Key Takeaways
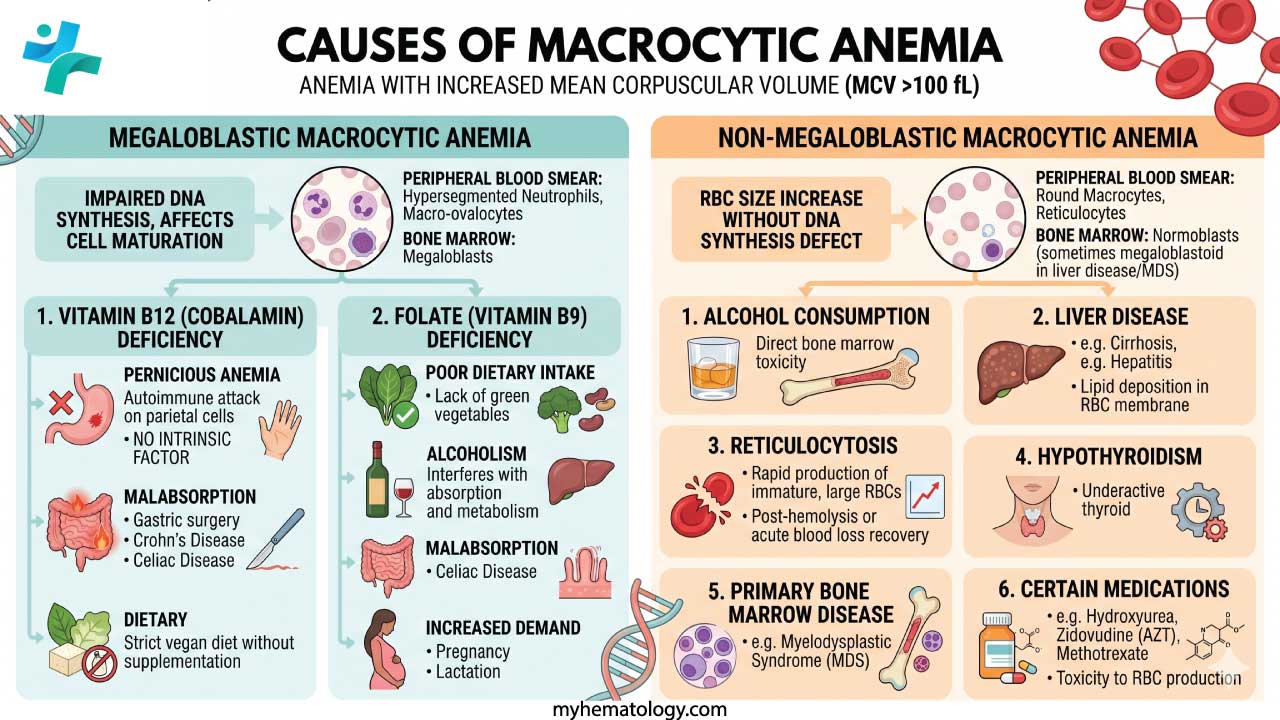
Macrocytic anemia is defined by the presence of abnormally large red blood cells, indicated by a Mean Corpuscular Volume (MCV) strictly greater than 100 fL.
- Categories: Megaloblastic (caused by impaired DNA synthesis) or non-megaloblastic (caused by other mechanisms, such as membrane lipid alterations or altered marrow release)
- Megaloblastic Anemias ▾: Vitamin B12 (cobalamin) and Folate (vitamin B9) deficiencies are the most common causes of megaloblastic anemia.
- Non-Megaloblastic Anemias ▾: Common causes include chronic alcohol consumption, liver disease, severe hypothyroidism, myelodysplastic syndromes (MDS), and marked reticulocytosis (where immature, larger red blood cells falsely elevate the MCV).
- Medication Triggers: Extreme macrocytosis (MCV > 130 fL) should immediately raise suspicion for drug-induced causes, particularly chemotherapeutics (e.g., methotrexate, hydroxyurea) and antiretrovirals (e.g., zidovudine).
*Click ▾ for more information
Introduction
Macrocytic anemia is a type of anemia in which red blood cells are abnormally large. To understand it, it helps to break the name down: macrocytic means “large cell,” and anemia means a shortage of healthy red blood cells. Together, they describe a situation where the bone marrow is producing oversized red blood cells that often can’t do their job properly — including carrying oxygen around the body.
The condition is detected through a routine blood test called a complete blood count (CBC). One of the values this test measures is the mean corpuscular volume (MCV) — essentially the average size of your red blood cells. A normal MCV falls between 80 and 100 femtoliters (fL). In macrocytic anemia, the MCV is above 100 fL, signalling that the cells are too large.
Because macrocytic anemia has many possible causes, working out which one is responsible — the differential diagnosis — is the real clinical challenge. Getting this right matters enormously. Some causes are simple to fix; others point to serious underlying conditions that need urgent attention.
There are two broad categories of macrocytic anemia, and the distinction between them shapes everything that follows:
- Megaloblastic anemia — caused by impaired DNA synthesis, most commonly due to vitamin B12 or folate deficiency. This is the most common category.
- Non-megaloblastic anemia — caused by other mechanisms, such as liver disease, alcohol use, or bone marrow disorders, where DNA synthesis is not the primary problem.
Why MCV Matters: Sizing up the Red Blood Cell
The MCV is your first navigational tool when investigating anemia. It doesn’t give you the diagnosis — but it tells you which direction to look. Here’s how it divides the landscape:
- Microcytic anemia (MCV < 80 fL) — smaller than normal red blood cells. Think iron deficiency or thalassemia.
- Normocytic anemia (MCV 80–100 fL) — normal-sized cells, but not enough of them. Seen in aplastic anemia or hemolytic anemia.
- Macrocytic anemia (MCV > 100 fL) — larger than normal cells. This is the focus of this article.
An MCV above 100 fL is not a diagnosis in itself — it’s a prompt to investigate further. The size of the MCV elevation can also offer useful clues. Mild macrocytosis (100–110 fL) is more likely to reflect liver disease or alcohol use. A very high MCV (above 130 fL) should immediately raise suspicion for drug-induced causes or myelodysplastic syndrome (MDS).
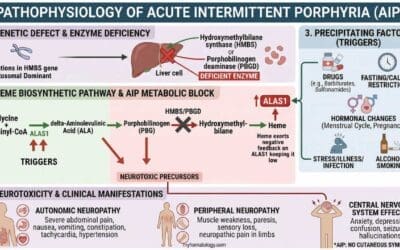
Megaloblastic Anemia
Megaloblastic anemia is the most common form of macrocytic anemia. It gets its name from megaloblasts — abnormally large, immature red blood cell precursors that appear in the bone marrow. These cells look busy, but they’re dysfunctional. They struggle to divide normally and often die before they can enter the bloodstream.
