Key Takeaways
Primary myelofibrosis is a rare chronic blood cancer originating in the bone marrow. Characterized by abnormal blood cell production, bone marrow fibrosis (scarring), and enlarged spleen.
- Pathogenesis ▾: Caused by clonal mutations in a hematopoietic stem cell, leading to overproduction of abnormal blood cells and bone marrow fibrosis due to inflammatory mediators.
- Epidemiology ▾:
- Prevalence: Around 1-2 cases per 100,000 individuals.
- Incidence: Approximately 0.5-1 case per 100,000 individuals annually.
- Age and gender: More common in older adults and slightly more prevalent in men.
- Geographical variations: Slightly higher rates in Europe and North America compared to other regions.
- Risk Factors and Causes ▾:
- Age: Risk increases significantly with age.
- Environmental factors: Exposure to certain chemicals and ionizing radiation.
- Family history: Having a close relative with MPNs.
- Genetic mutations: JAK2, CALR, and MPL mutations are the main culprits.
- Signs and Symptoms ▾:
- Early: Fatigue, weakness, night sweats, unexplained weight loss.
- Later: Enlarged spleen, bone pain, easy bruising/bleeding, symptoms from low blood cell counts (anemia, infections).
- Complications ▾: Infections, transformation to AML, bleeding/thrombosis, portal hypertension, extramedullary hematopoiesis, bone pain, fatigue, and splenomegaly-related symptoms.
- Laboratory investigations ▾: Complete blood count, peripheral blood smear, bone marrow aspiration and biopsy, molecular testing.
- Diagnostic criteria: Based on a combination of major and minor criteria, including bone marrow fibrosis, splenomegaly, and other factors.
- Treatment and Management ▾:
- Supportive care (transfusions, hydroxyurea, pain management, aspirin)
- JAK inhibitors (ruxolitinib, fedratinib)
- Interferon-alpha
- Splenectomy
- Low-dose radiation
- Stem cell transplantation (rarely)
- Prognosis: Varies depending on age, mutations, and other factors. DIPSS Plus score helps assess risk stratification.
*Click ▾ for more information
Introduction
Primary myelofibrosis (PMF) is a rare chronic blood cancer that originates in the bone marrow. It is characterized by:
- Abnormal blood cell production: The bone marrow produces too many abnormal blood cells, leading to imbalances in the different cell types (red blood cells, white blood cells, and platelets).
- Bone marrow fibrosis: Scarring (fibrosis) develops within the bone marrow, making it difficult for healthy blood cells to be produced.
- Enlarged spleen (splenomegaly): The spleen tries to compensate for the abnormal blood cell production, leading to its enlargement.
Classification
Primary myelofibrosis (PMF) is classified as a myeloproliferative neoplasm (MPN). MPNs are a group of blood cancers characterized by the overproduction of one or more types of blood cells. Other MPNs include:
Primary myelofibrosis (PMF) is considered a primary MPN because it arises on its own, without developing from another blood disorder.
Pathogenesis and Pathophysiology

The exact cause of primary myelofibrosis (PMF) is not fully understood, but it arises from the development of acquired (not inherited) genetic mutations in the DNA of hematopoietic stem cells within the bone marrow. These mutated stem cells and their progeny lead to an overproduction of abnormal blood cells, particularly megakaryocytes (precursors of platelets). These abnormal megakaryocytes release signaling molecules that stimulate fibroblasts, specialized cells in the bone marrow, to produce excessive collagen and other fibers, resulting in the characteristic scarring or fibrosis.
Several gene mutations have been frequently identified in individuals with primary myelofibrosis (PMF), with the most common being:
- JAK2 (Janus Kinase 2) mutation: Found in approximately 50-60% of primary myelofibrosis (PMF) patients. The most frequent JAK2 mutation is V617F, which leads to the JAK2 protein being constitutively active, promoting excessive blood cell production and contributing to fibrosis.
- CALR (Calreticulin) mutation: Present in about 25-35% of primary myelofibrosis (PMF) patients who do not have the JAK2 mutation. These mutations, often insertions or deletions in the CALR gene, result in an altered calreticulin protein that can also activate signaling pathways leading to MPN development.
- MPL (Myeloproliferative Leukemia virus oncogene) mutation: Found in a smaller proportion of primary myelofibrosis (PMF) patients (around 5-10%) who are negative for both JAK2 and CALR mutations. MPL encodes the thrombopoietin receptor, and mutations in this gene can also lead to its overactivation, driving the disease.
Around 10-15% of primary myelofibrosis (PMF) patients do not have any of these three common mutations and are referred to as "triple-negative." Other less common gene mutations can also be present and are being actively researched for their role in primary myelofibrosis (PMF) pathogenesis and prognosis.
While these genetic mutations are key drivers of primary myelofibrosis (PMF), the specific triggers that cause these mutations to occur in the first place remain largely unknown. In rare instances, exposure to certain industrial chemicals like benzene or high doses of radiation has been suggested as potential risk factors. It's also important to note that in a small subset of individuals, PMF can evolve from other myeloproliferative neoplasms like essential thrombocythemia (ET) or polycythemia vera (PV).
Epidemiology of Primary Myelofibrosis
- Prevalence: Primary myelofibrosis (PMF) is a relatively rare blood cancer. The estimated global prevalence is around 1-2 cases per 100,000 individuals.
- Age and Gender Distribution: Primary myelofibrosis (PMF) typically occurs in older adults, with the average age of diagnosis being around 65-70 years. The disease is more common in men compared to women, with a male-to-female ratio of approximately 1.5:1.
- Geographical Variations: The incidence of primary myelofibrosis (PMF) shows some variations across different geographical regions with slightly higher rates are observed in Europe and North America compared to other regions.
Risk Factors and Causes of PMF
Risk Factors
Several factors are considered risk factors for developing the disease:
- Age: The risk of primary myelofibrosis (PMF) increases significantly with age, with the highest incidence seen in individuals over 60 years old.
