Key Takeaways
An abnormal WBC on a peripheral blood smear is a white blood cell with shape, staining, or structural changes that differ from a healthy leukocyte. These changes give clues to infection, nutritional deficiency, inherited disorders, drug effects, or bone marrow disease.
- Cytoplasmic clues include toxic granulation (severe bacterial infection), Döhle bodies (inflammation), Alder-Reilly bodies (lysosomal storage disorders), vacuoles (drug toxicity, metabolic disorders, sepsis), and Auer rods (AML, especially APL).
- Nuclear clues include hypersegmentation (B12 or folate deficiency), left shift (acute infection or marrow stress), and Pelger-Huët anomaly (benign genetic trait — but the acquired pseudo-Pelger-Huët form points to dysplasia or drug effect).
- Reactive lymphocytes usually signal a viral infection, but persistent or unusual forms warrant flow cytometry to exclude leukemia or lymphoma.
- Modern labs increasingly pair manual review with digital morphology analyzers and AI pre-classification, which improves sensitivity for spotting abnormal WBCs but does not replace the trained eye [7,8].
*Click ▾ for more information
Introduction
When a blood smear is reviewed under the microscope, the white blood cells tell a story. Their size, nuclear shape, granule pattern, and staining together hint at what is happening inside the patient. An abnormal WBC is rarely a diagnosis on its own. It is a clue. Sometimes the clue points to a simple infection. Sometimes it points to a genetic disorder a child has lived with since birth. Sometimes it points to bone marrow failure or leukemia.
This article walks through the most important morphological changes in a hematology report. Each section explains what the cell looks like, why it looks that way, and which follow-up investigations the change usually prompts.
A Brief Refresher on WBCs
White blood cells, or leukocytes, are the body's immune sentinels. Five main types do most of the work. Neutrophils are the first responders to bacterial infection. Lymphocytes (B cells, T cells, and NK cells) handle viral defense and antibody production. Monocytes mature into tissue macrophages and present antigens. Eosinophils fight parasites and drive allergic responses. Basophils release histamine in allergy and inflammation.
A complete blood count gives a total WBC number, but it cannot tell which type is rising or whether the cells look normal. That is what the smear adds.
Why Morphology Still Matters
A differential count tells you the percentage of each WBC type. Morphology tells you whether those cells are healthy. Together they can:
- Suggest a diagnosis when other labs are inconclusive — for example, hypersegmented neutrophils alongside a macrocytic anemia point to B12 deficiency [4].
- Confirm a clinical suspicion, such as bacterial sepsis backed up by toxic granulation and a left shift.
- Guide treatment, since neutrophilia points toward antibiotics while eosinophilia points toward allergy or parasites.
- Monitor response, as morphology often normalizes when the underlying cause is treated.
Modern labs now combine the trained eye with digital morphology analyzers such as the Sysmex DI-60 and Mindray MC-100i. These instruments pre-classify cells using artificial intelligence, then a technologist verifies the call. AI pre-classification improves sensitivity for catching abnormal WBCs and shortens review time, but performance is weaker for rare cells and basophils, so manual review remains essential [7,8].
Cytoplasmic Inclusions
Toxic Granulation in Neutrophils
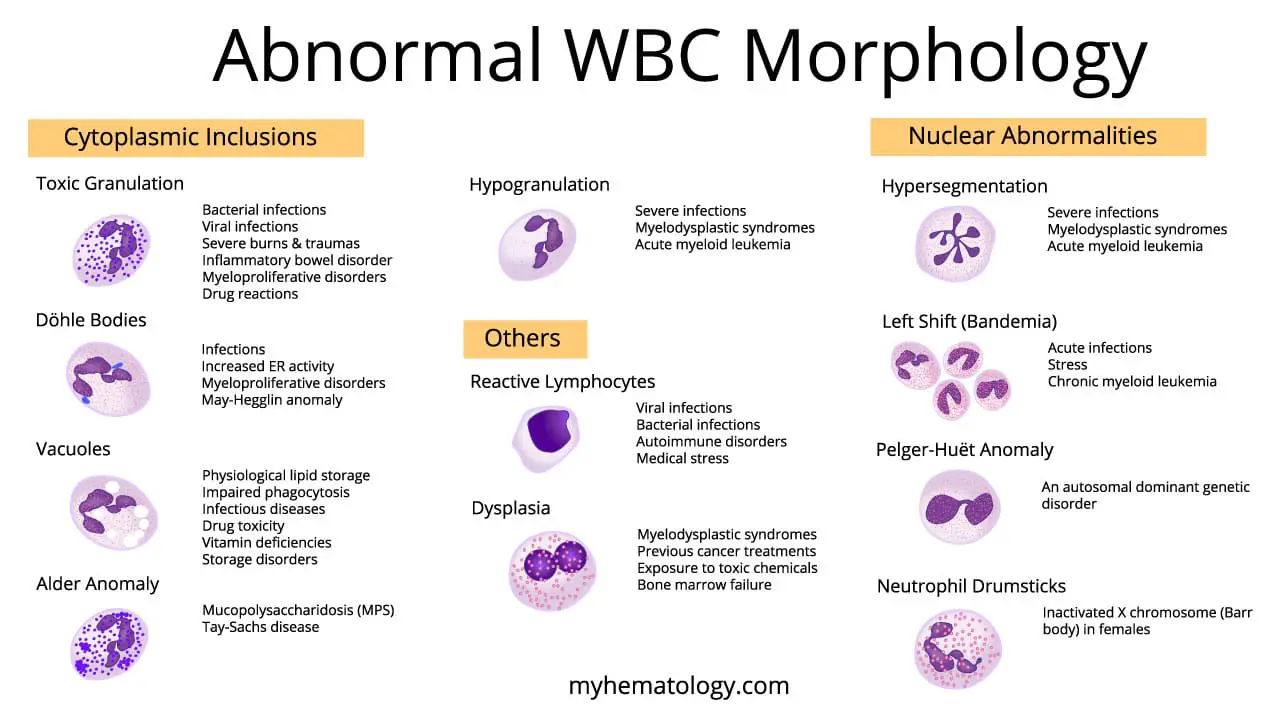
Toxic granulation refers to coarse, dark-staining granules inside neutrophils. They form when the bone marrow speeds up production so much that primary (azurophilic) granules are incompletely packaged.
What it looks like. The granules are larger, denser, and stain a darker blue or purple than the fine pink granules of a healthy mature neutrophil. They are best seen with a Romanowsky stain such as Leishman or Wright-Giemsa.