The root cause is almost always a deficiency in vitamin B12 or folate (vitamin B9). Both nutrients are essential for DNA synthesis — the process by which cells copy their genetic material before dividing. Without enough of either vitamin, cell division slows down. The cell keeps growing, but the nucleus (which contains the DNA) falls behind. This creates a distinctive pattern called nuclear-cytoplasmic asynchrony: a large cell with an immature nucleus.
Vitamin B12 Deficiency

Vitamin B12 — also called cobalamin — is a workhorse nutrient. It supports DNA synthesis and helps maintain the myelin sheath, the protective coating around nerve fibres. A deficiency therefore causes two distinct sets of problems: blood abnormalities and neurological damage.
How it happens
When B12 levels fall, DNA synthesis is disrupted, leading to the megaloblastic changes described above. At the same time, a compound called methylmalonic acid (MMA) accumulates in the blood. MMA is thought to damage myelin, which is why B12 deficiency — unlike folate deficiency — can cause lasting nerve damage (Bernetti et al., 2025).
B12 acts as a cofactor for two key reactions in the body:
- The conversion of homocysteine to methionine
- The conversion of methylmalonyl-CoA to succinyl-CoA
When these reactions stall, both MMA and homocysteine build up — a pattern that can be detected on blood tests.
Common causes
- Pernicious anemia — an autoimmune condition where the immune system destroys the stomach cells that produce intrinsic factor (IF). Intrinsic factor is a protein that the body must have to absorb B12 in the small intestine. Without it, even a good diet cannot prevent deficiency.
- Strict vegan or plant-based diet — B12 is found almost exclusively in animal products. People who eat no meat, fish, dairy, or eggs are at risk if they don’t supplement.
- Malabsorption — conditions that damage the stomach or small intestine, such as Crohn’s disease, coeliac disease, gastrectomy (stomach removal), or chronic pancreatitis, can prevent B12 from being absorbed properly.
- Long-term medication use — proton pump inhibitors (PPIs, used for acid reflux) and metformin (used for type 2 diabetes) can both interfere with B12 absorption over time.
Neurological risk: a critical warning
B12 deficiency can cause subacute combined degeneration (SCD) of the spinal cord — damage to the nerve tracts that control movement and sensation. Always screen for early symptoms: pins and needles (paraesthesia), loss of vibration sense or proprioception (awareness of body position), and unsteady walking (ataxia).
This is clinically urgent. Neurological damage from B12 deficiency can become permanent if not treated promptly. Diagnosing and treating the blood abnormality is not enough — the neurological picture must be assessed independently.
Treatment
B12 supplementation is the cornerstone of treatment. Intramuscular (IM) injections have traditionally been the gold standard because they bypass the gut entirely — particularly important when absorption is the problem. However, modern evidence shows that high-dose oral B12 (1000–2000 mcg daily) is equally effective in many patients, thanks to a secondary absorption pathway in the gut that doesn’t require intrinsic factor (Kuzminski et al., 1998; Butler et al., 2006; Mazur et al., 2025).
The Beefy Red Tongue
A classic sign of B12 deficiency is atrophic glossitis — the tongue loses its normal bumpy texture (papillae), becoming smooth, shiny, and reddened. Clinicians sometimes call it a “beefy red tongue.” If a patient presents with macrocytosis and a sore, unusually smooth tongue, B12 or folate deficiency should be your first suspicion.

Folate Deficiency
Folate — vitamin B9 — is the other primary cause of megaloblastic anemia. It shares many features with B12 deficiency on the blood film, but there is one important practical difference: the body’s folate stores run out much faster. While B12 stores can last two to four years, folate stores are typically exhausted within weeks to months of inadequate intake.
How it happens
Folate is essential for making the building blocks of DNA — specifically, it drives the conversion of deoxyuridylate (dUMP) to deoxythymidylate (dTMP), a step that is critical for DNA replication. Without enough folate, cell division slows. The cytoplasm keeps growing while the nucleus stalls — producing the same nuclear-cytoplasmic asynchrony seen in B12 deficiency (Ames et al., 2002; Wickramasinghe & Fida, 1994; Chon et al., 2019).

Common causes
- Poor diet — folate is found in leafy green vegetables, fruits, and fortified grains. A diet lacking in fresh produce — particularly in elderly patients living alone — is a common cause. Importantly, overcooking vegetables destroys folate content.
- Increased demand — pregnancy and breastfeeding significantly increase the body’s folate requirements. Chronic haemolytic anemias (such as sickle cell disease) also raise demand, because the bone marrow is working overtime to replace destroyed red cells.
- Alcohol use — alcohol impairs folate absorption in the small intestine, disrupts its metabolism in the liver, and increases how much the kidneys excrete. This makes alcohol one of the leading causes of folate deficiency.
- Malabsorption — conditions that damage the upper small intestine — such as coeliac disease or tropical sprue — prevent folate from being absorbed effectively.
A critical diagnostic rule: always check B12 first
It is medically dangerous to treat a macrocytic anemia with folate supplementation before checking vitamin B12 levels. Here is why: if the patient actually has a B12 deficiency, giving them folic acid will correct the blood picture — but will not protect the nervous system. Worse, it may divert the body’s remaining B12 away from myelin maintenance and towards red blood cell production, potentially accelerating neurological damage (Green & Miller, 1999).
Always rule out B12 deficiency before starting folate treatment.
Treatment
Oral folic acid supplementation is highly effective — usually 1–5 mg daily. Because folate is water-soluble, the body simply excretes any excess, making toxicity extremely rare.
Clinical Clue: The “Tea and Toast” Diet
In clinical practice, folate deficiency often appears in elderly patients or those living in social isolation who rely on a limited diet — sometimes called a ‘tea and toast’ diet. Fresh vegetables and fruits are expensive and perishable; when access to a varied diet is limited, folate stores deplete quickly. If you encounter a patient with macrocytosis and poor nutritional history, folate deficiency should be near the top of your list.
Non-Megaloblastic Anemia
Not all macrocytic anemia is caused by vitamin deficiency. In non-megaloblastic causes, DNA synthesis is generally intact. Instead, the red cells become large because of structural changes to the cell membrane, altered bone marrow dynamics, or the direct effects of toxins or disease on cell development.
Liver Disease
Chronic liver disease is one of the most common causes of non-megaloblastic macrocytosis in clinical practice. The mechanism here is structural — it involves the red blood cell’s outer membrane rather than its DNA.
How it happens
The liver plays a central role in lipid (fat) metabolism. When liver function is impaired, excess cholesterol and phospholipids accumulate in the bloodstream. These lipids deposit directly onto the red blood cell membrane, expanding its surface area. The cell flattens and widens — becoming larger without any change in its DNA machinery.
Common causes
- Cirrhosis — chronic scarring of the liver from any cause, including hepatitis C, NAFLD/MASH (fatty liver disease), or long-term alcohol use.