- Exposure to certain chemicals: Long-term exposure to benzene (found in some industrial settings and gasoline) and certain other solvents has been linked to an increased risk of primary myelofibrosis (PMF).
- Ionizing radiation: Exposure to high levels of ionizing radiation, such as that from atomic bombs or certain medical procedures, can also increase the risk.
- Family history: Having a close relative (parent, sibling, child) with MPNs, including primary myelofibrosis (PMF), increases the risk, though it is still relatively low.
Causes
The specific cause of primary myelofibrosis (PMF) is still unknown. However, it is believed to involve a combination of genetic and environmental factors:
- Genetic mutations: As discussed earlier, mutations in genes like JAK2, CALR, and MPL are found in the majority of primary myelofibrosis (PMF) cases. These mutations lead to dysregulation of cell growth and survival pathways, ultimately contributing to the development of the disease.
- Environmental factors: While not fully understood, exposure to certain environmental toxins and chemicals may play a role in triggering these genetic mutations or promoting the development of primary myelofibrosis (PMF) alongside the mutations.
Signs and Symptoms of PMF
The primary myelofibrosis (PMF) symptoms can be varied and non-specific in the early stages, often making it difficult to diagnose. However, as the disease progresses, the symptoms become more pronounced.
Early Primary Myelofibrosis Symptoms
- Fatigue: This is a common and often the first noticeable myelofibrosis symptom. It can be described as a feeling of tiredness, exhaustion, and lack of energy that persists even after adequate rest.
- Weakness: This can manifest as a feeling of decreased muscle strength and difficulty performing daily activities.
- Night sweats: Excessive sweating during sleep, especially at night, can be a sign of an underlying inflammatory process, which can be present in primary myelofibrosis (PMF).
- Unexplained weight loss: This can occur due to various reasons in primary myelofibrosis (PMF), including decreased appetite, increased metabolic rate due to inflammation, and difficulty absorbing nutrients.

This image illustrates several key signs and symptoms associated with primary myelofibrosis (PMF). Anemia, characterized by a decrease in red blood cells, can lead to fatigue, weakness, and shortness of breath. Massive hepatosplenomegaly refers to a significant enlargement of both the liver (hepatomegaly) and spleen (splenomegaly), which can cause discomfort, early satiety (feeling full after eating a small amount), and other complications. Additionally, hypermetabolism (increased metabolic rate) can manifest as unintended weight loss, night sweats, and fatigue, potentially contributing to a reduced quality of life in individuals with PMF.
Later Myelofibrosis Symptoms
- Enlarged spleen (splenomegaly): This is a hallmark primary myelofibrosis (PMF) symptom and can be felt as a mass or fullness in the upper left abdominal region. The enlarged spleen can cause discomfort, early satiety (feeling full after eating a small amount), and other myelofibrosis symptoms.
- Bone pain: This can be a general ache or a sharp pain in the bones, often occurring in the back, legs, or ribs.
- Easy bruising or bleeding: This is due to a decrease in the number of platelets, which are essential for blood clotting.
- Myelofibrosis symptoms related to low blood cell counts:
- Anemia: This deficiency in red blood cells can lead to symptoms like pale skin, shortness of breath, and dizziness.
- Frequent infections: A decrease in white blood cells can weaken the immune system, making individuals more susceptible to infection
Complications of PMF
Primary myelofibrosis (PMF) can lead to various complications if left untreated or if the treatment is not effective.
Infection
Due to a weakened immune system caused by decreased white blood cell counts, individuals with primary myelofibrosis (PMF) are more susceptible to infections, including bacterial, viral, and fungal infections. These infections can be serious and require prompt medical attention.
Transformation to Acute Myeloid Leukemia (AML)
In some cases, primary myelofibrosis (PMF) can transform into AML, a more aggressive form of leukemia. This occurs due to the accumulation of additional genetic mutations in the bone marrow cells over time. The risk of transformation increases with the duration and severity of primary myelofibrosis (PMF).
Bleeding and Thrombosis
Primary myelofibrosis (PMF) can lead to both bleeding and thrombosis (blood clot formation) due to imbalances in blood cell production.
- Bleeding: A decrease in platelets can increase the risk of easy bruising, bleeding from the gums or nose, and internal bleeding.
- Thrombosis: Although less common than bleeding, blood clots can form in various locations, such as the deep veins of the legs (deep vein thrombosis) or the lungs (pulmonary embolism). These clots can be life-threatening and require immediate medical attention.
Portal Hypertension
An enlarged spleen (splenomegaly) can block the flow of blood through the portal vein, a major vein in the abdomen. This can lead to portal hypertension, a condition characterized by high blood pressure in the portal vein system. Myelofibrosis symptoms may include abdominal discomfort, ascites (fluid accumulation in the abdomen), and variceal bleeding (bleeding from enlarged veins in the esophagus or stomach).
Extramedullary hematopoiesis
In some cases, the bone marrow becomes so severely affected by fibrosis that it cannot produce enough blood cells. As a compensatory mechanism, blood cell production can start occurring outside the bone marrow, in organs like the liver and spleen. This is called extramedullary hematopoiesis. While it helps to maintain blood cell counts to some extent, it can also lead to organ dysfunction if not properly controlled.
Other complications
- Bone pain: The bone marrow fibrosis can cause chronic pain in the bones.
- Fatigue: This remains a significant issue even with treatment, impacting quality of life.
- Splenomegaly-related symptoms: The enlarged spleen can cause discomfort, early satiety, and other bothersome symptoms.
