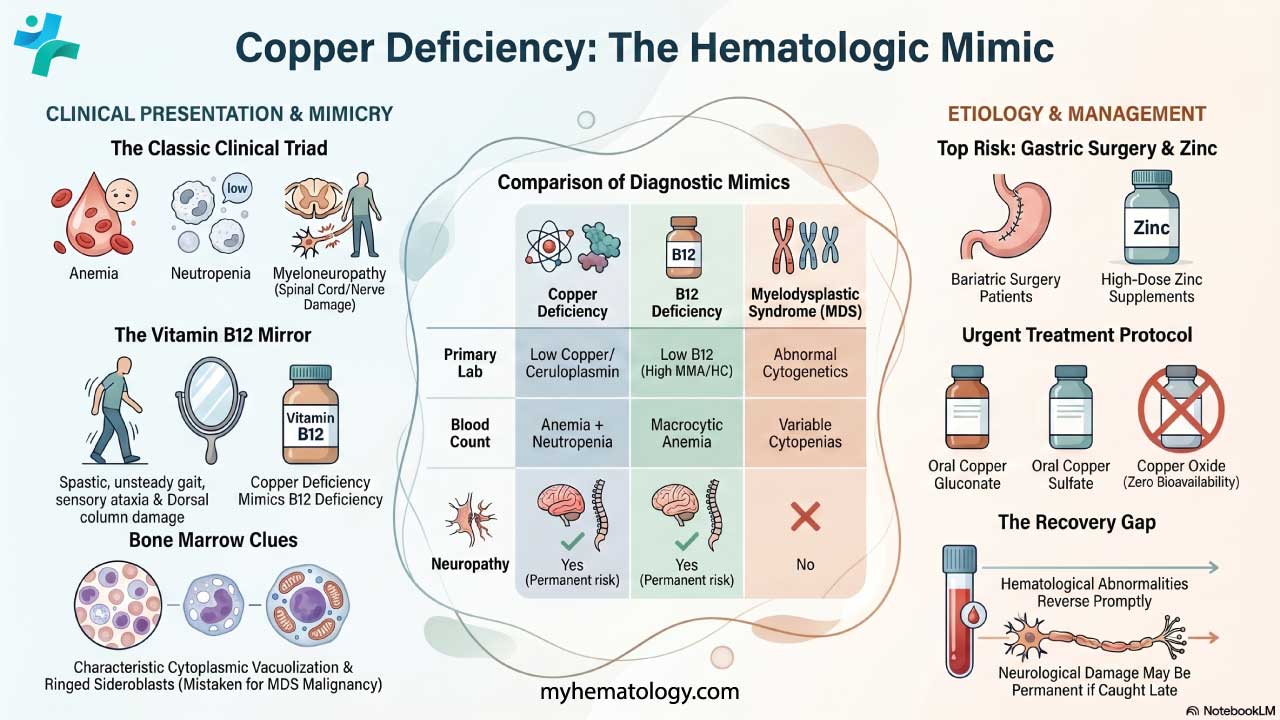
Key Takeaways
Anemia of Chronic Disease also known as Anemia of Inflammation is a type of anemia that develops as a consequence of chronic inflammatory conditions in the body.
- Causes ▾:
- Chronic infections
- Autoimmune diseases
- Other chronic inflammatory conditions e.g. chronic kidney disease
- Sarcoidosis
- Pathophysiology ▾: Chronic inflammation disrupts red blood cell production through multiple mechanisms:
- Suppressed EPO Production: Inflammation suppresses the production of erythropoietin (EPO), a hormone crucial for stimulating red blood cell production.
- Iron Dysregulation: Despite normal or elevated iron stores, hepcidin (an inflammatory protein) limits iron availability for red blood cell production.
- Shortened Red Blood Cell Lifespan: Chronic inflammation can slightly shorten the lifespan of circulating red blood cells.
- Signs and Symptoms ▾: Often has non-specific symptoms like fatigue, weakness, and shortness of breath. Symptoms can be easily overlooked.
- Laboratory Diagnosis ▾:
- CBC and PBF: Can be microcytic (small red blood cells) or normocytic (normal-sized red blood cells). Low reticulocyte count (impaired red blood cell production).
- Iron studies: Low serum iron with normal or elevated ferritin (iron entrapment, not deficiency). Low transferrin saturation (reflects limited iron availability).