- Obstructive jaundice — when bile flow is blocked, lipid exchange between the plasma and red cell membrane is significantly disrupted.
- Alcoholic liver disease — this often presents a ‘triple threat’: direct alcohol toxicity to the bone marrow, associated folate deficiency from poor nutrition, and liver-driven membrane changes to the red cell.
Treatment
There is no specific treatment for the macrocytosis itself. The priority is treating the underlying liver condition. Once liver function improves, lipid metabolism normalises and the MCV typically falls back into the normal range.
Clinical Clue: Round vs. Oval
When reviewing a blood film, the shape of the macrocytes gives you a quick clue. Megaloblastic macrocytes (from B12 or folate deficiency) are typically oval — they are called macro-ovalocytes. Macrocytes caused by liver disease tend to be round. You may also see target cells (codocytes) — red cells with a bullseye appearance caused by excess membrane — which strongly point towards liver pathology rather than a nutritional deficiency.
Hypothyroidism
Hypothyroidism — an underactive thyroid gland — is a classic but often overlooked cause of non-megaloblastic macrocytic anemia. The anemia is usually mild, but macrocytosis can sometimes be the first laboratory hint that thyroid function is declining.
How it happens
Thyroid hormones (T3 and T4) stimulate erythropoiesis — the process of making red blood cells in the bone marrow. When thyroid hormone levels fall, the body’s overall metabolism slows and its demand for oxygen drops, so red blood cell production decreases (Killeen & Adil, 2025). The exact reason why the cells become larger is not fully understood, but it appears to involve a combination of slower stem cell division and changes in red cell membrane lipid composition, similar to what happens in liver disease.
Common causes
- Hashimoto’s thyroiditis — the most common cause worldwide. Hashimoto’s is an autoimmune condition; importantly, people with one autoimmune disease are at higher risk of developing others. Pernicious anemia (also autoimmune) frequently co-exists. If the MCV is notably elevated in a hypothyroid patient, always screen for B12 deficiency too.
- Iatrogenic hypothyroidism — occurring after thyroidectomy (surgical removal of the thyroid) or radioactive iodine therapy for an overactive thyroid.
- Iodine deficiency — globally common, though less frequent in regions with iodised salt.
An important nuance: textbooks often describe the anemia of hypothyroidism as normocytic (normal cell size). However, up to 25% of hypothyroid patients develop mild macrocytosis, with MCV values typically between 100 and 105 fL. A rising MCV in a patient with fatigue and weight gain should prompt a thyroid-stimulating hormone (TSH) test.
Treatment
Thyroid hormone replacement — usually levothyroxine — resolves the macrocytosis entirely. Once the patient reaches a stable, normal thyroid state (euthyroidism), the MCV typically returns to the reference range within a few months as the red cell population turns over.
Clinical Clue: The Autoimmune Package Deal
When you find macrocytosis in a patient with hypothyroidism, don’t stop at the TSH. Hashimoto’s thyroiditis and pernicious anemia share an autoimmune basis and frequently co-exist as part of what is called Polyglandular Autoimmune Syndrome. Always check a serum B12 level to make sure you aren’t missing a megaloblastic process running in parallel.
Alcohol Abuse
Chronic alcohol consumption is arguably the most common cause of non-megaloblastic macrocytosis in everyday clinical practice. Alcohol exerts a multi-pronged assault on the bone marrow and red cell development — and crucially, it can raise the MCV before significant anemia or liver disease is even detectable.
How it happens
Alcohol causes macrocytosis through several overlapping mechanisms:
- Direct bone marrow toxicity — ethanol and its breakdown product, acetaldehyde, are directly toxic to developing red blood cell precursors, causing them to enlarge abnormally.
- Membrane disruption — alcohol interferes with lipid metabolism, leading to cholesterol accumulation in the red cell membrane and increasing cell size — a similar mechanism to liver disease.
- Nutritional depletion — people who drink heavily often eat poorly. Alcohol also directly impairs folate absorption in the gut and increases its excretion via the kidneys, creating a genuine folate deficiency on top of the direct toxic effects.
Key clinical indicators
- Isolated raised MCV — in many heavy drinkers, an MCV of 100–110 fL may be the only laboratory abnormality. Clinicians sometimes use this as a subtle marker for hidden alcohol use disorder.
- Vacuolisation — in acute alcohol toxicity, bone marrow biopsy may reveal characteristic vacuoles (small clear bubbles) inside developing red and white blood cell precursors.
- Sideroblastic changes — alcohol can interfere with haem (the iron-containing component of haemoglobin) synthesis, occasionally producing ringed sideroblasts in the marrow.
Treatment
The primary treatment is stopping alcohol and addressing nutritional deficits. Because folate deficiency is so common in this group, folic acid supplementation (1 mg daily) is almost always given as standard.
The good news: once alcohol intake stops, the MCV will begin to fall. It typically takes two to four months — roughly the lifespan of a red blood cell — for the MCV to return to normal.
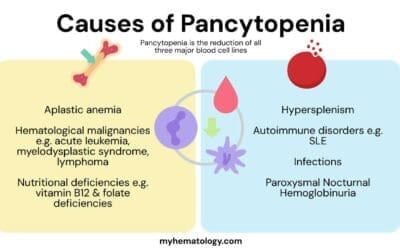
Myelodysplastic Syndromes (MDS)
Myelodysplastic syndromes (MDS) are a group of bone marrow disorders where the marrow is active and full of cells — but those cells are malformed and die before entering the bloodstream. The term dysplastic means abnormally shaped or developed. MDS is an important diagnosis to consider in older patients with unexplained macrocytic anemia that doesn’t improve with nutritional supplementation.
How it happens
MDS is a stem cell disorder. Genetic mutations prevent red blood cell precursors from maturing properly, producing a pattern of nuclear-cytoplasmic asynchrony very similar to megaloblastic anemia — but caused by DNA mutations rather than vitamin deficiency. These poorly developed cells often enter the circulation as large, misshapen macrocytes.
Common causes
- Primary (de novo) MDS — most common in patients over 65, arising from the gradual accumulation of genetic mutations over a lifetime.
- Secondary (treatment-related) MDS — seen in patients who have previously received chemotherapy or radiation therapy for other cancers, such as lymphoma or breast cancer.
What to look for
- A refractory macrocytic anemia — one that does not respond to B12, folate, or iron supplementation — should raise the suspicion of MDS.
- Pseudo-Pelger-Huët cells on the blood film — neutrophils (a type of white blood cell) with only two lobes instead of the usual three to five, often resembling a pair of spectacles or a dumbbell.
- Bone marrow biopsy — the gold standard for diagnosis. It typically reveals a hypercellular marrow (packed with developing cells) combined with peripheral cytopenia (low cell counts in the circulating blood).