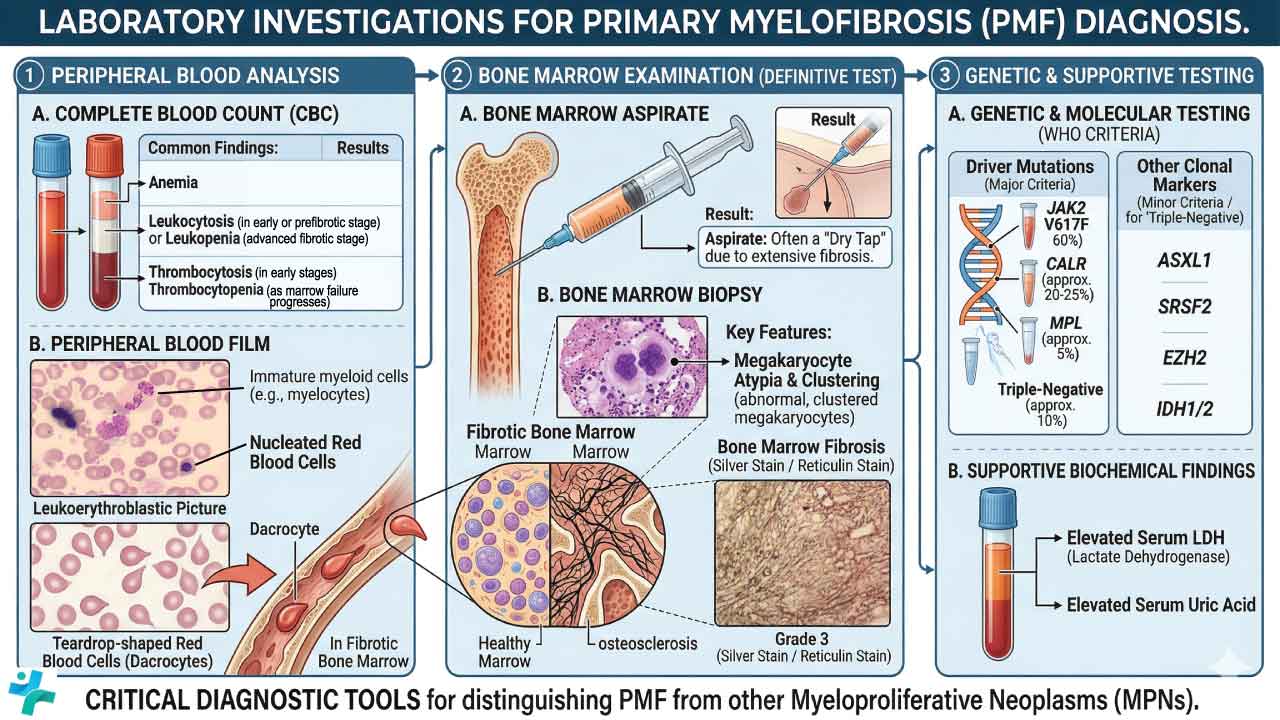
Laboratory Investigations for Primary Myelofibrosis
A comprehensive diagnostic workup for primary myelofibrosis (PMF) requires correlating peripheral blood findings with bone marrow histology and molecular genetics.
Complete Blood Count (CBC)
The CBC findings in primary myelofibrosis (PMF) are highly variable and depend on the disease stage (prefibrotic vs. overt fibrotic phase):
- Anemia: Usually normocytic and normochromic. It is the most common cytopenia and progressively worsens as bone marrow fibrosis increases.
- White Blood Cells (WBCs): Leukocytosis (elevated WBC) is common in early or prefibrotic stages, while leukopenia (low WBC) may develop in advanced fibrotic stages.
- Platelets: Thrombocytosis (elevated platelets) frequently occurs early in the disease course. As marrow failure progresses, severe thrombocytopenia ensues.
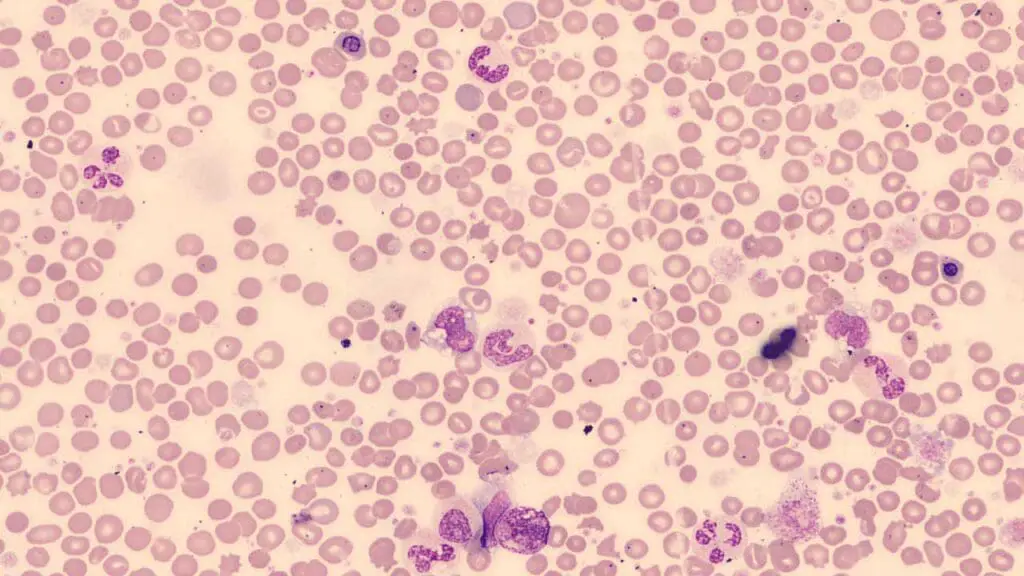
Peripheral Blood Smear (PBS)
The peripheral blood smear is a critical, low-cost screening tool that reveals the classic triad of primary myelofibrosis (PMF) morphology:
- Leukoerythroblastosis: The premature release of nucleated red blood cells (nRBCs) and immature myeloid cells (e.g., myelocytes, metamyelocytes, promyelocytes) into the peripheral circulation. This indicates the disruption of the bone marrow-blood barrier due to fibrosis.
- Dacrocytes (Tear-drop cells): Red blood cells that are stretched and deformed as they squeeze through the fibrotic, scarred bone marrow matrix or the enlarged spleen.
- Atypical Platelets: The presence of giant, hypogranular platelets and circulating megakaryoblast fragments.
