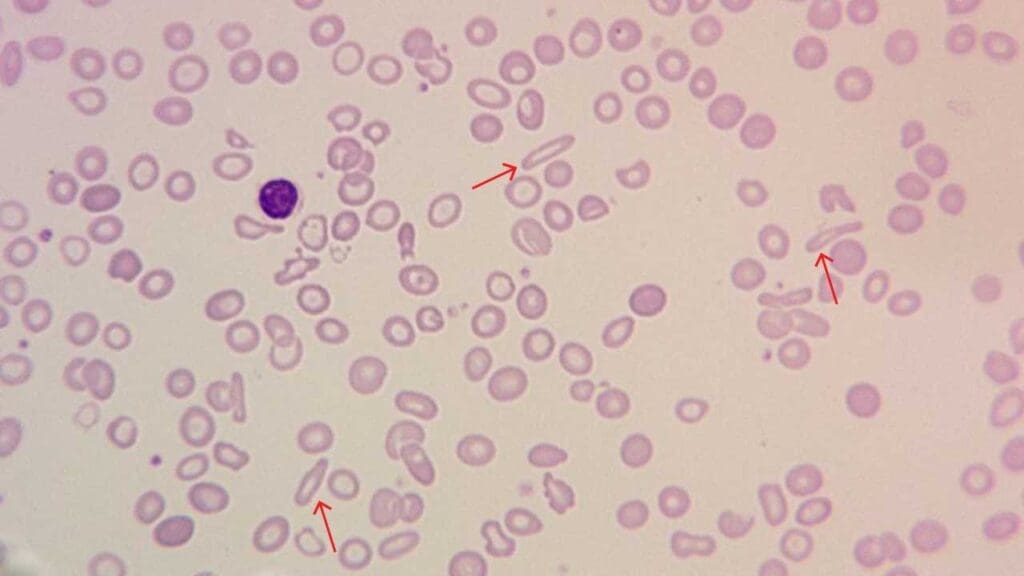
Pencil Cells
Pencil cells are markedly elongated, often microcytic, and frequently pale. They are most strongly associated with iron deficiency anemia but may also appear in thalassemia, lead poisoning, and sideroblastic anemia.
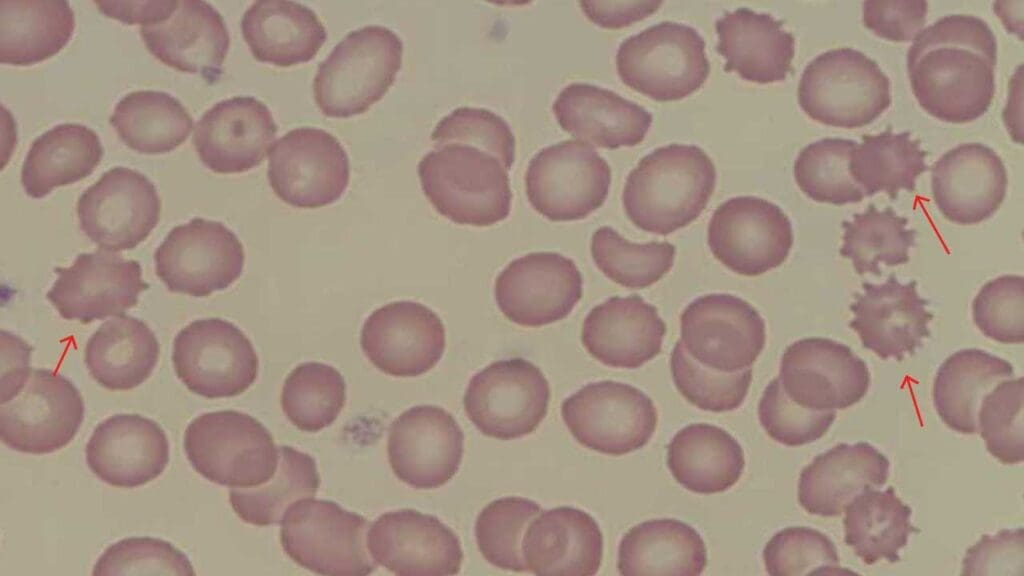
Echinocytes (Burr or Crenated Cells)
Echinocytes have numerous small, evenly spaced spikes around the cell.
Causes: artifact (very common — request a fresh smear if echinocytes are the only finding), uremia in kidney disease, severe liver disease, severe burns, sepsis, and electrolyte imbalances.
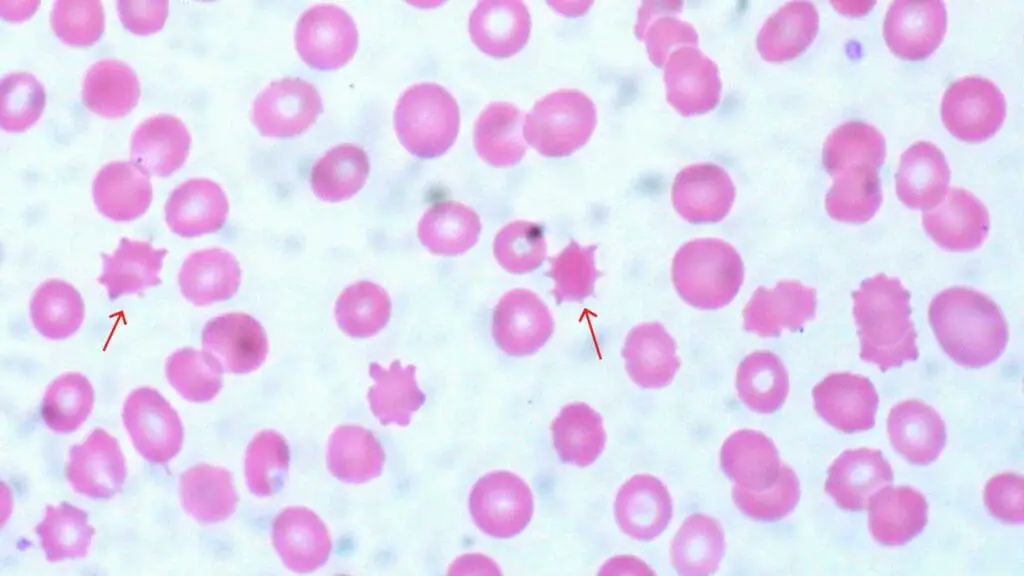
Acanthocytes
Acanthocytes have irregularly spaced, blunt-ended projections — fewer and more uneven than echinocytes.