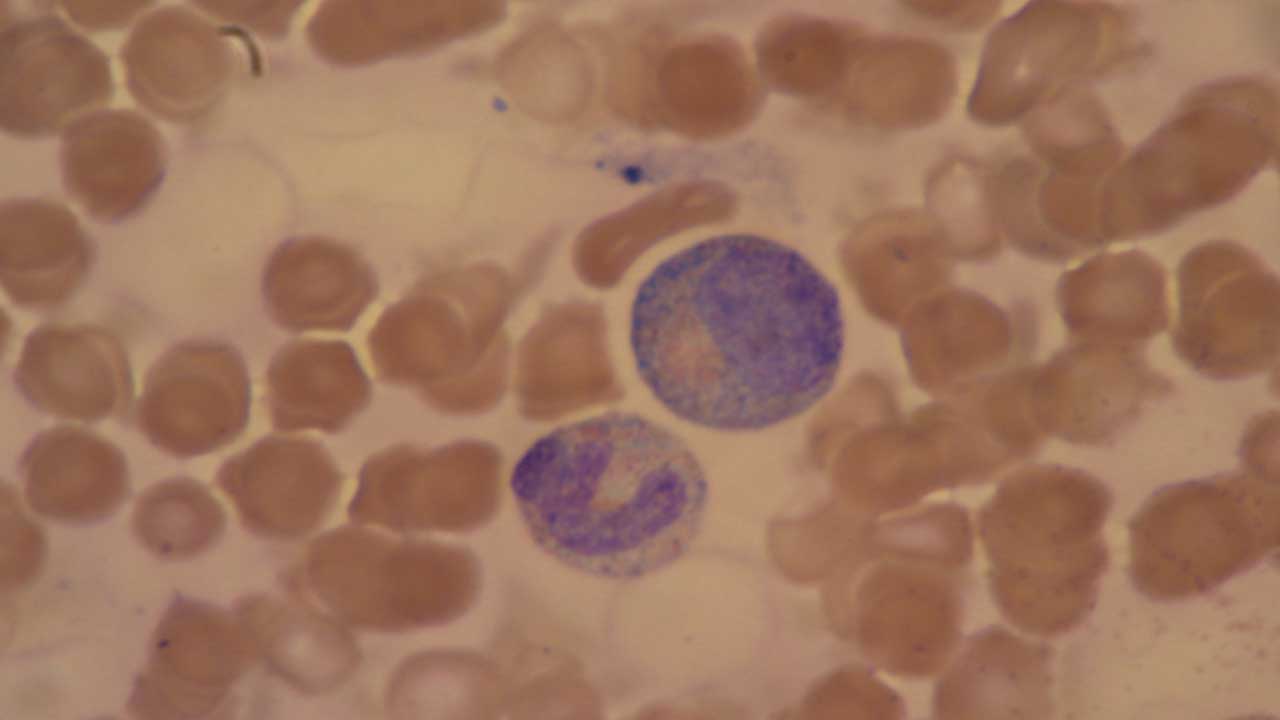
Common causes.
- Severe bacterial infection, especially sepsis, pneumonia, and meningitis.
- Severe viral infection in some cases.
- Tissue injury from burns, trauma, or surgery.
- Active inflammatory bowel disease.
- Granulocyte colony-stimulating factor (G-CSF) therapy and certain chemotherapy regimens.
Why it matters. Toxic granulation signals an active immune or inflammatory response. It is most useful when interpreted with the clinical picture, the white cell count, and the presence or absence of band cells.
When to investigate further. Blood cultures, CRP, procalcitonin, and source-specific imaging or serology are the usual next steps.
Döhle Bodies in Neutrophils
Döhle bodies are small, pale blue-gray oval inclusions sitting near the edge of the neutrophil cytoplasm. They are aggregates of rough endoplasmic reticulum, which contains ribosomal RNA — this is why older texts describe them as "agglutinated ribosomes" and newer texts as "RER remnants." Both views point to the same structure.
What it looks like. Round to oval, 1–3 micrometers across, located close to the cell membrane, and staining pale blue to gray.
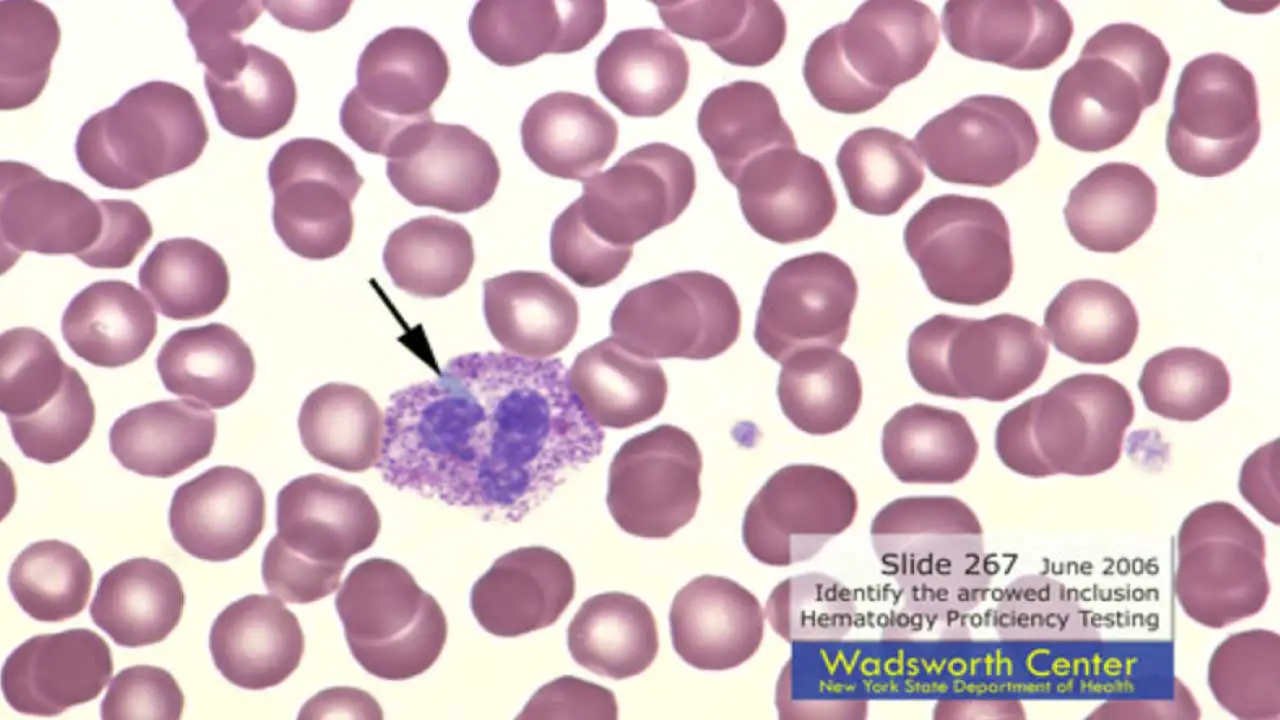
Common causes.
- Bacterial and viral infection.
- Severe burns, trauma, and pregnancy.
- Drug reactions and B12 or folate deficiency.
- The May-Hegglin anomaly, an inherited MYH9 mutation that produces giant platelets, mild thrombocytopenia, and large Döhle-like inclusions.
Why it matters. Like toxic granulation, Döhle bodies are part of the "toxic change" pattern of inflammation. Alone they are not diagnostic. With giant platelets and a family history, however, they raise the suspicion of May-Hegglin anomaly.