Bone Marrow Aspiration and Biopsy
Bone marrow evaluation is the gold standard for diagnosing PMF and establishing the baseline degree of fibrosis
- Aspiration: Often results in a "dry tap" (inability to draw liquid marrow) due to the dense reticulin and collagen fibrosis packing the marrow space.
- Biopsy (Morphology): Demonstrates a hypercellular marrow in early stages, progressing to hypocellularity. The hallmark is megakaryocytic proliferation and atypia where the megakaryocytes are clustered, have dense/hyperlobulated nuclei, and vary significantly in size.
- Biopsy (Fibrosis Grading): Reticulin staining is mandatory to grade fibrosis from MF-0 to MF-3:
- MF-0: Scattered linear reticulin with no intersections.
- MF-1: Loose network of reticulin with many intersections (Prefibrotic PMF).
- MF-2: Diffuse and dense increase in reticulin with extensive intersections, occasionally with focal collagen or osteosclerosis (Overt PMF).
- MF-3: Dense network of thick reticulin with coarse bundles of collagen, often associated with significant osteosclerosis (advanced Overt PMF).
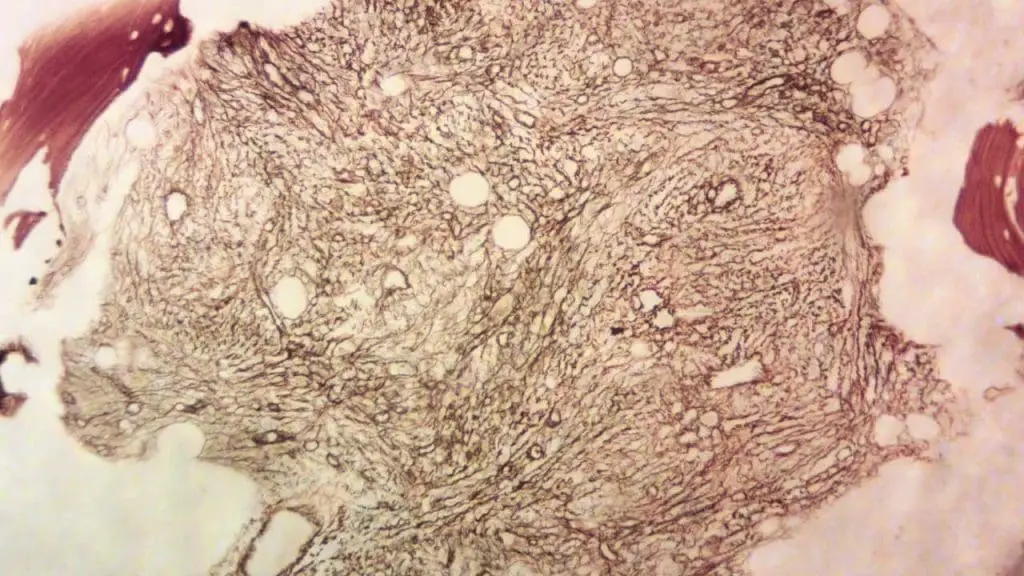
WHO/European Consensus Grading System
| Grade | Description of Fibers | Clinical Interpretation |
| MF-0 | Scattered linear reticulin with no intersections (cross-overs) corresponding to normal bone marrow. | Normal / No fibrosis |
| MF-1 | Loose network of reticulin with many intersections, particularly in perivascular areas. | Prefibrotic PMF (pre-PMF) |
| MF-2 | Diffuse and dense increase in reticulin with extensive intersections; occasionally with focal bundles of collagen and/or osteosclerosis. | Overt PMF |
| MF-3 | Diffuse and dense increase in reticulin with extensive intersections and coarse bundles of collagen; often associated with significant osteosclerosis. | Advanced Overt PMF |
- Prefibrotic PMF (pre-PMF): This requires a grading of MF-0 or MF-1. Pathologically, the focus is on megakaryocytic atypia and increased granulopoiesis rather than the fiber density itself.
- Overt PMF: This requires a grading of MF-2 or MF-3. At this stage, the mechanical obstruction of the marrow by collagen fibers leads to the classic clinical features like a "dry tap" and tear-drop cells on the peripheral smear.
Diagnostic Criteria for Primary Myelofibrosis
According to the updated 2022 World Health Organization (WHO) and International Consensus Classification (ICC) guidelines, PMF is divided into two distinct clinical phases: Prefibrotic PMF (pre-PMF) and Overt PMF. Diagnosis requires meeting all 3 Major Criteria, AND at least 1 Minor Criterion.
Criteria for Overt PMF
Major Criteria (Must meet all 3)
- Bone Marrow Morphology: Megakaryocytic proliferation and atypia, accompanied by reticulin and/or collagen fibrosis grade 2 or 3 (MF-2 or MF-3).
- Exclusion of other Neoplasms: Does not meet WHO/ICC criteria for Essential Thrombocythemia (ET), Polycythemia Vera (PV), BCR-ABL1 positive Chronic Myeloid Leukemia (CML), Myelodysplastic Syndromes (MDS), or other myeloid neoplasms.
- Molecular Demonstration: Presence of JAK2, CALR, or MPL mutation. (Note: In "triple-negative" cases, the presence of another clonal marker like ASXL1 or EZH2, or the absence of reactive bone marrow fibrosis, fulfills this criterion).
Minor Criteria (Must meet at least 1, confirmed on two consecutive occurrences)
- Anemia: Not attributed to a comorbid condition (e.g., iron deficiency or bleeding).
- Leukocytosis: WBC count ≥ 11 x 109/L.
- Palpable Splenomegaly: Enlarged spleen detected on physical examination.
- Elevated LDH: Above the upper limit of normal for the institution's reference range.
- Leukoerythroblastosis: Present on the peripheral blood smear.
Criteria for Prefibrotic PMF (pre-PMF)
The diagnostic criteria for pre-PMF are nearly identical to Overt PMF, with the key difference being the bone marrow grading.
- Major Criterion 1 Difference: The bone marrow shows megakaryocytic proliferation and atypia, but without reticulin fibrosis greater than grade 1 (must be MF-0 or MF-1). It often presents with age-adjusted hypercellularity and granulocytic proliferation.