Causes: abetalipoproteinemia (a rare lipid absorption disorder), severe liver disease (especially cirrhosis), neuroacanthocytosis syndromes, and certain medications (high-dose phenytoin, misoprostol).
Spherocytes
Spherocytes are nearly round and lack the central pallor. They are often slightly smaller than normal because the spherical shape reduces surface area.
Causes
- Hereditary spherocytosis — an inherited membrane disorder leading to chronic hemolysis. Confirmed by osmotic fragility or flow cytometry.
- Autoimmune hemolytic anemia (AIHA) — antibodies coat red cells, which are then partially eaten by splenic macrophages, producing spherocytes. The Direct Antiglobulin Test (Coombs) confirms the autoimmune nature.
- Hemolytic transfusion reactions — a medical emergency.
- Microangiopathic hemolytic anemia — schistocytes dominate but spherocytes can also appear.
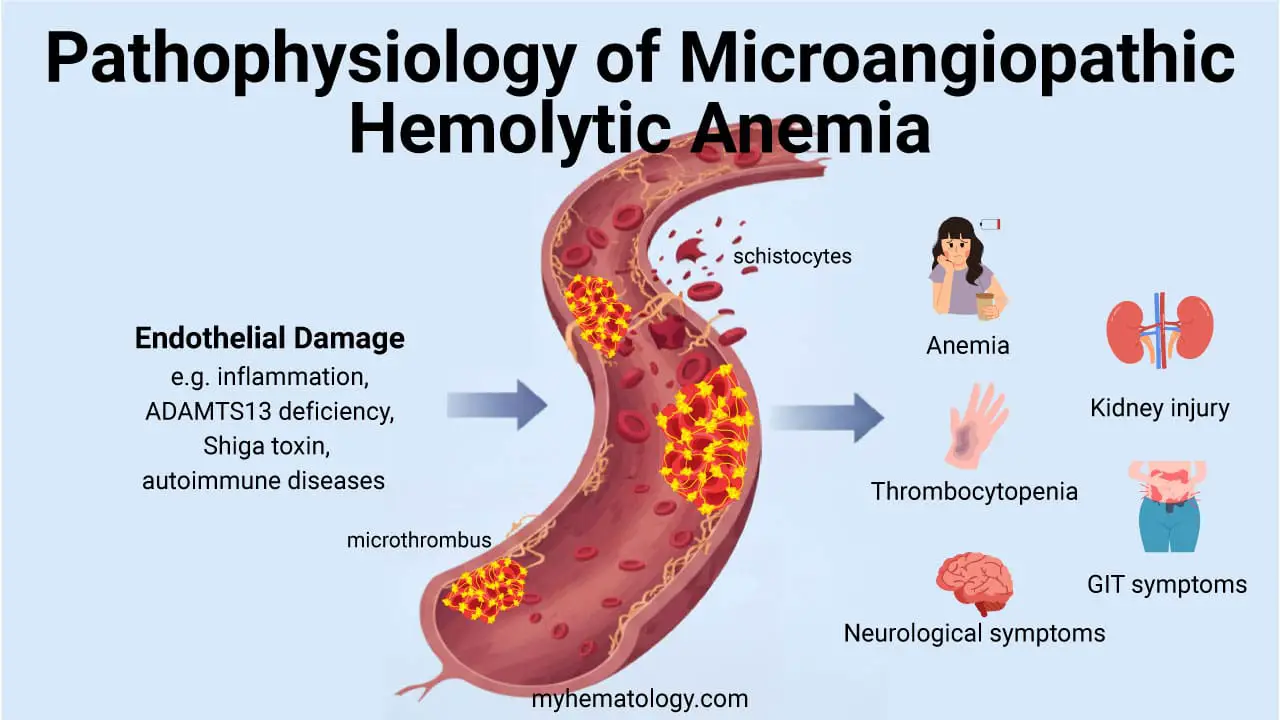
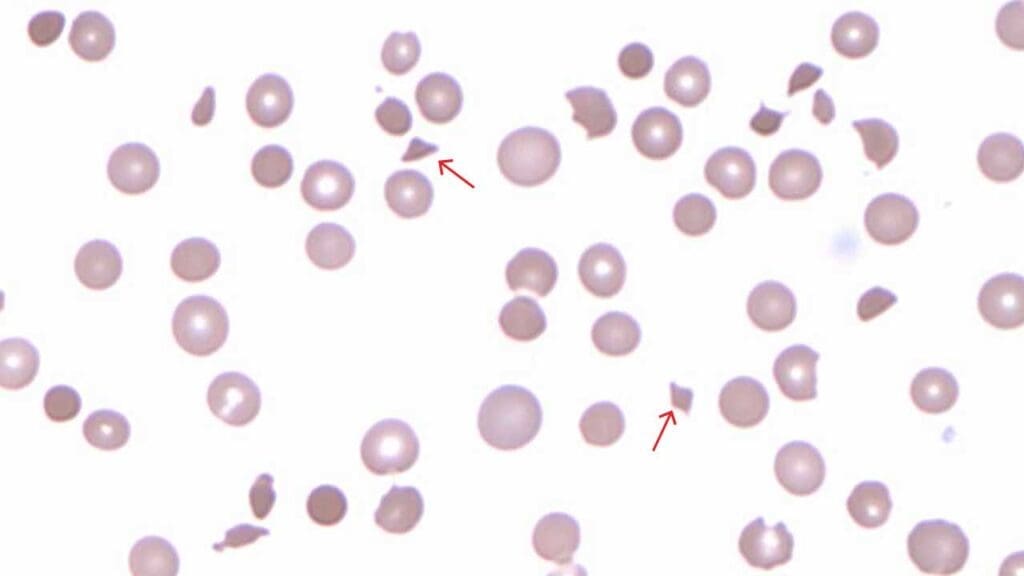
Schistocytes (Fragmented Cells)
Schistocytes are fragmented red cells: triangles, helmets, and small irregular pieces. They form when red cells are sheared by mechanical forces in the bloodstream.