Toxic Granulation vs. Döhle Bodies at a Glance
| Feature | Toxic Granulation | Döhle Bodies |
| Composition | Incompletely packaged primary granules | Rough ER with ribosomes |
| Appearance | Coarse dark granules throughout cytoplasm | Pale blue ovals at cell edge |
| Cause | Severe infection, inflammation, G-CSF | Infection, burns, pregnancy, May-Hegglin |
| Significance | Strong sign of active inflammation | Component of toxic change; may signal inherited disorder if persistent |
Vacuolation in Abnormal WBCs
Vacuoles are clear, round, unstained spaces in the cytoplasm. They appear empty under the microscope because they hold fluid or undigested material that does not take up stain.
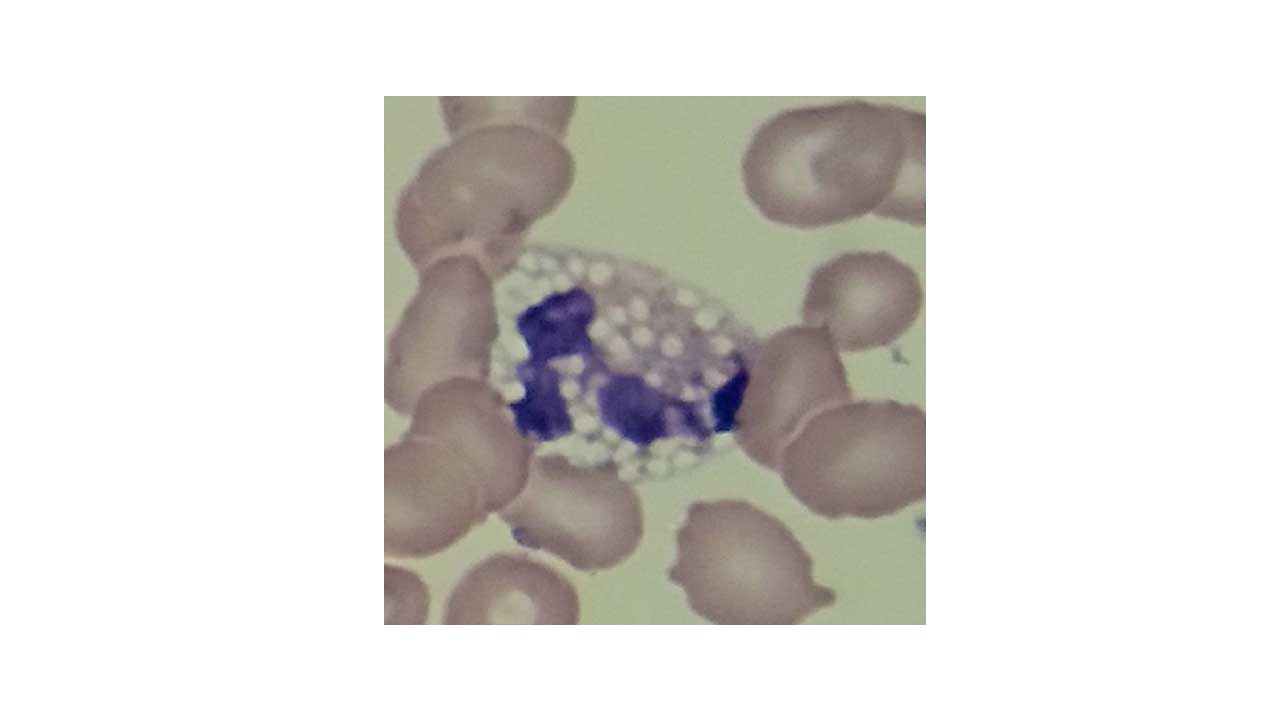
Common causes
- Active phagocytosis or sepsis. Neutrophils develop vacuoles after engulfing bacteria or in the setting of endotoxemia.
- Drug toxicity, especially from chemotherapy.
- Vitamin B12 or folate deficiency.
- Lysosomal storage disorders such as Gaucher disease, Niemann-Pick disease, and Chanarin-Dorfman syndrome [6]. Vacuolated lymphocytes in a child are a major clue.
- Chédiak-Higashi syndrome, in which giant lysosomal granules form because of impaired vesicle fusion.
Why it matters. In neutrophils, vacuolation correlates with severity in sepsis and systemic inflammatory response syndrome. In children, vacuolated lymphocytes should trigger evaluation for an inherited metabolic disorder.
When to investigate further. Blood cultures and lactate in suspected sepsis; urine glycosaminoglycans, white cell enzyme assays, and genetic testing in suspected storage disorders.
Alder-Reilly Anomaly
Alder-Reilly anomaly (sometimes called Alder anomaly) is a rare inherited finding. Coarse, darkly staining metachromatic granules — called Reilly bodies — sit in the cytoplasm of granulocytes, monocytes, and lymphocytes. The granules contain partially digested mucopolysaccharides that cells cannot break down because a lysosomal enzyme is missing.
What it looks like. Large, coarse, basophilic granules, most prominent in neutrophils but also visible in eosinophils, basophils, monocytes, and lymphocytes. Granules may be surrounded by a clear halo, which is best seen in lymphocytes.
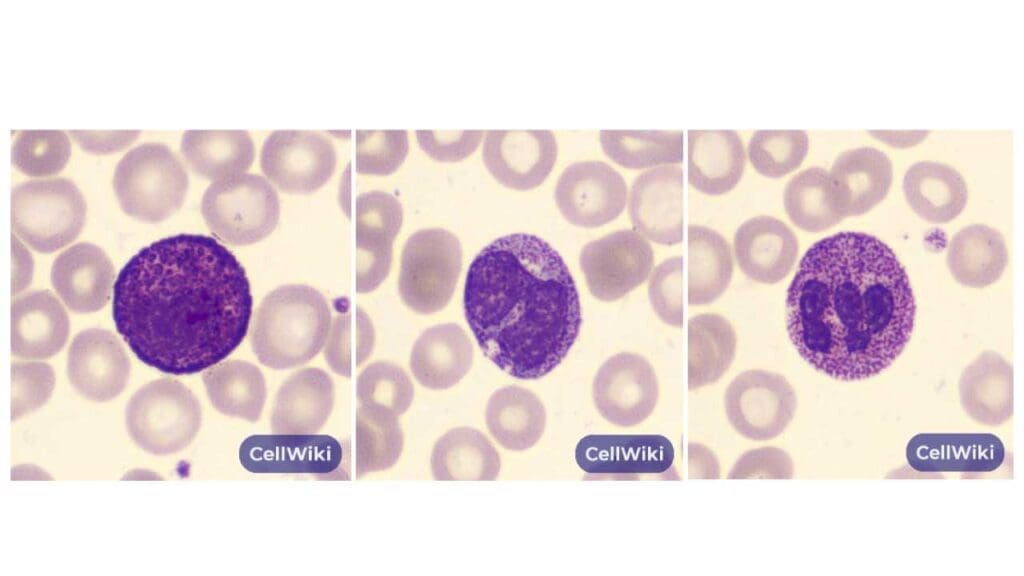
Cause and inheritance. Alder-Reilly bodies are most often associated with the mucopolysaccharidoses (MPS) — a group of lysosomal storage disorders that are mostly autosomal recessive, except for MPS II (Hunter syndrome), which is X-linked recessive. Tay-Sachs disease can also cause the finding.