- Minor Criteria Difference: Leukoerythroblastosis is not required as a minor criterion for pre-PMF, but the other minor criteria (anemia, leukocytosis, splenomegaly, elevated LDH) apply.
Treatment and Management of PMF
Risk Stratification
Before initiating any treatment, patients must be risk-stratified using prognostic models like DIPSS Plus or the newer, molecularly integrated MIPSS70. Treatment goals diverge significantly based on these scores:
- Low-Risk Patients: Goal is maximizing quality of life and observing for progression.
- High-Risk Patients: Goal is extending survival and evaluating for curative stem cell transplantation.
Asymptomatic and Low-Risk PMF
For patients classified as Low or Intermediate-1 risk who do not have debilitating symptoms, severe anemia, or massive splenomegaly, the standard of care is "Watch and Wait" (active observation). Premature initiation of therapies like JAK inhibitors in asymptomatic patients exposes them to unnecessary drug toxicities without proven survival benefits.
Targeted Therapies
For symptomatic patients (especially those with constitutional symptoms and symptomatic splenomegaly), JAK inhibitors are the cornerstone of medical therapy. They do not cure the disease or completely reverse bone marrow fibrosis, but they drastically reduce spleen size and systemic inflammation.
- Ruxolitinib (Jakafi):
- Mechanism: JAK1 and JAK2 inhibitor.
- Indication: The first-line standard for intermediate or high-risk PMF with symptomatic splenomegaly.
- Limitation: Can cause or worsen anemia and thrombocytopenia.
- Fedratinib (Inrebic):
- Mechanism: Highly selective JAK2 inhibitor.
- Indication: Used primarily in patients who are intolerant to, or have relapsed after, Ruxolitinib.
- Limitation: Carries a black box warning for Wernicke’s encephalopathy; thiamine levels must be monitored.
- Pacritinib (Vonjo):
- Mechanism: JAK2 and IRAK1 inhibitor.
- Indication: Specifically approved for PMF patients with severe thrombocytopenia (platelet count < 50 x 109/L). This fills a massive clinical gap, as Ruxolitinib is difficult to dose in these patients.
- Momelotinib (Ojjaara):
- Mechanism: Inhibits JAK1, JAK2, and uniquely, ACVR1.
- Indication: The go-to choice for primary myelofibrosis (PMF) patients with significant anemia. By inhibiting ACVR1, it decreases hepcidin production, which restores iron homeostasis and improves red blood cell production.
Management of Cytopenias
When primary myelofibrosis (PMF) presents primarily with bone marrow failure rather than proliferative features, treatment shifts to managing the specific cytopenias.
Managing Anemia
- Erythropoiesis-Stimulating Agents (ESAs): Epoetin alfa or darbepoetin can be used if endogenous erythropoietin levels are low (< 500 mU/mL).
- Danazol: A synthetic androgen that can improve anemia in a subset of patients by stimulating red blood cell production.
- Luspatercept: An erythroid maturation agent increasingly utilized for transfusion-dependent anemia in myeloproliferative neoplasms.
- Blood Transfusions: The mainstay for severe, symptomatic anemia, though it carries the long-term risk of iron overload (requiring iron chelation therapy).
Managing Thrombocythemia & Leukocytosis
- Hydroxyurea: A cytoreductive agent used to control high white blood cell or platelet counts, and it can also moderately reduce spleen size.
Management of Refractory Splenomegaly
If targeted therapies and cytoreduction fail to control a massive, painful spleen, mechanical interventions are considered.
- Splenectomy: Surgical removal is high-risk in primary myelofibrosis (PMF) due to the potential for massive bleeding, thrombosis, or extreme compensatory liver enlargement (hepatomegaly) post-surgery. It is generally a last resort.
- Splenic Irradiation: Low-dose radiation can provide temporary relief (usually 3–6 months) for patients who are not surgical candidates, though it carries a high risk of prolonged, severe cytopenias.
Allogeneic Stem Cell Transplantation (Allo-HSCT)
Allo-HSCT is the only potentially curative therapy for primary myelofibrosis (PMF). It replaces the fibrotic, mutated marrow with healthy donor hematopoietic stem cells.
- Patient Selection: Because allo-HSCT carries a high rate of transplant-related mortality (graft-versus-host disease, severe infections), it is strictly reserved for:
- Patients with Intermediate-2 or High-risk disease.
- Younger, fit patients (typically under 70-75 years old) without severe comorbidities.
- Bridge to Transplant: JAK inhibitors are often used to shrink the spleen and improve the patient's physical condition before proceeding to transplant.
Emerging Therapies (Clinical Trials)
To keep the website authoritative and forward-looking, note that the future of primary myelofibrosis (PMF) treatment lies in combination therapies (e.g., adding a second agent to Ruxolitinib). Notable classes under investigation include:
- BET Inhibitors (e.g., Pelabresib): Show promise in reducing bone marrow fibrosis and spleen volume.
- BCL-xL Inhibitors (e.g., Navitoclax): Target the apoptotic pathways to induce cell death in the mutated clone.
Prognosis
Key Prognostic Scoring Systems
Modern primary myelofibrosis (PMF) prognosis is divided by whether the patient is a candidate for Hematopoietic Cell Transplantation (HCT).
MIPSS70+ Version 2.0 (The Integrated Standard)
This is currently the most utilized model for transplant-age patients (usually <70 years). It weights molecular data heavily alongside clinical severity.
| Risk Category | Total Points | Median Overall Survival (OS) | 10-Year OS Rate |
| Very Low | 0 | Not Reached | ~92% |
| Low | 1–2 | ~16.4 Years | ~56% |
| Intermediate | 3–4 | ~7.7 Years | ~37% |
| High | 5–8 | ~4.1 Years | ~13% |
| Very High | ≥9 | ~1.8 Years | <5% |
GIPSS (Genetically Inspired Prognostic Scoring System)
GIPSS is unique because it relies exclusively on mutations and karyotype, making it useful when clinical data (like symptoms or exact blood counts) are fluctuant.