Causes: microangiopathic hemolytic anemia (TTP, HUS, preeclampsia, DIC), mechanical heart valves, severe aortic stenosis, severe burns, and sickle cell disease during crises. Their presence often signals a serious, sometimes life-threatening, condition.
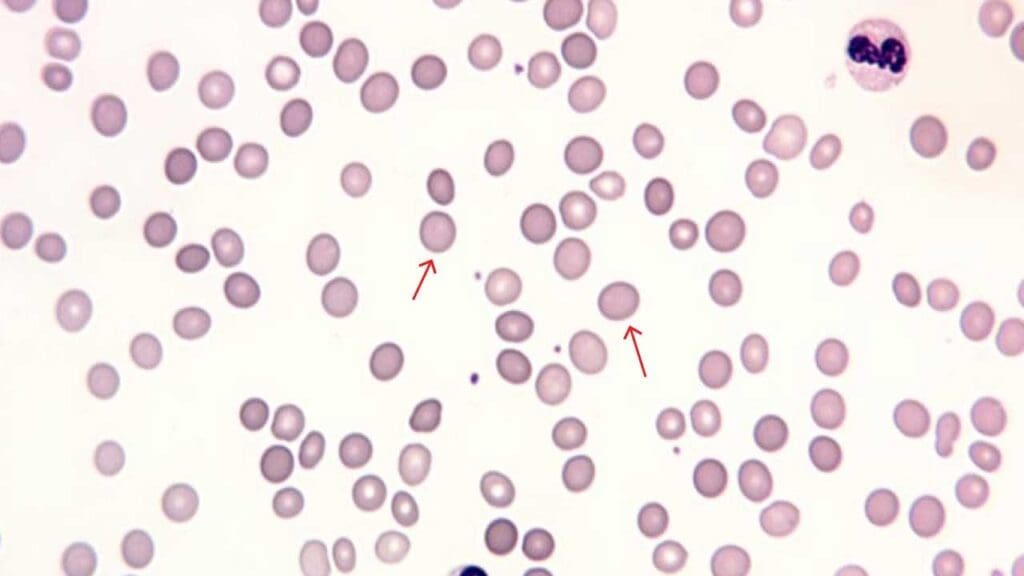
Elliptocytes
Elliptocytes are oval-shaped red cells.
Causes: hereditary elliptocytosis (often asymptomatic) and, less commonly, mild iron deficiency.
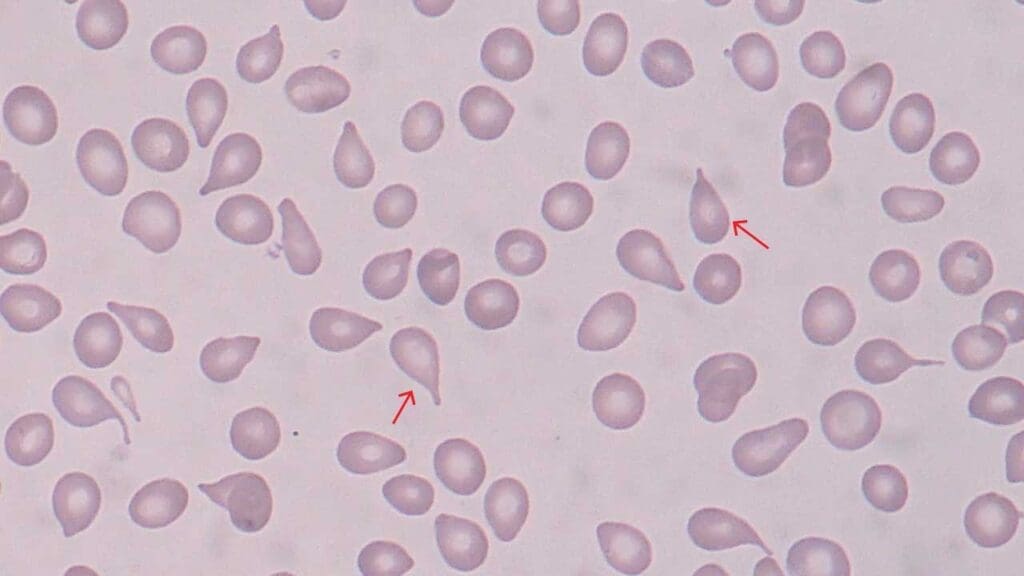
Tear Drop Poikilocytes (Tear Drop Cells)
Teardrop-shaped red cells with a narrow, blunt point at one end.