Why it matters. The granulocytes themselves work normally. The value of the finding is that it is a non-invasive clue to a serious underlying genetic disorder. Reilly bodies can be the first hint that prompts enzyme assays and a confirmed diagnosis [9].
Common pitfall. Alder-Reilly bodies are easily confused with toxic granulation, but two clues separate them. Reilly bodies appear in all leukocyte lineages, not just neutrophils, and they are permanent rather than transient.
Auer Rods and Faggot Cells
Auer rods are needle-shaped, pink-red crystalline structures in the cytoplasm of myeloid blasts. They form when azurophilic granules fuse abnormally.
What it looks like. Slim, straight or slightly curved pink rods inside immature myeloid cells. When multiple rods bundle together inside a single promyelocyte, the cell is called a faggot cell.
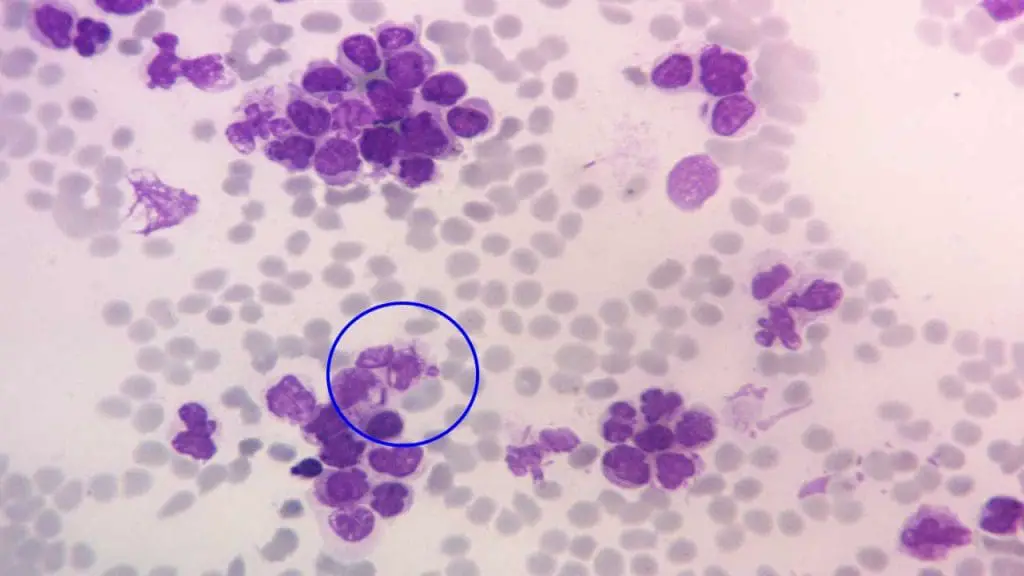
Why it matters. Auer rods are pathognomonic for myeloid lineage and signal acute myeloid leukemia (AML). Faggot cells are a hallmark of acute promyelocytic leukemia (APL), a hematologic emergency because of its risk of disseminated intravascular coagulation. Suspected APL requires immediate flow cytometry, PML-RARA testing, and initiation of all-trans retinoic acid (ATRA) [1].
Hypogranularity in WBCs
Hypogranularity is a reduction or absence of the normal cytoplasmic granules in granulocytes, most noticeable in neutrophils.
What it looks like. Granulocytes whose cytoplasm appears clear, pale, or only sparsely speckled.
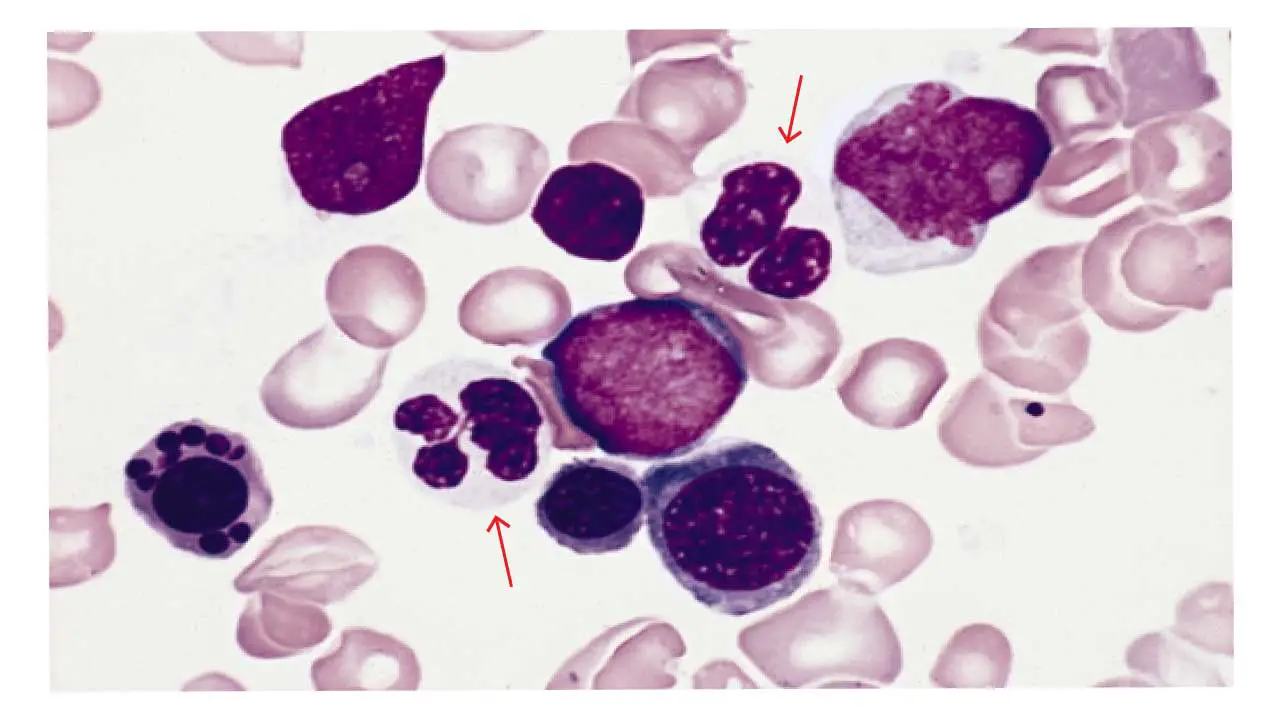
Common causes.
- Myelodysplastic neoplasms (MDS) and AML — hypogranularity is one of the hallmark dysplastic features.
- B12 or folate deficiency.
- Severe infection releasing immature neutrophils.
Why it matters. Hypogranularity is one of the strongest cytological markers of dysplasia. Its diagnostic weight goes up when it appears with other dysplastic features such as pseudo-Pelger-Huët cells and nuclear abnormalities [1,2].
Nuclear Abnormalities
Hypersegmented Neutrophils
A healthy neutrophil has two to five nuclear lobes connected by thin chromatin strands. A hypersegmented neutrophil has six or more.

Common causes.
- Megaloblastic anemia from vitamin B12 or folate deficiency. Disrupted DNA synthesis allows the nucleus to keep dividing.