- Low Risk: Median OS of ~26 years.
- High Risk: Median OS of ~2.6 years.
DIPSS-Plus (Clinical/Dynamic)
Still widely used for bedside assessment. It identifies 8 risk factors: Age >65, Hb <10g/dL, WBC >25 x 109/L, Blasts ≥1%, Constitutional symptoms, Unfavorable karyotype, Platelets <100 x 109/L, and RBC transfusion dependency.
High Molecular Risk (HMR) Mutations
The presence and number of non-driver mutations significantly shorten survival. If a patient has ≥ 2 HMR mutations, they are automatically shifted into a higher risk tier regardless of their blood counts.
- ASXL1: The most common HMR mutation; strongly associated with shorter survival and fibrotic progression.
- SRSF2 & U2AF1 (Q157): Spliceosome mutations that significantly increase the risk of Leukemic Transformation.
- EZH2: Linked to poor outcomes and aggressive myeloproliferation.
- IDH1/2: Associated with rapid progression to Blast Phase.
CALR Type 1/Like mutations are considered "prognostically favorable" compared to JAK2 or MPL mutations, often predicting longer survival and lower thrombosis risk.
Risk of Leukemic Transformation (Blast Phase)
Approximately 10–20% of primary myelofibrosis (PMF) patients will progress to secondary Acute Myeloid Leukemia (sAML), also known as PMF in Blast Phase (PMF-BP).
- Transformation Timing: Often occurs within the first 5 years if HMR mutations (SRSF2, IDH1/2) are present.
- Post-Transformation Prognosis: Exceedingly poor, with a median survival of only ~5–6 months unless the patient can successfully bridge to an urgent stem cell transplant.
Summary of Prognosis
| Factor | Favorable Prognosis | Poor Prognosis |
| Genetics | CALR Type 1 Mutation | Triple Negative, ASXL1, SRSF2, EZH2 |
| Karyotype | Normal or 13q-, 20q- | inv(3), -7, i(17q), 12p- |
| Clinical | Asymptomatic | Transfusion-dependent, Blasts ≥2% |
| Spleen | Small/Non-palpable | Massive (>10cm), Progressive enlargement |
Post-Essential Thrombocythemia (ET) MF and Post-Polycythemia Vera (PV) MF
- Post-ET MF: This occurs when a patient diagnosed with essential thrombocythemia (ET), characterized by an increased platelet count, eventually progresses to develop myelofibrosis. This transition can happen years after the initial diagnosis of ET, with an average time of 7-20 years. The overall risk of developing post-ET MF is around 1.6-9%.
- Post-PV MF: This occurs when a patient diagnosed with polycythemia vera (PV), characterized by an increased red blood cell count, progresses to develop myelofibrosis. Similar to post-ET MF, this transformation can take years to occur, with an average of 7-20 years after the initial PV diagnosis. The risk of developing post-PV MF is slightly higher compared to post-ET MF, with an estimated range of 5-14%.
Frequently Asked Questions (FAQs)
Is primary myelofibrosis curable?
No, primary myelofibrosis (PMF) is generally not considered curable with currently available treatment options. Allogeneic stem cell transplantation (SCT) offers the only potential for cure in PMF. This procedure replaces the patient's bone marrow with healthy stem cells from a donor but it's a high-risk procedure typically reserved for younger patients with high-risk disease and a suitable donor.
What are the 4 hallmarks of myelofibrosis?
The four hallmarks of myelofibrosis are:
- Enlarged spleen (splenomegaly): This is a characteristic feature of primary myelofibrosis (PMF) and can be felt as a mass or fullness in the upper left abdominal region. The enlarged spleen can cause various symptoms, including:
- Early satiety (feeling full after eating a small amount)
- Discomfort in the upper left abdomen
- Shortness of breath in severe cases
- Anemia: This occurs due to a decrease in red blood cell production in the bone marrow. Symptoms of anemia can include:
- Fatigue
- Weakness
- Pale skin
- Shortness of breath
- Dizziness
- Bone marrow fibrosis: This is a hallmark feature of primary myelofibrosis (PMF) and refers to the scarring and hardening of the bone marrow. This scarring disrupts the normal architecture of the bone marrow, making it difficult for healthy blood cells to be produced.
- Debilitating disease-associated myelofibrosis symptoms: These myelofibrosis symptoms can significantly impact a patient's quality of life and include:
- Fatigue: This is a common and often the first noticeable myelofibrosis symptom. It can be described as a feeling of tiredness, exhaustion, and lack of energy that persists even after adequate rest.
- Night sweats: These are excessive sweating episodes that occur during sleep and can be related to the body's attempt to regulate temperature due to inflammation.
- Unexplained weight loss: This can occur due to various reasons in primary myelofibrosis (PMF), including decreased appetite, increased metabolic rate due to inflammation, and difficulty absorbing nutrients.
- Bone pain: This can be a general ache or a sharp pain in the bones, often occurring in the back, legs, or ribs.
- Easy bruising or bleeding: This is due to a decrease in the number of platelets, which are essential for blood clotting.
What are the signs myelofibrosis is getting worse?
While the course of myelofibrosis (MF) can vary considerably from person to person, there are signs and symptoms that might indicate the disease is worsening:
Progression of Existing Myelofibrosis Symptoms
- Increased fatigue and weakness: This is a common myelofibrosis symptom, but if it worsens significantly and interferes significantly with daily activities, it could suggest disease progression.
- Worsening pain: Bone pain, often in the back, legs, or ribs, can worsen as the disease progresses.
- More pronounced weight loss: Unexplained weight loss can occur in myelofibrosis and may become more noticeable as the disease worsens.
- Splenomegaly progression: An enlarged spleen (splenomegaly) is a hallmark of myelofibrosis. If the spleen continues to grow significantly, it can cause discomfort, early satiety (feeling full after eating a small amount), and other myelofibrosis symptoms.