The primary clinical red flag for dacrocytes is myelofibrosis — and other space-occupying lesions of the marrow that cause extramedullary hematopoiesis. The cell "tears" as it tries to squeeze out of a fibrotic marrow [5]. This finding should immediately prompt a bone marrow biopsy.
Other causes: marrow infiltration by tumor or granuloma, vitamin B12 or folate deficiency (typically alongside macrocytosis and hypersegmented neutrophils).
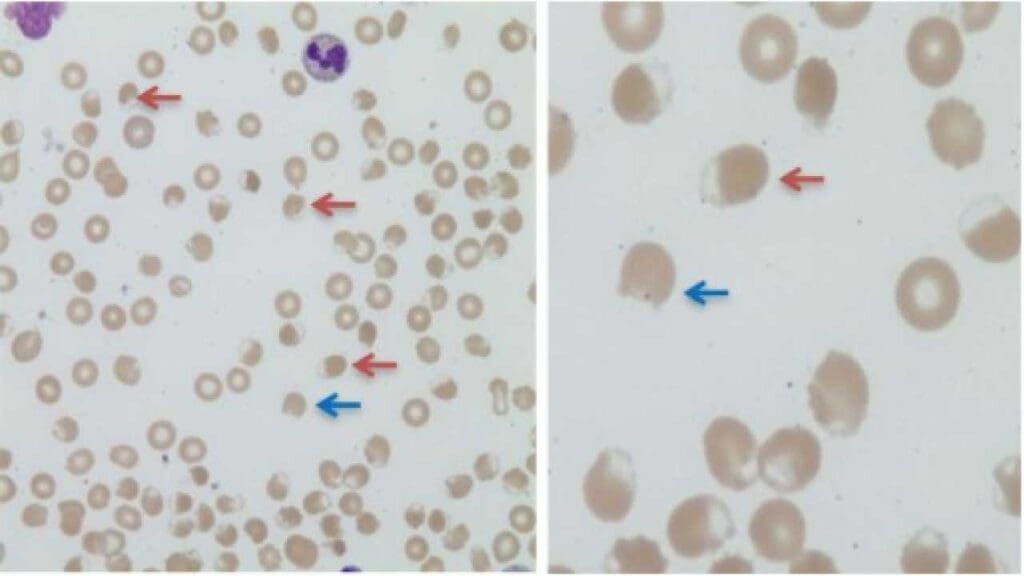
Bite Cells
Bite cells appear as red cells with a circular "bite" missing — the result of splenic macrophages removing Heinz bodies (denatured hemoglobin clumps).
Causes: G6PD deficiency (most common), other red cell enzyme deficiencies, oxidative drugs (dapsone, sulfasalazine, primaquine), and unstable hemoglobin variants.
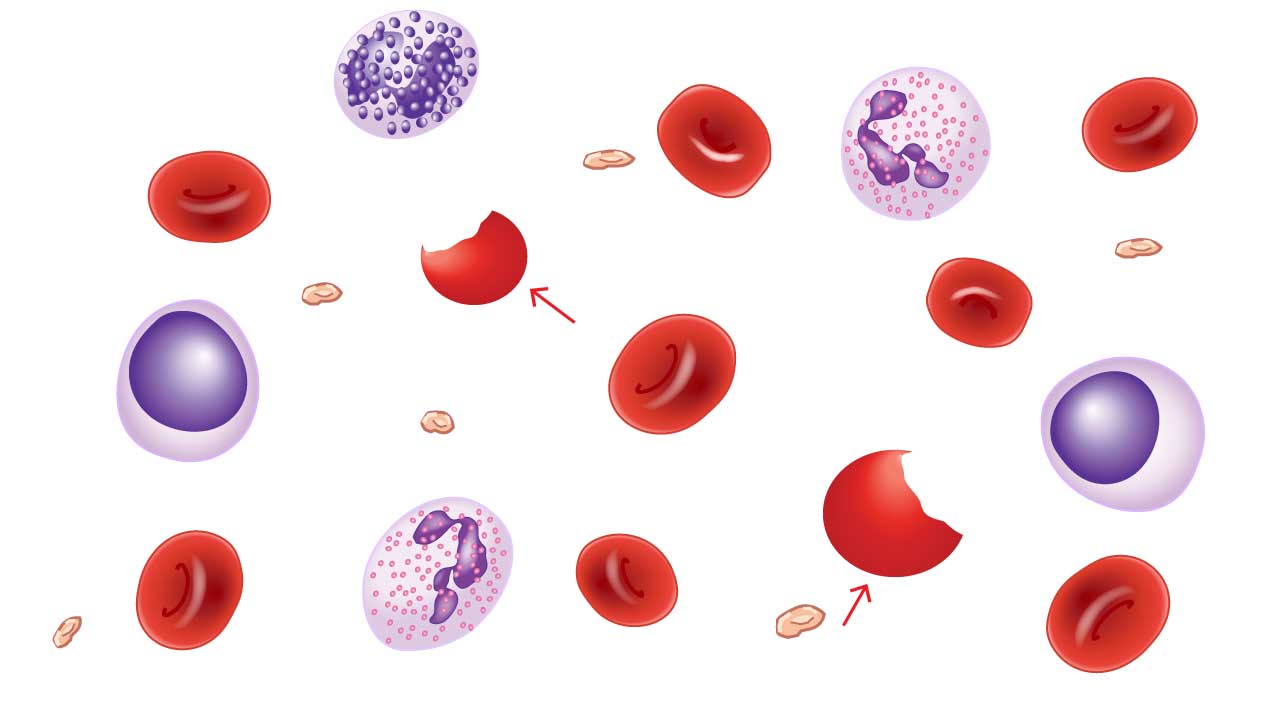
Blister Cells
Blister cells show a vacuole or "blister" pushed to one side, with hemoglobin displaced to the other side. They are precursors to bite cells.