Treatment
The management of Myelodysplastic Syndromes (MDS) is highly risk-stratified, focusing on improving quality of life and preventing transformation into Acute Myeloid Leukemia (AML). For lower-risk patients, the primary goal is managing symptomatic anemia and reducing transfusion dependency; this typically begins with supportive care and Erythropoiesis-Stimulating Agents (ESAs), but has recently expanded to include targeted therapies like luspatercept—specifically for those with ring sideroblasts—and imetelstat, a first-in-class telomerase inhibitor that targets malignant clones to restore healthy red cell production (Della Porta et al., 2024; Platzbecker et al., 2024). In higher-risk cases, treatment shifts toward more aggressive strategies such as hypomethylating agents (e.g., azacitidine) to slow disease progression, while allogeneic stem cell transplantation remains the only potentially curative option for eligible, fit individuals (National Comprehensive Cancer Network (NCCN) Guidelines for MDS (Version 1.2025/2026)).
Drug-Induced Macrocytosis
Several medications cause macrocytosis by interfering with DNA synthesis — technically producing a megaloblastic-type picture, but grouped separately in practice because the management is focused on the drug rather than vitamin supplementation.
How it happens
Most offending drugs block DNA polymerase or disrupt the metabolic pathways needed to make the building blocks of DNA (purines and pyrimidines). The result is the familiar nuclear-cytoplasmic asynchrony: the cell grows while the nucleus falls behind.
Key offenders
- Nitrous Oxide (N₂O) — Known colloquially as “laughing gas,” N₂O causes a dramatic, functional B12 deficiency. It irreversibly oxidizes the cobalt atom in Vitamin B12, rendering the vitamin completely inactive. This halts the conversion of homocysteine to methionine and inhibits DNA synthesis immediately (Saji et al., 2024).
- Hydroxyurea — used in sickle cell disease and myeloproliferative disorders. It almost universally causes macrocytosis (MCV 110–130 fL). In fact, a rising MCV is often used as a marker that the patient is taking their medication as prescribed.
- Zidovudine (AZT) — an older antiretroviral for HIV. A classic cause of marked macrocytosis, often without significant anaemia.
- Methotrexate — used in rheumatoid arthritis and cancer treatment. It blocks the enzyme dihydrofolate reductase, effectively mimicking a folate deficiency.
- Anticonvulsants (phenytoin, valproate) — interfere with folate absorption and metabolism in the gut and liver.
- Chemotherapy agents — cytarabine, 5-fluorouracil (5-FU), and azathioprine frequently produce large, stressed red blood cells.
Important threshold: if you encounter an MCV above 130 fL, your first suspicion should be drug-induced macrocytosis — particularly from hydroxyurea or zidovudine — or MDS. A simple nutritional deficiency rarely produces MCV values this extreme.
Treatment
Unlike nutritional anemias, drug-induced macrocytosis usually doesn’t respond to B12 or Folate supplementation unless a true deficiency co-exists. For N₂O toxicity, immediate cessation of exposure and high-dose B12 supplementation is required to “bypass” the inactivated stores. For other drugs, the MCV will typically remain elevated as long as the patient is on the medication. Treatment is only necessary if the patient becomes symptomatically anemic, in which case the drug dosage may need adjustment.
Copper Deficiency
Copper deficiency is an easily overlooked cause of macrocytic anemia — and a clinically important one. It is well known for producing a hematological masquerade: the blood count and bone marrow findings can be almost indistinguishable from Myelodysplastic Syndrome (MDS), leading to misdiagnosis in patients who simply need a mineral supplement (Kumar et al., 2004).
How it happens
Copper is an essential component of several enzymes involved in iron metabolism and red blood cell production — most notably hephaestin and ceruloplasmin. Without adequate copper, iron cannot be properly mobilised from the body’s storage sites or incorporated into hemoglobin. The result is ineffective red cell production with sideroblastic changes (abnormal iron accumulation in developing red cells). Copper deficiency also impairs white blood cell maturation, which is why it frequently causes neutropenia (a low white cell count) alongside the anemia.
Common causes
- Excess zinc intake — the most common hidden cause. High doses of zinc (from supplements or zinc-containing denture creams) trigger the production of a protein called metallothionein in the gut lining. This protein binds copper and traps it inside intestinal cells, blocking its absorption into the bloodstream.
- Bariatric (weight-loss) surgery — like vitamin B12, copper absorption depends heavily on the stomach and upper small intestine. Gastric bypass surgery significantly disrupts this process, putting patients at long-term risk (Gletsu-Miller & Wright, 2013).
- Malabsorption conditions — coeliac disease and inflammatory bowel disease can impair copper uptake from the gut.
- Total parenteral nutrition (TPN) — patients receiving long-term intravenous feeding without adequate trace element supplementation are at risk of developing copper deficiency over time.
The MDS mimicker: why this matters
Copper deficiency often presents with pancytopenia — low counts of red cells, white cells, and platelets — and ringed sideroblasts on bone marrow biopsy. These are also the hallmark findings of Myelodysplastic Syndrome. As a result, patients have been misdiagnosed with a serious bone marrow condition when the true cause was a correctable nutritional deficiency. If a patient presents with a refractory macrocytic anemia, pancytopenia, and bone marrow dysplasia, and MDS workup is inconclusive — check a serum copper and zinc level before accepting the MDS diagnosis (Halfdanarson et al., 2008).
Treatment
Treatment involves copper supplementation — oral in mild cases, intravenous in severe ones. Crucially, any source of excess zinc must be identified and stopped. Recovery is typically rapid: white cell counts begin to improve within days, and the anaemia resolves within weeks.
Clinical Clue: The Neurological Twin
Copper deficiency causes a neurological syndrome — copper deficiency myeloneuropathy — that is virtually identical to the subacute combined degeneration seen in B12 deficiency: loss of balance, tingling in the limbs, and progressive weakness. If a patient has B12-like neurological symptoms and macrocytic anemia, but their B12 levels are completely normal, check serum copper and zinc immediately. This diagnosis is easily missed and, if left untreated, can cause permanent neurological disability.