Development of New Myelofibrosis Symptoms
- Fevers and night sweats: These can be signs of increased inflammation or potential infections, which are more common with advanced myelofibrosis.
- Increased bleeding or bruising: This can occur due to a decrease in platelets, which are essential for blood clotting.
- Shortness of breath: This can be caused by various factors in myelofibrosis, including anemia (low red blood cell count) or enlarged spleen compressing the lungs.
- Frequent infections: A weakened immune system due to myelofibrosis can increase susceptibility to infections.
Changes in Blood Tests
- Decreasing blood counts: Lower red blood cell (anemia), white blood cell, and platelet counts may be observed as the disease progresses.
- Increased blast count: A rise in the number of immature blood cells (blasts) in the bone marrow can indicate a more aggressive form of myelofibrosis or potential transformation to acute myeloid leukemia (AML).
How painful is myelofibrosis?
The pain experienced with myelofibrosis (MF) can vary considerably from person to person, ranging from no pain to significant and debilitating pain. Several factors contribute to this variability.
Causes of Pain in Myelofibrosis
- Bone marrow fibrosis: The hallmark feature of myelofibrosis involves scarring and hardening of the bone marrow. This process can irritate and inflame surrounding tissues, leading to pain, especially in the back, ribs, and legs.
- Splenomegaly: An enlarged spleen, a common feature of myelofibrosis, can press on nearby organs like the stomach and diaphragm, causing discomfort and pain in the upper left abdomen.
- Increased inflammation: Chronic inflammation associated with myelofibrosis can contribute to generalized aches and pains throughout the body.
- Individual pain tolerance: People have varying levels of pain tolerance, which influences how they perceive and experience pain.
Severity of Pain
- Mild pain: Some individuals with myelofibrosis experience mild, occasional aches that might not significantly interfere with daily activities.
- Moderate pain: This is more common and can be managed with medications like pain relievers and anti-inflammatory drugs.
- Severe pain: In some cases, the pain can be severe and debilitating, requiring additional pain management strategies like nerve blocks or stronger pain medications.
Management of Pain in Myelofibrosis
- Identifying the cause: Accurately determining the source of the pain is crucial for effective management.
- Medications: Pain relievers like nonsteroidal anti-inflammatory drugs (NSAIDs) and stronger pain medications like opioids may be prescribed depending on the severity of pain.
- Other strategies: Other approaches like physical therapy, massage therapy, and relaxation techniques can help manage pain and improve overall well-being.
- Addressing the underlying cause: When possible, addressing the underlying cause of the pain, such as managing inflammation and reducing spleen size, can significantly improve pain levels.
What causes death with myelofibrosis?
Myelofibrosis (MF) can lead to death due to a variety of causes.
- Transformation to Acute Myeloid Leukemia (AML): One of the most significant risks for patients with MF is the progression of the disease to acute myeloid leukemia (AML). AML is an aggressive form of blood cancer that can be rapidly fatal if not treated promptly. The risk of AML transformation varies, but it's a significant contributor to death in myelofibrosis patients.
- Complications of Bone Marrow Failure: As MF progresses, the bone marrow becomes increasingly scarred and fibrotic. This can severely impair its ability to produce healthy blood cells, leading to complications such as:
- Severe anemia: This can cause life-threatening fatigue, weakness, and shortness of breath.
- Infections: A weakened immune system due to bone marrow failure increases vulnerability to severe infections that can be difficult to treat.
- Bleeding: Low platelet counts can cause spontaneous bleeding, which can be life-threatening in some cases.
- Complications of Splenomegaly: An enlarged spleen (splenomegaly), a common feature of myelofibrosis, can lead to various complications:
- Splenic rupture: In rare cases, a severely enlarged spleen can rupture, causing massive internal bleeding and potentially leading to death.
- Portal hypertension: Increased pressure in the blood vessels around the liver due to splenomegaly can lead to portal hypertension, resulting in bleeding and other complications.
- Other Causes
- Heart failure: Complications like heart failure can occur due to chronic anemia, iron overload from transfusions, or other underlying medical problems.
- Liver failure: Advanced myelofibrosis, portal hypertension, and certain complications can lead to liver failure.
- Thrombosis (blood clots): Patients with myelofibrosis might have an increased risk of blood clots, which can cause serious complications like stroke or heart attack.
Comorbidities
Myelofibrosis mainly affects older individuals who often have other existing medical conditions (comorbidities). These conditions, along with complications of myelofibrosis, can contribute to death.
What organs does myelofibrosis affect?
While myelofibrosis (MF) primarily affects the bone marrow, it can indirectly impact various organs due to its influence on blood cell production and the presence of specific myelofibrosis symptoms.
1. Spleen
Splenomegaly (enlarged spleen): This is a hallmark feature of myelofibrosis. The spleen filters blood cells, removing old or damaged ones. An enlarged spleen can occur due to increased workload in trying to compensate for abnormal blood cell production and removal of abnormal cells. This can cause:
- Discomfort or pain in the upper left abdomen
- Early satiety (feeling full after eating a small amount)
- Pressure on nearby organs like the stomach and diaphragm
2. Liver
Portal hypertension: When the enlarged spleen disrupts blood flow in the portal vein (drains blood from the spleen and intestines to the liver), it can lead to increased pressure in the portal vein system. This, in turn, can cause:
- Variceal bleeding: Increased pressure can cause enlarged veins in the esophagus or stomach to rupture, leading to potentially life-threatening bleeding.
- Ascites: Accumulation of fluid in the abdomen.
3. Lungs
Extramedullary hematopoiesis (EMH): In rare cases, blood cell production can occur outside the bone marrow, including in the lungs. This can cause:
- Coughing
- Shortness of breath
- Chest pain
4. Skin
Pruritus (itching): This can be a common symptom in myelofibrosis, although the exact cause is not fully understood.
5. Other organs
- Bones: Bone pain can be a common symptom in myelofibrosis due to the abnormal bone marrow environment and inflammation.
- Heart: Chronic anemia and other factors associated with myelofibrosis can potentially increase the risk of heart failure.