Causes: the same as bite cells — predominantly oxidative damage from G6PD deficiency, oxidative drugs, unstable hemoglobinopathies, and occasionally microangiopathic hemolytic anemia.
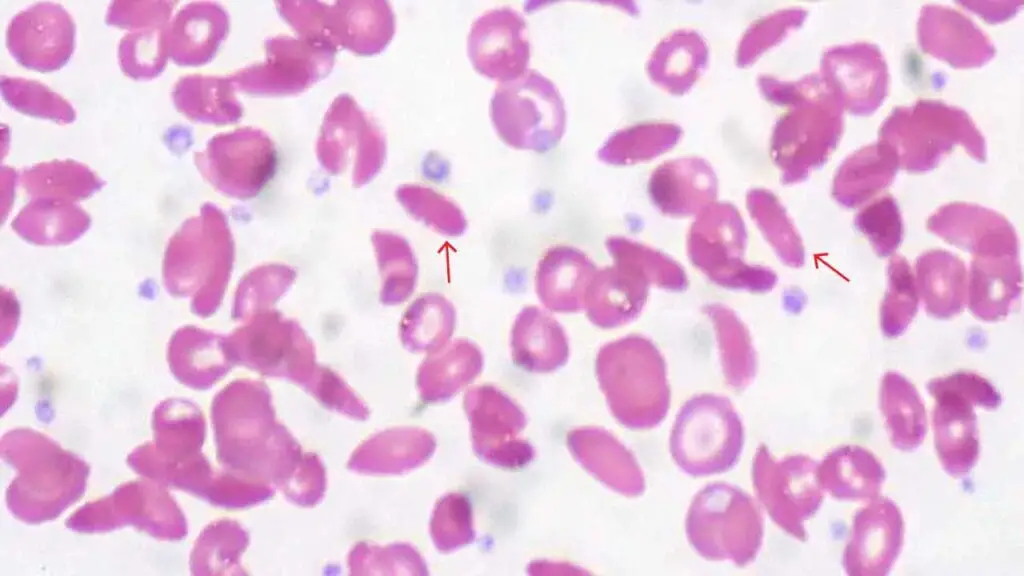
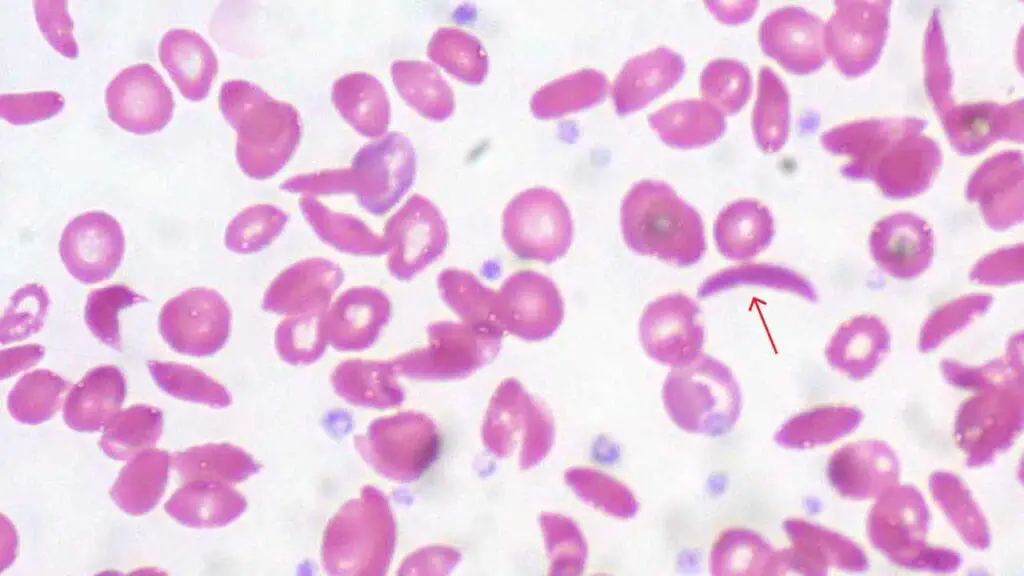
Sickle Cells
Sickle cells are crescent or C-shaped, formed when deoxygenated abnormal hemoglobin (HbS) polymerizes into rigid rods.
Cause: Sickle cell disease, an inherited mutation in the beta-globin gene. The clinical implications are major: lifelong hemolytic anemia, painful vaso-occlusive crises, and risk of stroke, acute chest syndrome, and kidney damage.