Reticulocytosis
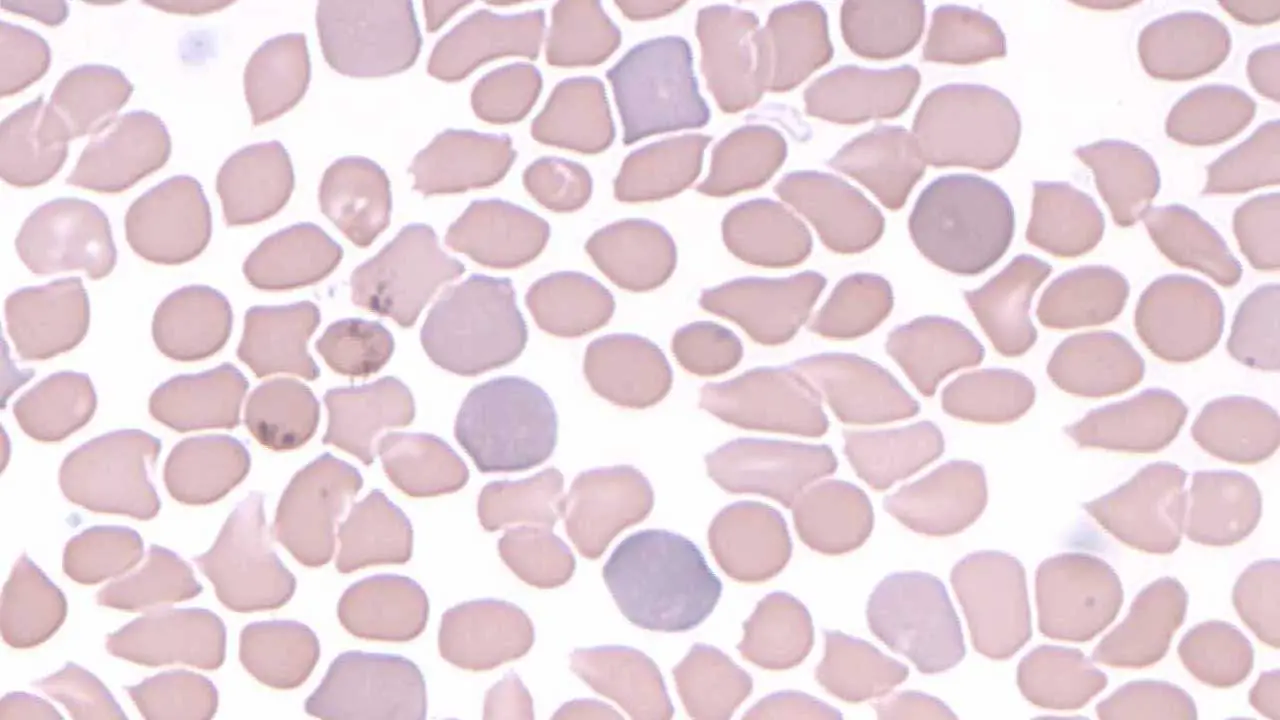
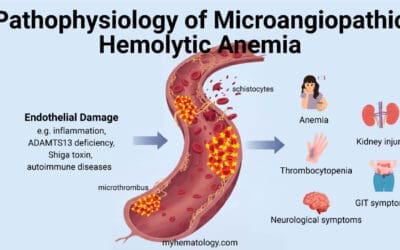
Reticulocytosis — an increase in circulating reticulocytes (immature red blood cells) — is an important and often underappreciated cause of a raised MCV. It is best understood as a pseudo-macrocytosis: the red cells are large not because of any defect in DNA synthesis or cell membrane, but simply because they are young.
On a blood film, reticulocytes appear slightly larger than mature red cells and stain with a faint blue-grey tint rather than the usual pink. This blue tinge, caused by residual ribosomal RNA inside the cell, is called polychromasia. Seeing many of these cells on a smear is a reassuring sign — it tells you the bone marrow is active and responding.
How it happens
When the body detects a sudden fall in red blood cells or oxygen-carrying capacity — from hemorrhage, hemolysis, or the start of nutritional treatment — the kidneys release erythropoietin (EPO). EPO signals the bone marrow to accelerate red cell production. To meet demand quickly, the marrow releases red blood cells before they have fully matured. These young reticulocytes are physically larger than mature erythrocytes (103–120 fL versus 80–100 fL) and carry enough remnant RNA to stain blue on a film.
Common causes
- Hemolytic anemias — rapid destruction of red blood cells (for example in sickle cell crisis, G6PD deficiency, or autoimmune hemolytic anemia) floods the circulation with reticulocytes as the marrow tries to compensate.
- Significant blood loss — acute hemorrhage triggers an immediate marrow response.
- Response to nutritional treatment — when a patient with iron, B12, or folate deficiency begins supplementation, a ‘reticulocyte flare’ occurs. As the marrow surges back to life, a wave of large young cells enters the blood, which can temporarily push the MCV even higher before it normalises.
Treatment
Reticulocytosis is a sign, not a disease. The reticulocyte count itself requires no treatment. The clinical task is to identify why the marrow is working overtime — whether that is active hemolysis, bleeding, or a response to therapy — and address the underlying cause. Once the trigger resolves and the red cell population matures, the MCV will return to the normal range on its own.
Clinical Clue: The “False” Macrocytosis
Always check the reticulocyte count before committing to an expensive workup for B12 deficiency or MDS. If the reticulocyte count is high — typically above 3–5% — the elevated MCV is likely a mathematical artefact caused by the large volume of young cells circulating in the blood. A high reticulocyte count tells you the bone marrow is working. A low reticulocyte count in the face of anemia tells you the bone marrow is failing. That single distinction shapes the entire diagnostic direction.
Summary of Non-Megaloblastic Causes
| Cause | Distinctive Smear Finding | Key Diagnostic Test |
| Liver Disease | Target Cells (Codocytes) | LFTs, Ultrasound |
| Alcohol Abuse | Round Macrocytes | GGT, Patient History |
| Hypothyroidism | Normal/Mild Macrocytes | TSH, Free T4 |
| MDS | Pseudo-Pelger-Huët cells | Bone Marrow Biopsy |
| Copper Deficiency | Neutropenia/Sideroblasts | Serum Copper & Zinc |
| Reticulocytosis | Polychromasia | Reticulocyte Count |
Approach to Macrocytic Anemia
When a patient presents with a raised MCV, the clinical approach is systematic. It moves from history and examination through to targeted laboratory testing — narrowing the possibilities at each stage.
Step 1: Clinical History
A focused history often points you toward the diagnosis before you order a single test. Key questions include:
- Symptoms — fatigue, weakness, shortness of breath, and pale skin are common to all anemias. Neurological symptoms (tingling, numbness, unsteady gait) specifically raise the concern for B12 deficiency.
- Diet — ask about intake of meat, fish, dairy, eggs, and vegetables. A strict plant-based diet without supplementation flags B12 risk; a diet poor in fresh produce flags folate risk.
- Alcohol — quantity and duration of alcohol use. Chronic heavy drinking is a common and easily missed cause.
- Medications and supplements — a full medication list is essential. Metformin, PPIs, methotrexate, anticonvulsants, and antiretrovirals can all contribute. Don’t forget over-the-counter supplements: high-dose zinc is a leading cause of copper deficiency and is easily missed if not specifically asked about.
- Past medical history — liver disease, autoimmune conditions, thyroid problems, gastrointestinal surgery, or a previous diagnosis of pernicious anemia are all relevant.
Step 2: Physical Examination
Physical findings support — and sometimes reveal — the underlying cause:
General and Dermatologic Findings
- Lemon-Yellow Skin Tinge — This classic sign is a hallmark of severe megaloblastic anemia, resulting from the combination of marked pallor (due to anemia) and mild jaundice (resulting from “ineffective erythropoiesis,” or intramedullary hemolysis of fragile megaloblasts).