- ESR and CRP: Elevated
- Differential Diagnosis ▾: Important to distinguish ACD from other types of anemia like iron deficiency anemia, vitamin B12 or folate deficiency anemia, and aplastic anemia through clinical features, laboratory tests, and sometimes bone marrow examination.
- Treatment ▾: Focuses on addressing the underlying chronic inflammatory condition. Erythropoietin-Stimulating Agents (ESAs) might be considered in specific cases, but their effectiveness is limited in ACD.
*Click ▾ for more information
Introduction
Anemia of Chronic Disease also known as Anemia of Inflammation is a type of anemia that develops as a consequence of chronic inflammatory conditions in the body. These chronic inflammatory conditions can be diverse, ranging from infections and autoimmune diseases to chronic kidney disease and cancer.
There are several key factors contributing to Anemia of Chronic Disease (Anemia of Inflammation) being one of the most common types of anemia:
- High Prevalence of Chronic Diseases: Chronic inflammatory conditions like infections, autoimmune diseases, and chronic kidney disease are widespread globally. As the prevalence of these underlying conditions rises, so does the likelihood of developing Anemia of Chronic Disease (Anemia of Inflammation) as a secondary consequence.
- Multiple Pathways to Anemia of Chronic Disease (Anemia of Inflammation): Chronic inflammation disrupts red blood cell production in several ways. Suppressed EPO production, iron dysregulation due to hepcidin, and shortened red blood cell lifespan all contribute to Anemia of Chronic Disease (Anemia of Inflammation) development. This multifaceted approach to disrupting red blood cell production makes Anemia of Chronic Disease (Anemia of Inflammation) more likely to occur.
- Subtle Symptoms in Early Stages: Anemia of Chronic Disease (Anemia of Inflammation) often presents with mild, non-specific symptoms like fatigue and weakness, which can be easily overlooked or attributed to the underlying chronic illness itself. This can lead to delayed diagnosis and potentially higher prevalence estimates as some cases might go undetected.
- Aging Population: As the global population ages, the prevalence of chronic diseases associated with aAnemia of Chronic Disease (Anemia of Inflammation), such as rheumatoid arthritis and heart failure, is also expected to rise. This demographic shift might further contribute to the increasing prevalence of Anemia of Chronic Disease (Anemia of Inflammation).
- Improved Diagnostics, But Still Challenging: While diagnostic techniques for anemia have improved, definitively diagnosing Anemia of Chronic Disease (Anemia of Inflammation) can still be challenging, especially in its early stages. The presence of other types of anemia alongside Anemia of Chronic Disease (Anemia of Inflammation) can further complicate diagnosis. This might lead to underdiagnosis and potentially contribute to a higher perceived prevalence.
Definition of Anemia
Anemia is a condition in which the blood has a reduced ability to carry oxygen. Anemia is defined as when the hemoglobin level in the blood falls below the reference or normal range for the particular age and gender of the person. This can happen due to three main reasons:
- Low number of red blood cells: Red blood cells are the carriers of hemoglobin, a protein that binds to oxygen in the lungs and delivers it throughout the body. If we don't have enough red blood cells, our blood will have less capacity to transport oxygen.
- Reduced amount of hemoglobin: Hemoglobin is the iron-rich protein inside red blood cells that gives blood its red color. If there is a deficiency in hemoglobin, even with a normal number of red blood cells, the blood will be less efficient at carrying oxygen.
- Abnormal hemoglobin function: In some cases, the hemoglobin itself might be abnormal and not function properly, even if the red blood cell count and hemoglobin quantity are within normal range. This can impair oxygen delivery.
Types of Anemia
Anemia can be classified in several ways, but one of the most approaches is based on the size of the red blood cells morphologically.