- Myelodysplastic neoplasms with disordered nuclear maturation.
- Severe liver disease.
- Hydroxyurea therapy, used in sickle cell disease and CML.
Why it matters. Hypersegmented neutrophils alongside macrocytic red cells form one of the most teachable, recognizable patterns in hematology. The combination should prompt serum B12, folate, and a reticulocyte count [4].
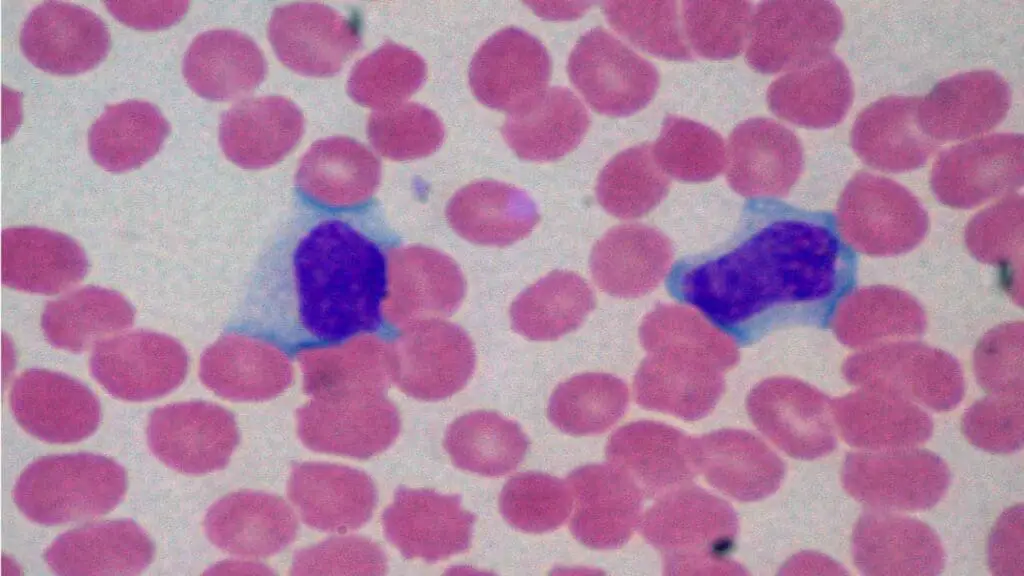
Left Shift (Bandemia)
A left shift is the appearance of immature neutrophils (particularly band cells) in peripheral blood. Band cells have a curved, unsegmented nucleus shaped like a "C" or horseshoe.
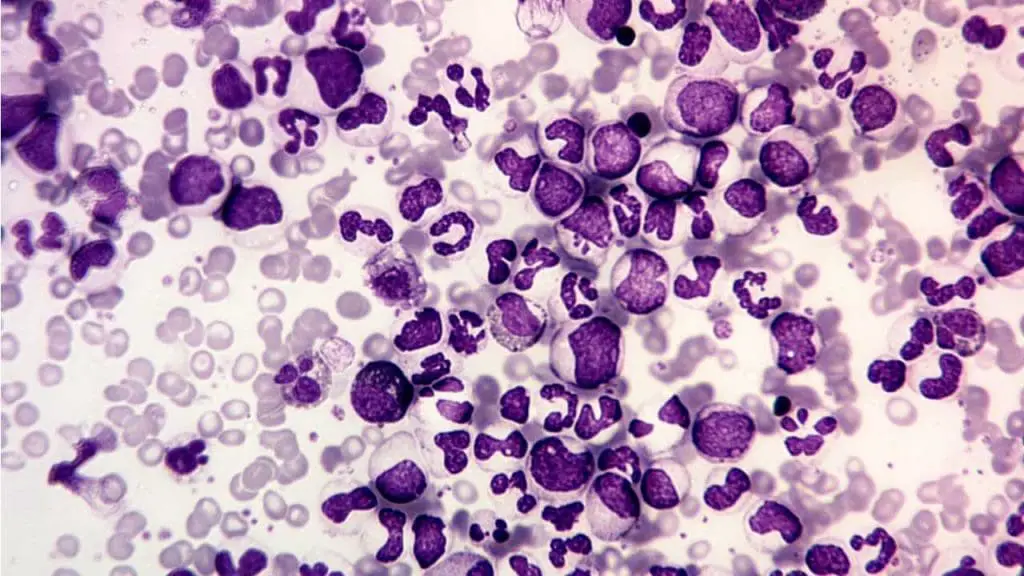
Common causes.
- Acute bacterial infection. The bone marrow releases immature cells to meet demand.
- Severe stress, trauma, or burns.
- Chronic myeloid leukemia and other bone marrow disorders, which release a much wider spectrum of immature granulocytes (metamyelocytes, myelocytes, even blasts).
Why it matters. A left shift confirms the inflammatory leukogram in sepsis but, when more immature forms (myelocytes, promyelocytes) appear, the question shifts toward bone marrow disease. CML is the classic example, especially when combined with basophilia, eosinophilia, and splenomegaly. BCR-ABL1 testing confirms the diagnosis. can be a sign of a blood cancer or other bone marrow-related diseases.
Pelger-Huët Anomaly (PHA)
Pelger-Huët anomaly is an inherited disorder of neutrophil nuclear segmentation caused by mutations in the lamin B receptor (LBR) gene. Inheritance is autosomal dominant. The cells are abnormal in shape but normal in function.
What it looks like. Neutrophils with bilobed, "pince-nez" (resembling a pair of spectacles), peanut-shaped, or dumbbell-shaped nuclei. Chromatin appears dense and coarsely clumped.
Why it matters. PHA itself is benign and needs no treatment. Its main clinical relevance is that it can be mistaken for a marked left shift on a smear, prompting unnecessary infection workups [5]. Recognizing the family pattern and the consistent involvement of almost all neutrophils prevents this error.
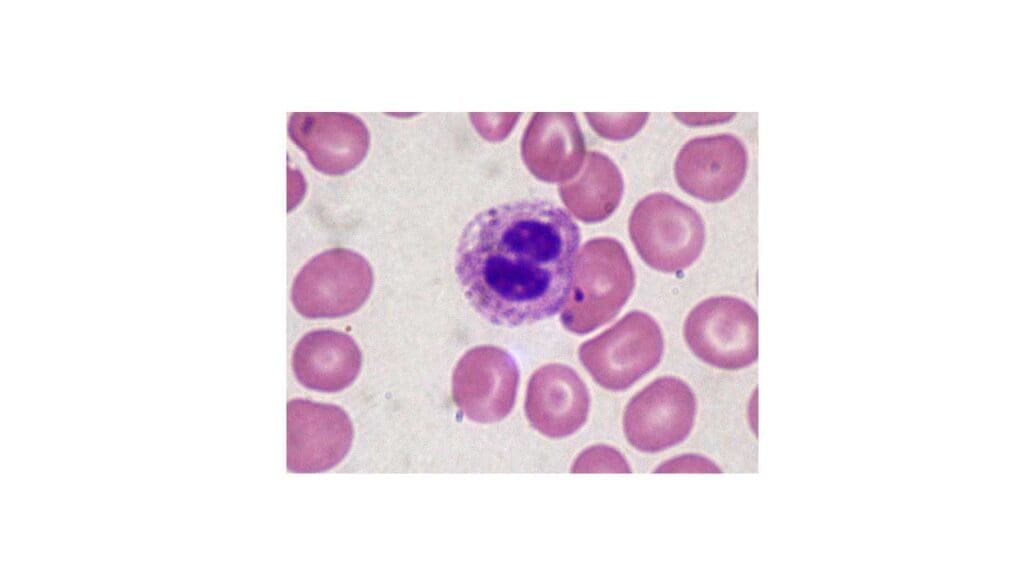
Pseudo-Pelger-Huët Anomaly (PPHA)
This is the acquired look-alike of PHA, and it is arguably more important for students to know. PPHA is one of the most reliable smear markers of granulocytic dysplasia.