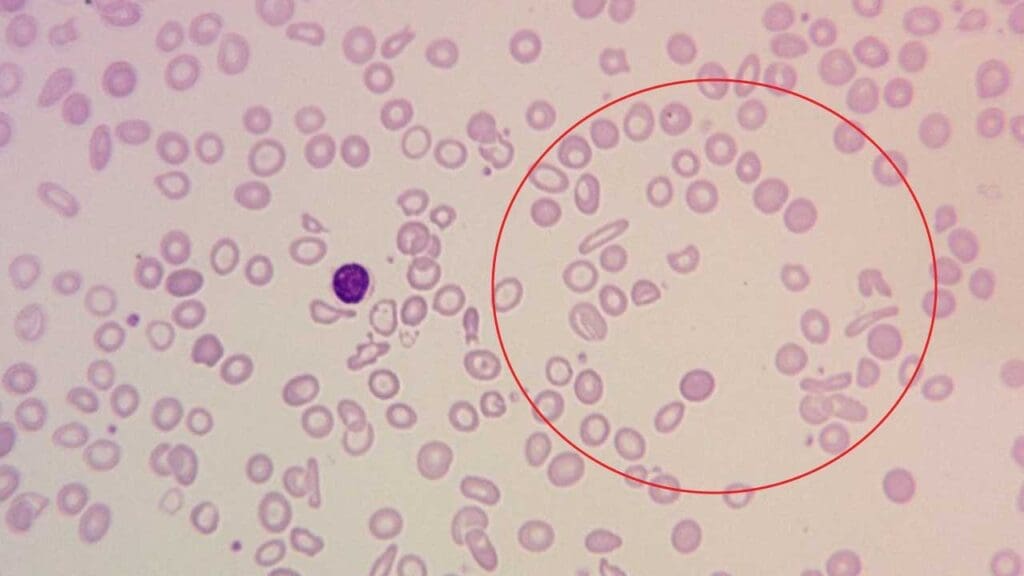
Anisopoikilocytosis
This term combines anisocytosis (size variation) and poikilocytosis (shape variation). It is not a single abnormality but a description of a smear showing both.
Causes: nutritional deficiencies (iron, B12, folate), bone marrow disorders (myelofibrosis, aplastic anemia), hemoglobinopathies, severe liver disease, splenomegaly, and significant blood loss.
The RDW is typically markedly elevated in anisopoikilocytosis, reflecting the size variation quantitatively.
Dimorphic Picture
A dimorphic blood picture shows two clearly distinct populations of red cells on the same smear — typically differing in size, sometimes also in color.
Causes:
- Post-transfusion sample — the classic teaching example. A patient with iron deficiency receives a transfusion, and the smear shows the patient's small, pale cells alongside the donor's normal-sized, normochromic cells.
- Mixed nutritional deficiencies — for example, simultaneous iron and folate deficiency.
- Treated iron deficiency — old microcytic cells alongside newly produced normocytic cells.
- Sideroblastic anemia.
- Bone marrow disorders such as MDS.
The RDW is significantly elevated in dimorphic pictures, which is one of the most reliable laboratory clues to this finding [7].
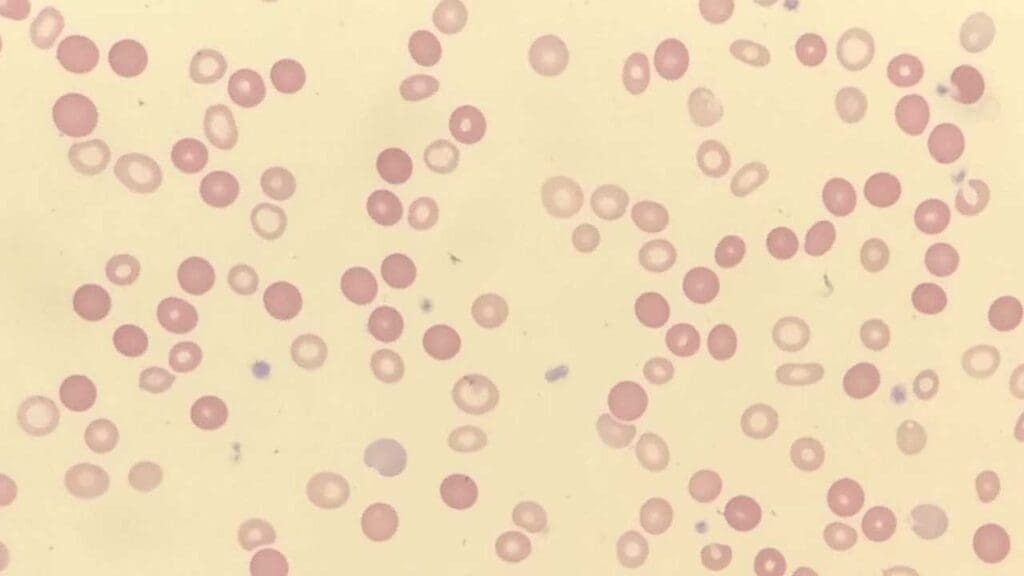
Rouleaux Formation
Rouleaux are stacks of red cells lined up like coins, caused by elevated plasma proteins reducing the repulsive forces between cells.
Causes:
- Multiple myeloma — a strong association. Marked rouleaux should prompt serum protein electrophoresis and consideration of a bone marrow biopsy.
- Inflammatory conditions — elevated fibrinogen.
- Autoimmune diseases.
- Dehydration — by concentrating plasma proteins.