Classification Based on Red Blood Cell Size
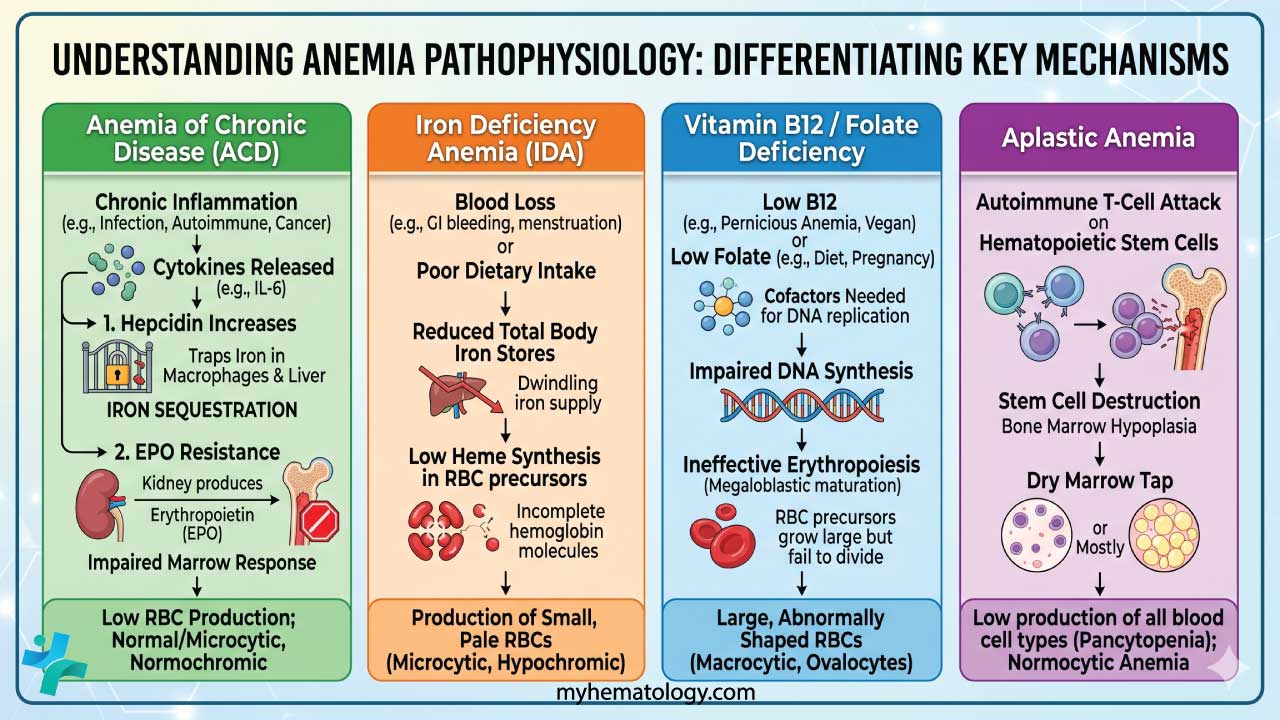
- Microcytic Anemia: This type is characterized by smaller than normal red blood cells. Iron deficiency anemia is the most common cause, where the body lacks sufficient iron to produce healthy red blood cells. Other causes can include chronic blood loss or conditions affecting iron absorption.
- Normocytic Anemia: In this type, the red blood cells are normal-sized. Normocytic anemia can arise from various causes, including chronic diseases (Anemia of Chronic Disease (Anemia of Inflammation)), bone marrow problems (aplastic anemia), or hemolytic anemia (where red blood cells are destroyed prematurely).
- Macrocytic Anemia: This type features red blood cells larger than normal. It's often linked to deficiencies in vitamin B12 or folate, both essential for red blood cell production. Other causes can include liver disease or alcohol abuse.
Additional Classification Systems
While red blood cell size is a helpful initial classification, anemia can also be categorized based on:
- Hemoglobin level: This considers the severity of anemia by classifying anemia based on hemoglobin concentration, such as normal, mild, moderate or severe.
- Cause of the Anemia: This delves deeper into the underlying factors causing the anemia, such as deficiencies, chronic conditions, or blood loss.