What is the difference between primary and secondary myelofibrosis?
Primary myelofibrosis develops de novo, meaning it occurs on its own. Secondary myelofibrosis evolves from a pre-existing myeloproliferative neoplasm, most commonly essential thrombocythemia (post-ET MF) or polycythemia vera (post-PV MF).
What does a "triple-negative" myelofibrosis diagnosis mean?
A triple-negative diagnosis in primary myelofibrosis indicates the absence of the three classic driver mutations: JAK2, CALR, and MPL. This subtype often requires comprehensive genetic profiling to identify other somatic mutations and is associated with a distinct clinical prognosis.
How do JAK inhibitors work in treating primary myelofibrosis?
JAK inhibitors target the overactive JAK-STAT signaling pathway. While they do not reverse the bone marrow fibrosis or cure the disease, they significantly reduce spleen volume and alleviate debilitating constitutional symptoms caused by excess inflammatory cytokines in primary myelofibrosis.
Can primary myelofibrosis transform into acute leukemia?
Yes, in approximately 10-20% of cases, primary myelofibrosis can undergo leukemic transformation into acute myeloid leukemia (AML), also known as blast-phase MPN. The risk is heavily influenced by the patient's specific genetic mutations and the duration of the disease.
Glossary of Related Medical Terms
- Constitutional Symptoms: Systemic symptoms caused by underlying inflammation, including fatigue, drenching night sweats, low-grade fever, and unintended weight loss.
- Dacrocyte (Tear-drop cell): An abnormally shaped red blood cell commonly observed on the peripheral blood smear in conditions involving bone marrow fibrosis.
- Extramedullary Hematopoiesis (EMH): The production of blood cells outside the bone marrow, most frequently occurring in the spleen and liver.
- Fibroblast: Connective tissue cells in the bone marrow that, when overstimulated by cytokines from abnormal cells, produce excessive collagen leading to scarring.
- Hepatomegaly: Abnormal enlargement of the liver.
- Leukoerythroblastosis: The presence of immature white blood cells and nucleated red blood cells in the peripheral blood circulation, a classic finding in myelophthisic processes like PMF.
- Megakaryocyte: A large bone marrow cell responsible for producing platelets; atypical, clustered megakaryocytes are a primary driver of the fibrotic process in PMF.
- Osteosclerosis: An abnormal increase in bone density and hardening of the bone trabeculae, frequently seen alongside reticulin fibrosis on bone marrow biopsies.
- Splenomegaly: Abnormal enlargement of the spleen, typically serving as a compensatory mechanism for compromised bone marrow function.
Disclaimer: This article is intended for informational purposes only and is specifically targeted towards medical students. It is not intended to be a substitute for informed professional medical advice, diagnosis, or treatment. While the information presented here is derived from credible medical sources and is believed to be accurate and up-to-date, it is not guaranteed to be complete or error-free. See additional information.
References
- https://mpn-hub.com/medical-information/international-consensus-classification-2022-for-myeloproliferative-neoplasms
- Gianelli U, Thiele J, Orazi A, Gangat N, Vannucchi AM, Tefferi A, Kvasnicka HM. International Consensus Classification of myeloid and lymphoid neoplasms: myeloproliferative neoplasms. Virchows Arch. 2023 Jan;482(1):53-68. doi: 10.1007/s00428-022-03480-8. Epub 2022 Dec 29. PMID: 36580136; PMCID: PMC9852206.
- Goldberg S, Hoffman J. Clinical Hematology Made Ridiculously Simple, 1st Edition: An Incredibly Easy Way to Learn for Medical, Nursing, PA Students, and General Practitioners (MedMaster Medical Books). 2021.
- Keohane EM, Otto CN, Walenga JM. Rodak's Hematology 6th Edition (Saunders). 2019.
- Takenaka K, Shimoda K, Akashi K. Recent advances in the diagnosis and management of primary myelofibrosis. Korean J Intern Med. 2018 Jul;33(4):679-690. doi: 10.3904/kjim.2018.033. Epub 2018 Apr 20. PMID: 29665657; PMCID: PMC6030412.
- Oon, S.F., Singh, D., Tan, T.H. et al. Primary myelofibrosis: spectrum of imaging features and disease-related complications. Insights Imaging 10, 71 (2019). https://doi.org/10.1186/s13244-019-0758-y
- Tefferi A. Primary myelofibrosis: 2023 update on diagnosis, risk-stratification, and management . American Journal of Hematology; 2023;98(5):801-821.
- Verstovsek, S., Mesa, R.A., Livingston, R.A. et al. Ten years of treatment with ruxolitinib for myelofibrosis: a review of safety. J Hematol Oncol 16, 82 (2023). https://doi.org/10.1186/s13045-023-01471-z
- Rampal, R. K., Grosicki, S., Chraniuk, D., Abruzzese, E., Bose, P., Gerds, A. T., Vannucchi, A. M., Palandri, F., Lee, S. E., Gupta, V., Lucchesi, A., Oh, S. T., Kuykendall, A. T., Patriarca, A., Álvarez-Larrán, A., Mesa, R., Kiladjian, J. J., Talpaz, M., Scandura, J. M., Lavie, D., … Mascarenhas, J. (2025). Pelabresib plus ruxolitinib for JAK inhibitor-naive myelofibrosis: a randomized phase 3 trial. Nature medicine, 31(5), 1531–1538. https://doi.org/10.1038/s41591-025-03572-3
- Mascarenhas, J., Hoffman, R., Talpaz, M., Gerds, A. T., Stein, B., Gupta, V., Szoke, A., Drummond, M., Pristupa, A., Granston, T., Daly, R., Al-Fayoumi, S., Callahan, J. A., Singer, J. W., Gotlib, J., Jamieson, C., Harrison, C., Mesa, R., & Verstovsek, S. (2018). Pacritinib vs Best Available Therapy, Including Ruxolitinib, in Patients With Myelofibrosis: A Randomized Clinical Trial. JAMA oncology, 4(5), 652–659. https://doi.org/10.1001/jamaoncol.2017.5818