- Hyperpigmentation — Chronic vitamin B12 deficiency may present with darkening of the skin, particularly on the knuckles, palms, and soles.
- Pale Nail Beds and Eyelids — General indicators of significant anemia where hemoglobin levels are severely reduced.
Oral and Mucosal Manifestations
- Angular Cheilitis — Inflammation and fissuring at the corners of the mouth are common in mixed anemias, often indicating a concurrent iron deficiency alongside macrocytic etiologies.
- Hunter’s Glossitis — The tongue may appear beefy-red, smooth, and “bald” due to the atrophy of lingual papillae, which is a frequent finding in B12 and folate deficiencies.
Neurologic and Psychiatric Signs
- “Megaloblastic Madness” — This refers to the cognitive and psychiatric manifestations of vitamin B12 deficiency, which can include depression, psychosis, memory loss, and dementia. These symptoms can occur even in the absence of hematologic abnormalities.
- Subacute Combined Degeneration (SCD) — Characterized by the loss of vibration and position sense due to demyelination of the dorsal and lateral columns of the spinal cord.
- Positive Romberg Sign — An indicator of sensory ataxia resulting from impaired proprioception.
Abdominal Findings
- Splenomegaly — While not universal, an enlarged spleen can be found in patients with hemolytic causes of macrocytosis or advanced Myelodysplastic Syndromes (MDS) where extramedullary hematopoiesis is present.
Step 3: Laboratory Investigations
Laboratory testing proceeds in tiers, from broad screening to targeted confirmatory tests.
Tier 1: Initial Screening
These tests confirm the presence of macrocytosis and provide clues about the bone marrow’s health.
- Complete Blood Count (CBC) —
- MCV > 100 fL: Confirms macrocytosis.
- Hemoglobin/Hematocrit: Determines the severity of the anemia.
- Pancytopenia: If white blood cells and platelets are also low, it suggests a bone marrow process like MDS or severe B12 deficiency.
- Reticulocyte Count —
- Elevated: Suggests the marrow is responding to blood loss or hemolysis (the “large” cells may just be young reticulocytes).
- Low/Normal: Suggests a production problem, like DNA synthesis failure or marrow suppression.
- Peripheral Blood Smear —
- Macro-ovalocytes + Hypersegmented Neutrophils: Highly indicative of Megaloblastic Anemia (B12/Folate).
- Round Macrocytes + Target Cells: Suggests Liver Disease.
- Basophilic Stippling: May suggest lead poisoning or sideroblastic processes.
Tier 2: Distinguishing Nutritional Deficiencies
If the smear suggests megaloblastic changes, the focus shifts to vitamins.
- Serum Vitamin B12 —
- < 200 pg/mL: Deficient.
- 200–400 pg/mL: “Gray zone”. Requires further testing with MMA.
- Serum or RBC Folate — RBC (Red Blood Cell) folate is often preferred as it reflects long-term stores, whereas serum folate can fluctuate based on the patient’s last meal. However, RBC folate can be falsely low in B12 deficiency because impaired B12-dependent methionine synthase activity traps folate as 5-methyltetrahydrofolate inside red cells, preventing its normal polyglutamate incorporation. This can lead to misdiagnosis of folate deficiency in a patient who actually has B12 deficiency.
- Methylmalonic Acid (MMA) & Homocysteine —
- High MMA + High Homocysteine: Confirms Vitamin B12 deficiency.
- Normal MMA + High Homocysteine: Confirms Folate deficiency.
- Holotranscobalamin (HoloTC / Active B12) — this active, transcobalamin-bound fraction of circulating B12 available to cells is now considered a more sensitive early marker of B12 deficiency than total serum B12. Because total B12 measures biologically inactive haptocorrin-bound B12 alongside active B12, it can remain deceptively normal in early deficiency states (Golding, 2016; Dastidar & Sikder, 2022). A study by Dastidar & Sikder (2022) demonstrated that active B12 was more sensitive (82.35% vs 65%) and specific than total B12 for detecting deficiency. Its omission from the diagnostic algorithm is a missed opportunity for clinical currency.
Tier 3: Specialized Testing
- Autoantibody testing (for pernicious anaemia) — anti-intrinsic factor antibodies are highly specific (confirmatory); anti-parietal cell antibodies are sensitive (screening).
- Thyroid-stimulating hormone (TSH) — to rule out hypothyroidism.
- Liver function tests (LFTs) — to investigate liver disease or alcohol-related damage. An elevated GGT is a useful marker of alcohol use.
- Bone marrow aspiration and biopsy — indicated when the cause remains unclear after Tier 1 and 2 investigations, or when MDS or leukaemia is suspected. It allows direct visualisation of megaloblasts and chromosomal analysis (cytogenetics).
Differential Diagnosis of Macrocytosis
| Investigation | Megaloblastic (B12/Folate) | Non-Megaloblastic (Liver/Alcohol) | Myelodysplastic Syndromes (MDS) | Drug-Induced (e.g., Hydroxyurea, AZT) |
| MCV | Often > 110 fL | Often 100–110 fL | Often 100–110 fL | Often > 110 fL (Frequently > 130 fL) |
| Blood Smear | Macro-ovalocytes, Hypersegmented Neutrophils | Round macrocytes, Target cells (liver) | Dysplastic red cells, Pseudo-Pelger-Huët cells | Extreme macrocytes, usually normal neutrophils |
| RDW | Elevated | Variable | Elevated (dimorphic population) | Variable to Elevated |
| MMA | Elevated (in B12 deficiency) | Normal | Normal | Normal (Except with Nitrous Oxide) |
| Bilirubin / LDH | Elevated (due to ineffective erythropoiesis) | Normal (unless direct liver damage) | Elevated (due to ineffective erythropoiesis) | Usually Normal |
| Key Secondary Finding | Low reticulocytes, potential pancytopenia | Elevated GGT / Abnormal LFTs | Pancytopenia, hypercellular marrow | History of specific pharmacotherapy |
The “Normal MCV” Diagnostic Trap (Mixed Anemias)
A patient can have severe B12 or folate deficiency but present with a normal MCV. This happens when a concurrent microcytic process — such as iron deficiency or thalassemia — offsets the macrocytosis, producing a falsely averaged normal result. The clue is a markedly elevated red cell distribution width (RDW), which reflects the wide variation in cell sizes. Always pair the MCV with an RDW and a peripheral blood smear to avoid missing this combination.
Frequently Asked Questions (FAQs)
What is the difference between macrocytic and megaloblastic anemia?
Macrocytic anemia is the broader term — it means any anemia where red blood cells are larger than normal (MCV > 100 fL), regardless of cause. Megaloblastic anemia is a specific subtype of macrocytic anemia caused by impaired DNA synthesis. All megaloblastic anemias are macrocytic, but not all macrocytic anemias are megaloblastic.