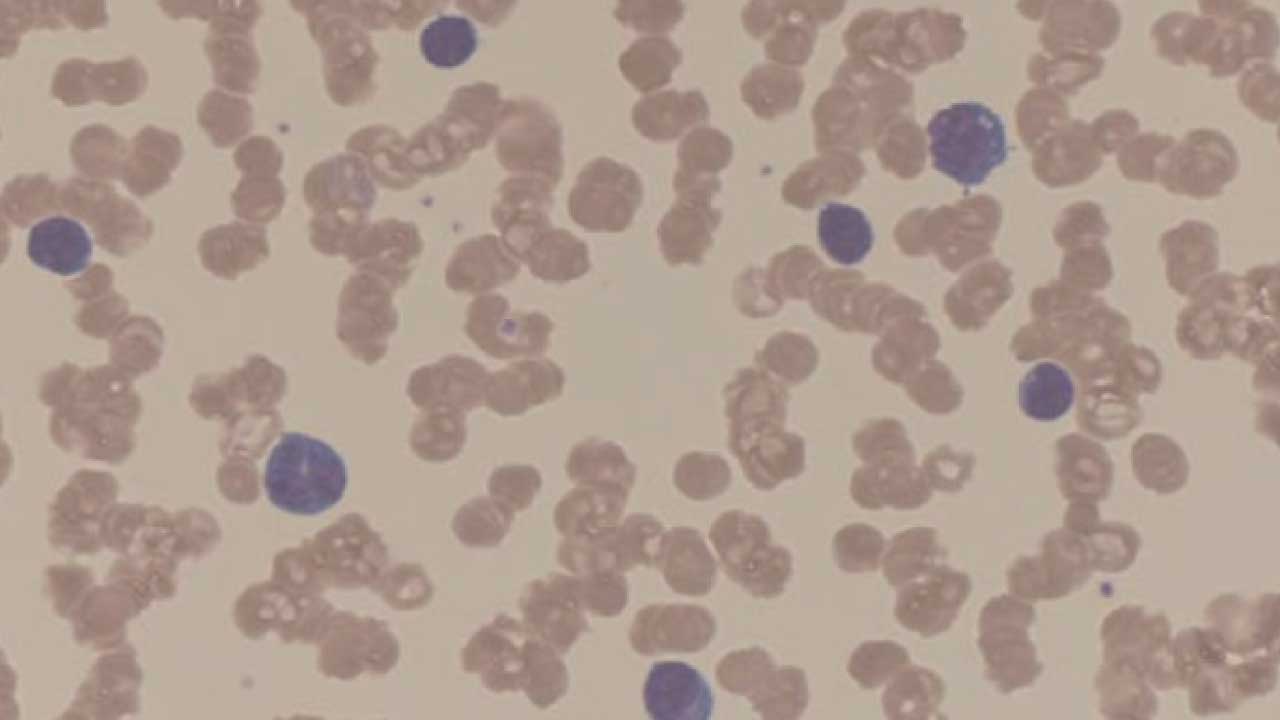
Red Cell Clumping (Agglutination)
Agglutination is irregular, dense clumping driven by antibodies binding to the red cell surface, distinct from the orderly stacks of rouleaux.
Causes: autoimmune hemolytic anemia (positive Coombs test), incompatible transfusion reactions, certain infections (Mycoplasma pneumoniae, Epstein-Barr virus), cold agglutinin disease, and some medications (quinine, certain cephalosporins).
Bringing It Together
Red blood cell morphology is one of the most cost-effective diagnostic tools available. A peripheral blood smear, paired with a CBC and a reticulocyte count, can narrow a differential diagnosis dramatically — sometimes faster than expensive molecular tests.
Frequently Asked Questions (FAQs)
What is red blood cell morphology and why does it matter?
Red blood cell morphology is the size, shape, and color of red cells seen under a microscope on a peripheral blood smear. It matters because abnormal morphology is often the first clue to an underlying disease, including anemia, liver disease, bone marrow disorders, and inherited hemoglobin conditions. Morphology turns a blood smear into a diagnostic snapshot that complements CBC numbers.
What is the difference between polychromasia and a reticulocyte?
A reticulocyte is a young red blood cell, identified specifically with a supravital stain like new methylene blue. Polychromasia is what those same young cells look like on a routine Wright-Giemsa stain — slightly larger and bluish-tinged because of residual RNA. The terms are often used interchangeably, but morphology (polychromasia) only gives a visual impression, while a reticulocyte count gives the actual number, which tells you whether the bone marrow is responding adequately to anemia.
What does a high RDW mean?
RDW, or Red Cell Distribution Width, measures how much red blood cell sizes vary in a sample. A high RDW means the cells are unequal in size (anisocytosis). It is commonly elevated in iron deficiency anemia, mixed nutritional deficiencies, and dimorphic pictures (such as after a blood transfusion). RDW is often normal in thalassemia trait, which helps distinguish it from iron deficiency.
Why are teardrop cells (dacrocytes) considered a red flag?
Teardrop cells are most strongly associated with myelofibrosis, a condition where the bone marrow becomes scarred. The cells "tear" as they squeeze through fibrotic marrow during extramedullary hematopoiesis. Their presence should prompt urgent investigation, including a bone marrow biopsy, to look for myelofibrosis or marrow infiltration by tumor or granuloma.
How can I tell if a "weird" cell shape is real or just an artifact?
True pathology is scattered and varies in intensity across the smear — some fields show the abnormal cells, others show fewer. An artifact is usually uniform: if every single cell in a field looks the same way (for example, every cell is a stomatocyte), it's almost always a slide-preparation issue. Stomatocytes seen only in the thick part of the smear are usually meaningless; they are clinically significant only when found in the feathered edge or monolayer.