Common causes.
- Myelodysplastic neoplasms and acute myeloid leukemia.
- Chronic myeloid leukemia, particularly after treatment.
- Drugs, including tacrolimus, mycophenolate, sulfa drugs, valproate, ibuprofen, taxanes (paclitaxel, docetaxel), and even some BTK inhibitors. Drug-induced PPHA is typically transient and reverses within weeks of stopping the drug [3].
- Less commonly, severe infection, multiple myeloma, malaria, and B12 or folate deficiency.
How to tell PHA and PPHA apart. In inherited PHA, almost every neutrophil looks abnormal and the patient is asymptomatic. In PPHA, usually fewer than 20% of neutrophils are affected, other dysplastic features are often present, and there is a clinical context — a known malignancy, a transplant, or a new medication [3].
When to investigate further. Bone marrow aspirate and biopsy with cytogenetics and a myeloid mutation panel, particularly if dysplastic features cluster or cytopenias are present. Always review the medication list first to spot a reversible drug cause.
Differences of PHA from Other Abnormal WBCs with Similar Morphology
| Feature | Pelger-Huët Anomaly (PHA) | Pseudo-Pelger-Huët | Left Shift (Bandemia) |
|---|---|---|---|
| Cause | Inherited LBR mutation | MDS, AML, drugs | Acute infection or stress |
| Proportion of neutrophils affected | Nearly all | Usually < 20% | Variable, with band cells dominant |
| Clinical state | Asymptomatic | Often dysplasia or drug exposure | Acute illness, infection signs |
| Action | Reassure; document for future smears | Bone marrow workup or drug review | Treat the infection |
Drumsticks in Neutrophils
Drumsticks are small, round chromatin appendages connected to a neutrophil nucleus by a thin stalk. They represent the inactive X chromosome (the Barr body) and appear in about 1–2% of neutrophils in healthy females [10].
Why it matters today. Drumsticks were historically used as a cytological marker of female sex. With modern karyotyping, FISH, and chromosomal microarray, this role is largely obsolete. The reason to recognize them now is to avoid misclassifying them as pathology. True drumsticks must be distinguished from non-specific nuclear projections like sessile nodules and racquet forms, which are smaller, less intensely staining, and clinically insignificant [10].
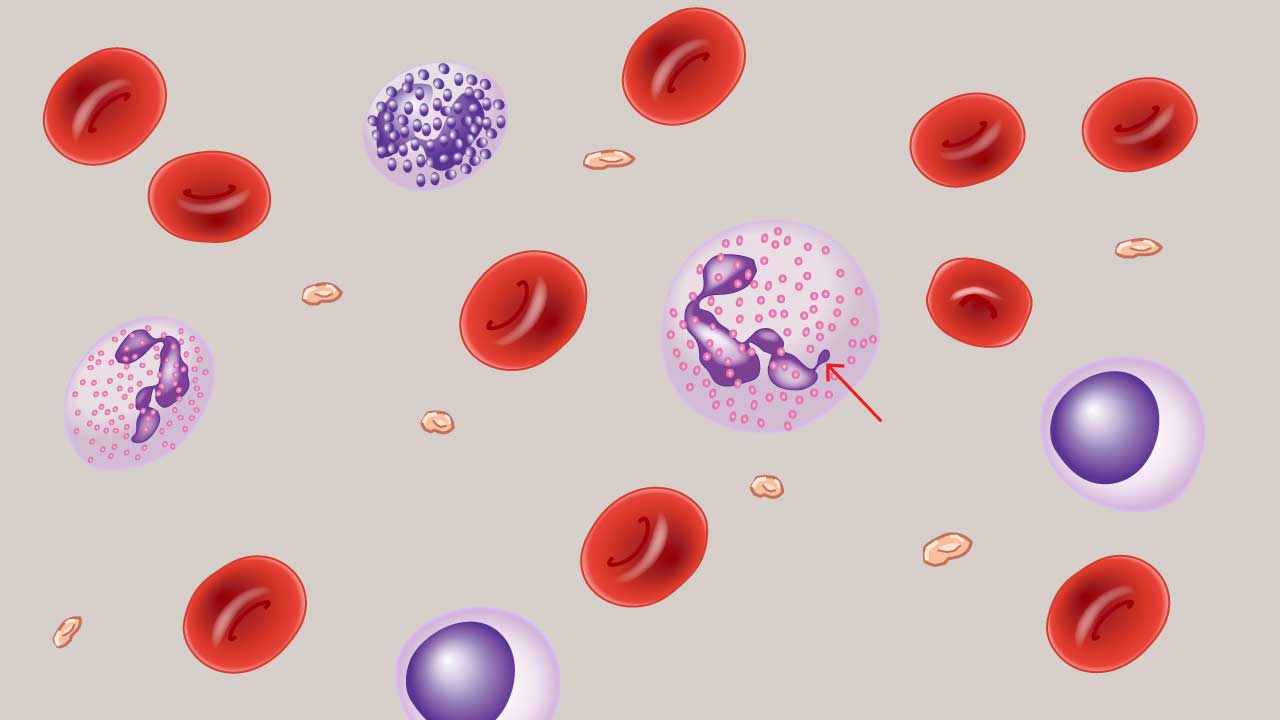
Other Important Findings
Reactive Lymphocytes
Reactive lymphocytes — sometimes called atypical lymphocytes — are lymphocytes that have enlarged and changed in response to antigenic or immune stimulation.
What they look like. Larger than typical lymphocytes, with abundant, deeply basophilic (blue) cytoplasm that often appears to "scallop" around adjacent red cells. The nucleus may be irregular, indented, or cleaved.
CausesCommon causes.
- Viral infections. Infectious mononucleosis (Epstein-Barr virus) is the classic example. CMV, hepatitis viruses, influenza, and HIV also drive reactive changes.
- Some bacterial infections — pertussis, brucellosis, cat-scratch disease.
- Autoimmune disease and drug hypersensitivity.
Why it matters. Most reactive lymphocytes signal a benign, self-limited immune response. Persistent atypical cells, very high counts, or unusual nuclear features should prompt flow cytometry to exclude leukemia or lymphoma. The presence of smudge cells (fragile lymphocytes that rupture on the slide) in an older adult with lymphocytosis points toward chronic lymphocytic leukemia (CLL).