Who is at the highest risk for macrocytic anemia?
Several groups are at elevated risk:
- Strict vegans or vegetarians who don’t supplement B12
- Elderly individuals, in whom B12 absorption becomes less efficient with age
- People with malabsorption conditions such as coeliac disease or Crohn’s disease
- Individuals who drink heavily or have a history of alcohol use disorder
- People with liver disease
- Those taking long-term metformin, PPIs, methotrexate, or anticonvulsants
- People with autoimmune conditions, particularly pernicious anaemia
Is macrocytic anemia serious or curable?
It depends on the underlying cause. Some causes — like dietary B12 or folate deficiency — are straightforward to treat and fully reversible. Others — like MDS — are more complex and carry a different prognosis. The macrocytosis itself is rarely the danger; it is the condition driving it that determines seriousness.
Can macrocytic anemia cause blood clots?
Macrocytic anemia, particularly the megaloblastic type caused by Vitamin B12 or folate deficiency, can indeed increase the risk of blood clots through a secondary metabolic mechanism involving homocysteine. When these vitamins are deficient, the body cannot effectively convert homocysteine into methionine, leading to an accumulation of homocysteine in the bloodstream. This condition, called hyperhomocysteinemia, is pro-thrombotic; it causes oxidative stress and damage to the vascular endothelium (the lining of the blood vessels) and interferes with natural anticoagulant pathways, significantly raising the risk for venous thromboembolism (VTE), pulmonary embolisms, and arterial thrombosis (den Heijer et al., 1996; Cattaneo, 1997; den Heijer et al., 2007). While the abnormally large red blood cells themselves do not cause the clots, the metabolic “bottleneck” created by the vitamin deficiency makes the circulatory environment much more prone to coagulation.
Can macrocytic anemia cause cancer?
Macrocytic anemia is a blood condition, not a form of cancer. However, the underlying cause behind the macrocytosis might be associated with a higher risk of certain cancers like myelodysplastic syndrome (MDS).
Which drugs cause macrocytic anemia?
Macrocytosis has been reported as a side effect of various medications, including:
- Antiviral medications: Reverse transcriptase inhibitors like zidovudine, lamivudine, and stavudine.
- Anticonvulsant medications: Valproic acid and phenytoin.
- Folate antagonists: Methotrexate.
- Chemotherapy drugs: Alkylating agents, pyrimidine and purine inhibitors.
- Other medications: Sulfasalazine (anti-inflammatory), pyrimethamine, trimethoprim, sulfamethoxazole, metformin, and nitrous oxide.
Can you have a normal MCV but still have vitamin B12 deficiency?
Yes — and this is an important pitfall. If iron deficiency (which makes cells smaller) coexists with B12 deficiency (which makes cells larger), the automated MCV average may appear falsely normal. An elevated RDW and a peripheral blood smear are essential to detect this mixed picture.
Is the Schilling test still used to diagnose pernicious anemia?
No. The Schilling test is now largely obsolete. Pernicious anaemia is diagnosed today through serum B12 measurement, MMA levels, and autoantibody testing (anti-intrinsic factor and anti-parietal cell antibodies).
How long does it take for macrocytic anemia to resolve with treatment?
The bone marrow responds quickly once B12 or folate supplementation begins. A surge of new red blood cells (reticulocytosis) typically peaks within five to seven days. Haemoglobin and MCV usually normalise over six to eight weeks. Neurological symptoms take longer and may not recover fully if the deficiency was prolonged and severe.
Glossary of Related Medical Terms
- Erythropoiesis — the process of producing red blood cells in the bone marrow.
- Glossitis — inflammation of the tongue; in B12 or folate deficiency it appears smooth, red, and shiny — a sign called atrophic glossitis.
- Homocysteine — an amino acid that requires both B12 and folate to be metabolised. Elevated levels signal a deficiency in one or both vitamins.
- Macrocytosis — the presence of abnormally large red blood cells (MCV > 100 fL) in the peripheral blood, with or without anaemia.
- Megaloblast — an abnormally large, immature red blood cell precursor found in the bone marrow, displaying nuclear-cytoplasmic asynchrony: the nucleus is underdeveloped relative to the cytoplasm.
- Methylmalonic acid (MMA) — a compound that accumulates specifically when B12 is deficient. Elevated MMA is a highly specific marker for B12 deficiency.
- Nuclear-cytoplasmic asynchrony — a pattern seen in developing blood cells where the cell body (cytoplasm) continues to grow normally while the nucleus (containing the DNA) lags behind. It is the cellular hallmark of megaloblastic anemia.
- Ceruloplasmin — a copper-carrying protein in the blood. Low ceruloplasmin levels confirm copper deficiency and are used alongside serum copper in diagnosis.
- Copper deficiency myeloneuropathy — a neurological condition caused by copper deficiency, producing damage to the spinal cord and peripheral nerves. It closely resembles the subacute combined degeneration seen in B12 deficiency.
- Metallothionein — a protein produced in the gut wall in response to high zinc intake. It traps copper inside intestinal cells, preventing absorption — the key mechanism by which excess zinc causes copper deficiency.
- Polychromasia — the appearance of red blood cells with a blue-grey tint on a blood film, indicating the presence of reticulocytes (immature red cells) that still contain ribosomal RNA.
- Red cell distribution width (RDW) — a laboratory measure of the variation in red blood cell sizes. A high RDW means the cells vary widely in size — useful for detecting mixed anemias.
- Reticulocyte — an immature red blood cell newly released from the bone marrow. Reticulocytes are larger than mature red cells (103–120 fL) and contain remnants of ribosomal RNA, giving them a blue tinge on a stained blood film.
- Reticulocytosis — an elevated reticulocyte count in the peripheral blood, indicating that the bone marrow is responding actively to anaemia, blood loss, or haemolysis. In the context of macrocytic anemia, a high reticulocyte count suggests pseudo-macrocytosis rather than a defect in DNA synthesis or cell membrane structure.
Disclaimer: This article is intended for informational purposes only and is specifically targeted towards medical students. It is not intended to be a substitute for informed professional medical advice, diagnosis, or treatment. While the information presented here is derived from credible medical sources and is believed to be accurate and up-to-date, it is not guaranteed to be complete or error-free. See additional information.
References
- Anemia: Diagnosis and Treatment (Willis, 2016).
- Management of Anemia: A Comprehensive Guide for Clinicians (Provenzano et al., 2018)
- Goldberg S, Hoffman J. Clinical Hematology Made Ridiculously Simple, 1st Edition: An Incredibly Easy Way to Learn for Medical, Nursing, PA Students, and General Practitioners (MedMaster Medical Books). 2021.