What is a dimorphic blood picture and when does it occur?
A dimorphic blood picture means there are two clearly different populations of red blood cells on the smear, typically differing in size or color. It is classically seen after a blood transfusion in a patient with iron deficiency, where small pale cells (the patient's) are mixed with larger normal cells (the donor's). It can also appear in mixed nutritional deficiencies, treated iron deficiency, and bone marrow disorders such as MDS or sideroblastic anemia. The RDW is usually markedly elevated.
Glossary of Related Medical Terms
- Anisocytosis — variation in red blood cell size on a smear.
- Poikilocytosis — variation in red blood cell shape on a smear.
- Erythropoiesis — the production of red blood cells in the bone marrow.
- Reticulocyte — a young red blood cell, identified specifically with a supravital stain like new methylene blue.
- Polychromasia — bluish-pink staining of young red cells on a standard Wright-Giemsa stain, reflecting residual RNA.
- RDW (Red Cell Distribution Width) — a CBC parameter that quantifies how much red cell sizes vary; the laboratory measure of anisocytosis.
- MCV (Mean Corpuscular Volume) — the average volume of a red blood cell, reported in femtoliters (fL); used to classify anemias as microcytic (<80 fL), normocytic (80–100 fL), or macrocytic (>100 fL).
- Hepcidin — a hormone made by the liver that locks iron inside macrophages during inflammation, making it unavailable for new red cell production.
- Hemolysis — the destruction of red blood cells, either inside blood vessels (intravascular) or in the spleen and liver (extravascular).
- Hypoxia — insufficient oxygen reaching the body's tissues.
- Hypoproliferative anemia — anemia where the bone marrow is not producing enough red cells (low reticulocyte count).
- Hyperproliferative anemia — anemia where the bone marrow is responding by making more red cells, usually because cells are being destroyed or lost (high reticulocyte count).
- Extramedullary hematopoiesis — blood cell production happening outside the bone marrow, typically in the spleen or liver, often when marrow is fibrotic.
- Myelofibrosis — scarring of the bone marrow, which forces cells to "squeeze out" through fibrotic tissue, producing teardrop-shaped cells.
- Heinz bodies — clumps of denatured hemoglobin inside red cells, often from oxidative damage; visible only with special stains.
- Hemoglobinopathy — an inherited disorder of hemoglobin structure or production (e.g., sickle cell disease, thalassemia).
- Splenectomy — surgical removal of the spleen.
- Cold agglutinin — an antibody that causes red blood cells to clump at lower temperatures.
Disclaimer: This article is intended for informational purposes only and is specifically targeted towards medical students. It is not intended to be a substitute for informed professional medical advice, diagnosis, or treatment. While the information presented here is derived from credible medical sources and is believed to be accurate and up-to-date, it is not guaranteed to be complete or error-free. See additional information.
References
- Khoury, J. D., Solary, E., Abla, O., Akkari, Y., Alaggio, R., Apperley, J. F., Bejar, R., Berti, E., Busque, L., Chan, J. K. C., Chen, W., Chen, X., Chng, W. J., Choi, J. K., Colmenero, I., Coupland, S. E., Cross, N. C. P., De Jong, D., Elghetany, M. T., Takahashi, E., … Hochhaus, A. (2022). The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia, 36(7), 1703–1719. https://doi.org/10.1038/s41375-022-01613-1
- Arber, D. A., Orazi, A., Hasserjian, R. P., Borowitz, M. J., Calvo, K. R., Kvasnicka, H. M., Wang, S. A., Bagg, A., Barbui, T., Branford, S., Bueso-Ramos, C. E., Cortes, J. E., Dal Cin, P., DiNardo, C. D., Dombret, H., Duncavage, E. J., Ebert, B. L., Estey, E. H., Facchetti, F., Foucar, K., … Tefferi, A. (2022). International Consensus Classification of Myeloid Neoplasms and Acute Leukemias: integrating morphologic, clinical, and genomic data. Blood, 140(11), 1200–1228. https://doi.org/10.1182/blood.2022015850
- Ganz, T., & Nemeth, E. (2012). Hepcidin and iron homeostasis. Biochimica et biophysica acta, 1823(9), 1434–1443. https://doi.org/10.1016/j.bbamcr.2012.01.014
- Weiss, G., Ganz, T., & Goodnough, L. T. (2019). Anemia of inflammation. Blood, 133(1), 40–50. https://doi.org/10.1182/blood-2018-06-856500
- Tefferi, A., & Pardanani, A. (2015). Myeloproliferative Neoplasms: A Contemporary Review. JAMA oncology, 1(1), 97–105. https://doi.org/10.1001/jamaoncol.2015.89
- Green, R., & Datta Mitra, A. (2017). Megaloblastic Anemias: Nutritional and Other Causes. The Medical clinics of North America, 101(2), 297–317. https://doi.org/10.1016/j.mcna.2016.09.013
- Salvagno, G. L., Sanchis-Gomar, F., Picanza, A., & Lippi, G. (2015). Red blood cell distribution width: A simple parameter with multiple clinical applications. Critical reviews in clinical laboratory sciences, 52(2), 86–105. https://doi.org/10.3109/10408363.2014.992064
- Ford J. (2013). Red blood cell morphology. International journal of laboratory hematology, 35(3), 351–357. https://doi.org/10.1111/ijlh.12082