Dysplasia
Dysplasia is a broad term for the disordered maturation of blood cells in the bone marrow. The cells that emerge look abnormal and often work poorly.
What it looks like on a smear.
- Granulocytic dysplasia — hypogranular neutrophils, pseudo-Pelger-Huët cells, and immature myeloid forms.
- Erythroid dysplasia — circulating nucleated red cells, anisopoikilocytosis, basophilic stippling.
- Megakaryocytic dysplasia — giant or hypogranular platelets.
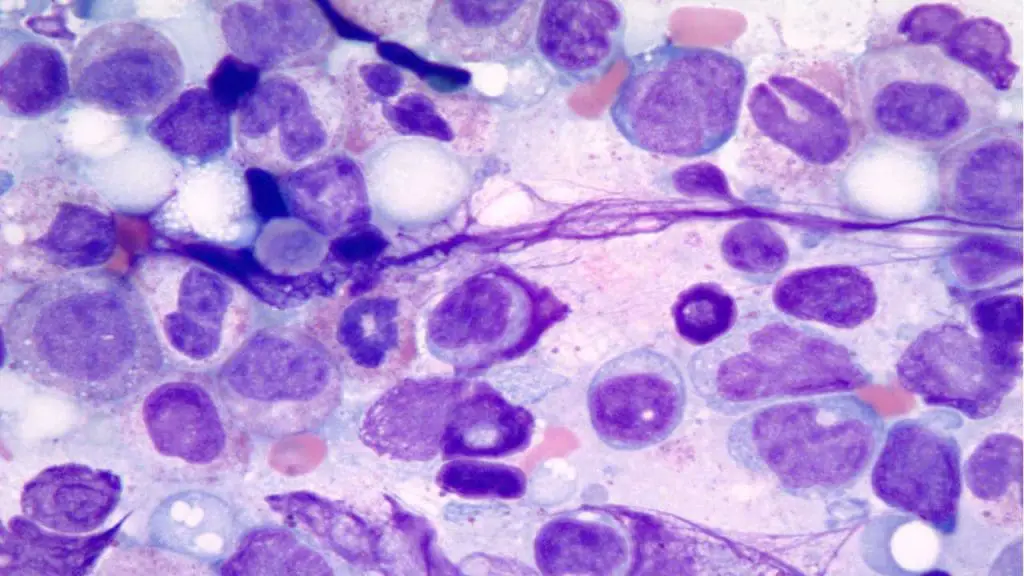
CausCauses.
- Primary myelodysplastic neoplasms (MDS). In 2022, the WHO 5th edition renamed myelodysplastic syndromes as myelodysplastic neoplasms and split them into genetically defined and morphology-defined groups. Examples of genetically defined subtypes include MDS with biallelic TP53 inactivation, MDS with low blasts and SF3B1 mutation, and MDS with del(5q). The International Consensus Classification (ICC) of 2022 runs in parallel with similar but not identical categories [1,2].
- Therapy-related dysplasia after chemotherapy or radiation for prior cancer.
- Toxic exposure, classically to benzene.
- Bone marrow failure syndromes.
Why it matters. Dysplastic features on a smear usually trigger a bone marrow aspirate and biopsy with cytogenetics, FISH, and a myeloid mutation panel. The combined morphologic, cytogenetic, and molecular data sit at the heart of the 2022 WHO and ICC classifications and shape both prognosis and treatment.
When to investigate further. Review the medication list to exclude reversible PPHA. Consider B12 and folate before committing to a malignancy workup. Refer to hematology for bone marrow evaluation.
Summary of Abnormal WBC Morphology
| Abnormality | Key Feature | Common Causes | Why It Matters |
| Toxic granulation | Coarse, dark blue granules in neutrophils | Severe bacterial infection, sepsis, G-CSF | Active inflammatory response |
| Döhle bodies | Pale blue oval inclusions near cell membrane | Infection, burns, pregnancy, May-Hegglin anomaly | Marker of "toxic change" |
| Vacuoles | Clear round spaces in cytoplasm | Sepsis, drug toxicity, storage disorders | Cell stress or accumulation of unprocessed material |
| Alder-Reilly bodies | Permanent coarse granules in all leukocytes | Mucopolysaccharidoses, Tay-Sachs | Clue to inherited lysosomal storage disease |
| Auer rods / faggot cells | Pink needle-like crystals in blasts | AML, especially APL | Pathognomonic for myeloid leukemia |
| Hypogranularity | Reduced or absent granules | MDS, AML, vitamin deficiency | Marker of dysplasia |
| Hypersegmented neutrophils | Six or more nuclear lobes | B12 or folate deficiency, hydroxyurea | Classic megaloblastic anemia clue |
| Left shift (bandemia) | Increased immature band cells | Acute infection, CML | Inflammatory response or marrow disorder |
| Pelger-Huët anomaly | Bilobed "pince-nez" or peanut nuclei | Inherited LBR mutation (PHA); MDS, AML, drugs (PPHA) | PHA is benign; PPHA points to dysplasia |
| Drumsticks | Small chromatin appendage from nucleus | Inactive X chromosome in females | Historical sex chromatin marker |
| Reactive lymphocytes | Large, blue-tinged cytoplasm; irregular nuclei | Viral infection, especially EBV/CMV | Immune activation |
| Smudge cells | Fragile, ruptured lymphocytes | CLL, sample handling | Diagnostic clue in CLL |
| Dysplasia | Disordered maturation features | MDS, AML | Triggers bone marrow biopsy |
Common Pitfalls to Remember
- PHA vs. PPHA vs. left shift. Inherited PHA affects nearly all neutrophils in a healthy patient. PPHA affects a minority of neutrophils, often with other dysplastic features or a drug exposure. A left shift adds band cells and metamyelocytes alongside infection.
- Toxic granulation vs. Alder-Reilly bodies. Toxic granulation is transient and limited to neutrophils. Alder-Reilly bodies are permanent and appear in all leukocyte lineages.
- Döhle bodies vs. May-Hegglin inclusions. Both are blue-gray cytoplasmic structures. May-Hegglin inclusions are larger, more elongated, and come with giant platelets and mild thrombocytopenia.
Frequently Asked Questions (FAQs)
What does "abnormal WBC morphology" mean on a blood smear report?
It means the white blood cells viewed under the microscope show shape, size, or staining changes that differ from healthy cells. These changes act as clues. They can point to infection, vitamin deficiency, bone marrow disorders, inherited conditions, or drug effects, and they usually prompt further blood tests or a bone marrow examination.
How are toxic granulation and Alder-Reilly bodies different?
Toxic granulation appears in neutrophils during severe bacterial infection or inflammation and is temporary. Alder-Reilly bodies look similar but are permanent inherited findings linked to mucopolysaccharidoses or Tay-Sachs disease. Alder-Reilly bodies also appear in monocytes and lymphocytes, while toxic granulation is limited to neutrophils.
Why is hypersegmentation of neutrophils important?
Neutrophils with six or more nuclear lobes are a classic clue for vitamin B12 or folate deficiency. Recognizing the change on a smear prompts testing for these deficiencies and treatment, which can reverse the anemia and prevent neurological complications from prolonged B12 deficiency.