- Butler, C. C., Vidal-Alaball, J., Cannings-John, R., McCaddon, A., Hood, K., Papaioannou, A., Mcdowell, I., & Goringe, A. (2006). Oral vitamin B12 versus intramuscular vitamin B12 for vitamin B12 deficiency: a systematic review of randomized controlled trials. Family practice, 23(3), 279–285. https://doi.org/10.1093/fampra/cml008
- Kuzminski, A. M., Del Giacco, E. J., Allen, R. H., Stabler, S. P., & Lindenbaum, J. (1998). Effective treatment of cobalamin deficiency with oral cobalamin. Blood, 92(4), 1191–1198. https://doi.org/10.1182/blood.V92.4.1191
- Mazur, M., Ndokaj, A., Salerno, C., Vallone, J., Ardan, R., Bietolini, S., Carrouel, F., Wilk, A., Sarig, R., Ottolenghi, L., & Bourgeois, D. (2025). Efficacy of sublingual and oral vitamin B12 versus intramuscular administration: insights from a systematic review and meta-analysis. Frontiers in pharmacology, 16, 1602976. https://doi.org/10.3389/fphar.2025.1602976.
- Saji AM, Lui F, De Jesus O. Spinal Cord Subacute Combined Degeneration. [Updated 2024 Apr 21]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2026 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK560728/.
- Bernetti, C., Cea, L., Buoso, A., Greco, F., Rossi, M., Pilato, F., Calandrelli, R., Di Gennaro, G., Di Lazzaro, V., Zobel, B. B., & Mallio, C. A. (2025). A Comprehensive Overview of Subacute Combined Degeneration: MRI Diagnostic Challenges and Treatment Pathways. Brain sciences, 15(9), 972. https://doi.org/10.3390/brainsci15090972.
- Ames, B. N., Elson-Schwab, I., & Silver, E. A. (2002). High-dose vitamin therapy stimulates variant enzymes with decreased coenzyme binding affinity (increased K(m)): relevance to genetic disease and polymorphisms. The American journal of clinical nutrition, 75(4), 616–658. https://doi.org/10.1093/ajcn/75.4.616
- Wickramasinghe, S. N., & Fida, S. (1994). Bone marrow cells from vitamin B12- and folate-deficient patients misincorporate uracil into DNA. Blood, 83(6), 1656–1661. https://doi.org/10.1182/blood.V83.6.1656.1656
- Chon, J., Field, M. S., & Stover, P. J. (2019). Deoxyuracil in DNA and disease: Genomic signal or managed situation?. DNA repair, 77, 36–44. https://doi.org/10.1016/j.dnarep.2019.02.014.
- Green, R., & Miller, J. W. (1999). Folate deficiency beyond megaloblastic anemia: hyperhomocysteinemia and other manifestations of dysfunctional folate status. Seminars in hematology, 36(1), 47–64.
- Kumar, N., Gross, J. B., Jr, & Ahlskog, J. E. (2004). Copper deficiency myelopathy produces a clinical picture like subacute combined degeneration. Neurology, 63(1), 33–39. https://doi.org/10.1212/01.wnl.0000132644.52613.fa
- Halfdanarson, T. R., Kumar, N., Li, C. Y., Phyliky, R. L., & Hogan, W. J. (2008). Hematological manifestations of copper deficiency: a retrospective review. European journal of haematology, 80(6), 523–531. https://doi.org/10.1111/j.1600-0609.2008.01050.x.
- Gletsu-Miller, N., & Wright, B. N. (2013). Mineral malnutrition following bariatric surgery. Advances in nutrition (Bethesda, Md.), 4(5), 506–517. https://doi.org/10.3945/an.113.004341.
- Della Porta, M. G., Garcia-Manero, G., Santini, V., Zeidan, A. M., Komrokji, R. S., Shortt, J., Valcárcel, D., Jonasova, A., Dimicoli-Salazar, S., Tiong, I. S., Lin, C. C., Li, J., Zhang, J., Pilot, R., Kreitz, S., Pozharskaya, V., Keeperman, K. L., Rose, S., Prebet, T., Lai, Y., … Platzbecker, U. (2024). Luspatercept versus epoetin alfa in erythropoiesis-stimulating agent-naive, transfusion-dependent, lower-risk myelodysplastic syndromes (COMMANDS): primary analysis of a phase 3, open-label, randomised, controlled trial. The Lancet. Haematology, 11(9), e646–e658. https://doi.org/10.1016/S2352-3026(24)00203-5
- Platzbecker, U., Santini, V., Fenaux, P., Sekeres, M. A., Savona, M. R., Madanat, Y. F., Díez-Campelo, M., Valcárcel, D., Illmer, T., Jonášová, A., Bělohlávková, P., Sherman, L. J., Berry, T., Dougherty, S., Shah, S., Xia, Q., Sun, L., Wan, Y., Huang, F., Ikin, A., … Zeidan, A. M. (2024). Imetelstat in patients with lower-risk myelodysplastic syndromes who have relapsed or are refractory to erythropoiesis-stimulating agents (IMerge): a multinational, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet (London, England), 403(10423), 249–260. https://doi.org/10.1016/S0140-6736(23)01724-5
- https://www.nccn.org/patients/guidelines/content/PDF/mds-patient.pdf
- Golding P. H. (2016). Holotranscobalamin (HoloTC, Active-B12) and Herbert’s model for the development of vitamin B12 deficiency: a review and alternative hypothesis. SpringerPlus, 5(1), 668. https://doi.org/10.1186/s40064-016-2252-z
- Dastidar, R., & Sikder, K. (2022). Diagnostic reliability of serum active B12 (holo-transcobalamin) in true evaluation of vitamin B12 deficiency: Relevance in current perspective. BMC research notes, 15(1), 329. https://doi.org/10.1186/s13104-022-06224-8
- den Heijer, M., Koster, T., Blom, H. J., Bos, G. M., Briet, E., Reitsma, P. H., Vandenbroucke, J. P., & Rosendaal, F. R. (1996). Hyperhomocysteinemia as a risk factor for deep-vein thrombosis. The New England journal of medicine, 334(12), 759–762. https://doi.org/10.1056/NEJM199603213341203
- Cattaneo M. (1997). Hyperhomocysteinemia: a risk factor for arterial and venous thrombotic disease. International journal of clinical & laboratory research, 27(3), 139–144. https://doi.org/10.1007/BF02912449.
- den Heijer, M., Willems, H. P., Blom, H. J., Gerrits, W. B., Cattaneo, M., Eichinger, S., Rosendaal, F. R., & Bos, G. M. (2007). Homocysteine lowering by B vitamins and the secondary prevention of deep vein thrombosis and pulmonary embolism: A randomized, placebo-controlled, double-blind trial. Blood, 109(1), 139–144. https://doi.org/10.1182/blood-2006-04-014654
- Killeen RB, Adil A. Macrocytic Anemia. [Updated 2025 Apr 4]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2026 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK459295/