Is Pelger-Huët anomaly dangerous?
The inherited form is benign. Affected neutrophils have only two lobes but work normally. The danger lies in misreading it as a left shift and ordering unnecessary tests for infection. An acquired version, called pseudo-Pelger-Huët anomaly, is different and may signal a bone marrow disorder or a drug reaction that needs follow-up.
What is the difference between inherited and acquired Pelger-Huët anomaly?
Inherited Pelger-Huët anomaly is a lifelong genetic trait that affects nearly all neutrophils and causes no symptoms. Pseudo-Pelger-Huët anomaly appears later in life. It usually affects a smaller proportion of neutrophils, and it is linked to myelodysplastic neoplasms, leukemia, or certain drugs such as tacrolimus, mycophenolate, ibuprofen, or chemotherapy agents.
When are reactive lymphocytes a sign of cancer rather than infection?
Most reactive lymphocytes reflect viral infections like infectious mononucleosis and resolve as the infection clears. They become concerning when their numbers stay elevated for weeks, when there are very abnormal-looking forms with cleaved nuclei, when smudge cells appear, or when they occur with weight loss, fever, or swollen lymph nodes. Flow cytometry and clinician review separate benign reactive changes from leukemia or lymphoma.
Glossary of Related Medical Terms
- Apoptosis — Programmed cell death; a controlled, orderly process by which damaged or unneeded cells self-destruct.
- Auer rods — Pink-red, needle-shaped crystalline inclusions made of fused azurophilic granules; found in myeloid blasts.
- Azurophilic — Stains purple-blue with azure dyes used in Romanowsky stains (e.g., Wright-Giemsa).
- Bandemia / left shift — A rise in immature neutrophils (band cells) in peripheral blood.
- Basophilic — Stains dark blue with basic dyes due to acidic cellular components like RNA.
- Cytopenia — A low count of one or more blood cell types (anemia, neutropenia, thrombocytopenia).
- Dysplasia — Abnormal cell development, often signaling disordered bone marrow maturation.
- Faggot cells — Promyelocytes containing bundles of multiple Auer rods; characteristic of acute promyelocytic leukemia (APL).
- Granulocyte — A WBC with visible cytoplasmic granules: neutrophils, eosinophils, and basophils.
- Hyposegmentation — Fewer nuclear lobes than normal in a granulocyte.
- Hypersegmentation — Six or more nuclear lobes in a neutrophil; classic clue for B12 or folate deficiency.
- Leukogram — The white blood cell portion of a complete blood count.
- Lysosomal storage disorder — Inherited disease in which a missing or faulty enzyme allows substances to build up inside cells.
- Megaloblastic anemia — Anemia caused by impaired DNA synthesis (typically B12 or folate deficiency), producing large red cells.
- Mucopolysaccharidosis (MPS) — A group of inherited disorders caused by enzyme defects that prevent breakdown of glycosaminoglycans.
- Myelodysplastic neoplasms (MDS) — Clonal bone marrow disorders with ineffective blood cell production and a risk of progression to AML.
- Peripheral blood smear — A thin layer of blood spread on a glass slide and stained for microscopic examination.
- Phagocytosis — The process by which cells engulf and digest particles like bacteria.
- Pseudo-Pelger-Huët anomaly (PPHA) — Acquired hyposegmentation of neutrophils, usually a marker of dysplasia or drug effect.
Reactive lymphocyte — A lymphocyte enlarged and altered in response to antigenic or immune stimulation.
Disclaimer: This article is intended for informational purposes only and is specifically targeted towards medical students. It is not intended to be a substitute for informed professional medical advice, diagnosis, or treatment. While the information presented here is derived from credible medical sources and is believed to be accurate and up-to-date, it is not guaranteed to be complete or error-free. See additional information.
References
- Khoury, J.D., Solary, E., Abla, O. et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia 36, 1703–1719 (2022). https://doi.org/10.1038/s41375-022-01613-1
- Arber, D. A., Orazi, A., Hasserjian, R. P., Borowitz, M. J., Calvo, K. R., Kvasnicka, H. M., Wang, S. A., Bagg, A., Barbui, T., Branford, S., Bueso-Ramos, C. E., Cortes, J. E., Dal Cin, P., DiNardo, C. D., Dombret, H., Duncavage, E. J., Ebert, B. L., Estey, E. H., Facchetti, F., Foucar, K., … Tefferi, A. (2022). International Consensus Classification of Myeloid Neoplasms and Acute Leukemias: integrating morphologic, clinical, and genomic data. Blood, 140(11), 1200–1228. https://doi.org/10.1182/blood.2022015850
- Wang, E., Boswell, E., Siddiqi, I., Lu, C. M., Sebastian, S., Rehder, C., & Huang, Q. (2011). Pseudo-Pelger-Huët anomaly induced by medications: a clinicopathologic study in comparison with myelodysplastic syndrome-related pseudo-Pelger-Huët anomaly. American journal of clinical pathology, 135(2), 291–303. https://doi.org/10.1309/AJCPVFY95MAOBKRS
- Farrelly, S. J., & O'Connor, K. A. (2017). Hypersegmented neutrophils and oval macrocytes in the setting of B12 deficiency and pancytopaenia. BMJ case reports, 2017, bcr2016218508. https://doi.org/10.1136/bcr-2016-218508
- Shah, S. S., Parikh, R. S., Vaswani, L. P., & Divkar, R. (2016). Familial Pelger-Huet Anomaly. Indian journal of hematology & blood transfusion : an official journal of Indian Society of Hematology and Blood Transfusion, 32(Suppl 1), 347–350. https://doi.org/10.1007/s12288-015-0508-3
- Akyay, A., Demir Şahin, F., & Şen, A. (2020). Vacuolated Leukocytes in the Peripheral Blood Smear of a Child with Chanarin-Dorfman Syndrome. Turkish journal of haematology : official journal of Turkish Society of Haematology, 37(4), 299–300. https://doi.org/10.4274/tjh.galenos.2020.2020.0242
- Zhao, Y., Diao, Y., Zheng, J., Li, X., & Luan, H. (2024). Performance evaluation of the digital morphology analyser Sysmex DI-60 for white blood cell differentials in abnormal samples. Scientific reports, 14(1), 14344. https://doi.org/10.1038/s41598-024-65427-0
- Xing, Y., Liu, X., Dai, J., Ge, X., Wang, Q., Hu, Z., Wu, Z., Zeng, X., Xu, D., & Qu, C. (2023). Artificial intelligence of digital morphology analyzers improves the efficiency of manual leukocyte differentiation of peripheral blood. BMC medical informatics and decision making, 23(1), 50. https://doi.org/10.1186/s12911-023-02153-z
- AlMozain, Nour; Bakshi, Nasir A. (2017). A Case of Hunter Syndrome and Alder-Reilly Anomaly. Journal of Applied Hematology 8(1):p 33-35. htpps://doi.org/10.4103/joah.joah_29_16
- Méhes K. (1966). Nuclear projections in neutrophils. Blood, 28(4), 598–601. https://doi.org/10.1182/blood.V28.4.598.598