Causes of Anemia of Chronic Disease (Anemia of Inflammation)
Chronic Infections
- Bacterial infections like tuberculosis, osteomyelitis (bone infection), and endocarditis (infection of the heart valves)
- Viral infections like HIV/AIDS and hepatitis B or C
- Parasitic infections like malaria
Autoimmune Diseases
- Rheumatoid arthritis
- Systemic lupus erythematosus (SLE)
- Inflammatory bowel disease (IBD) including Crohn's disease and ulcerative colitis
Other Chronic Inflammatory Conditions
- Chronic kidney disease (CKD)
- Certain cancers (lymphoma, carcinoma, sarcoma)
Additional Less Common Causes
- Certain autoimmune vasculitides (inflammation of blood vessels)
- Sarcoidosis (inflammatory disease affecting multiple organs)
Pathophysiology of Anemia of Inflammation
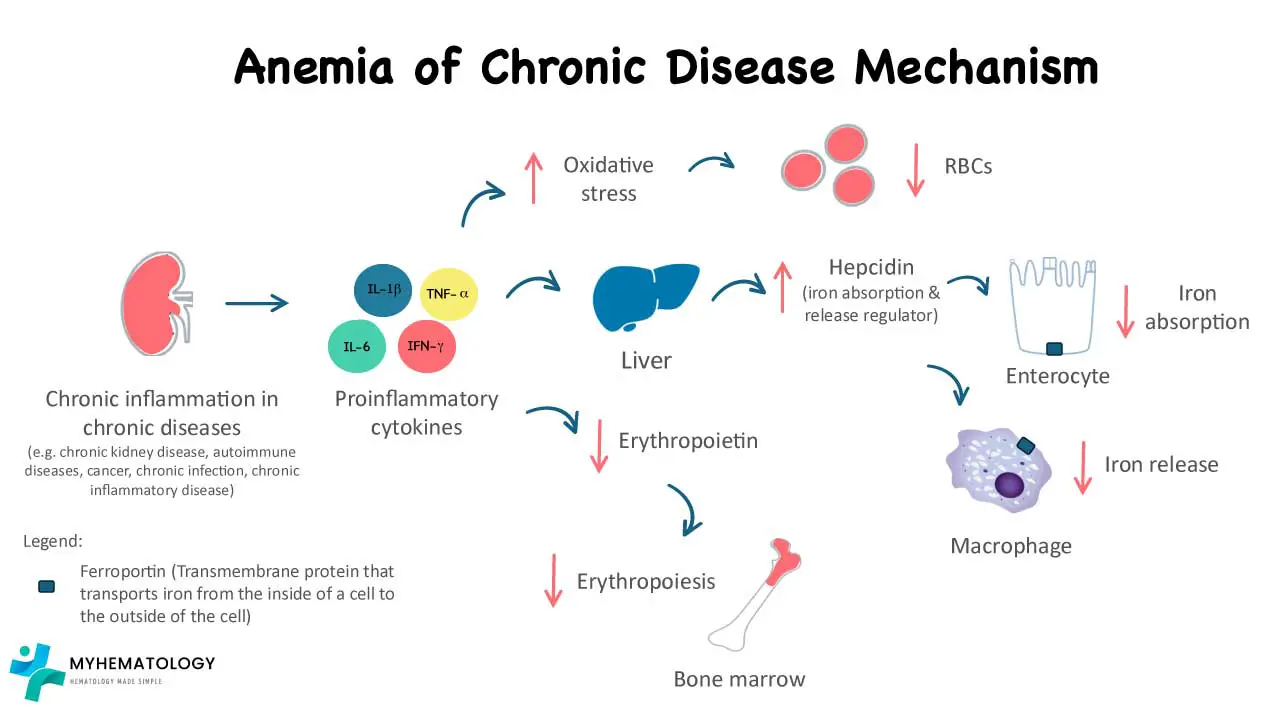
Anemia of Chronic Disease (Anemia of Inflammation) arises from a complex interplay between the chronic inflammatory response and several factors affecting red blood cell production.
Chronic Inflammation and Cytokines
Chronic inflammatory conditions trigger the release of various inflammatory cytokines, including interleukin-1 (IL-1) and interleukin-6 (IL-6).
- IL-1: This cytokine directly suppresses the production of erythropoietin (EPO) by the kidneys. EPO is a hormone crucial for stimulating red blood cell production in the bone marrow. Reduced EPO levels lead to a decrease in red blood cell production.
- IL-6: While its exact role is still being explored, IL-6 is believed to contribute to iron dysregulation and potentially shorten the lifespan of red blood cells.
Iron Dysregulation and Hepcidin
Chronic inflammation leads to the production of a protein called hepcidin by the liver. Hepcidin binds to ferroportin, a protein on the surface of macrophages and intestinal enterocytes, which are responsible for iron release and absorption, respectively, regulating iron release from storage sites in the body (macrophages and hepatocytes).
In Anemia of Chronic Disease (Anemia of Inflammation), even though total body iron stores might be normal or even elevated, hepcidin restricts iron release, making it less available for hemoglobin synthesis in red blood cells. This creates a state of "functional iron deficiency" within the bone marrow, despite having sufficient iron stores.
Beyond the Hepcidin-Ferroportin Axis
While hepcidin-mediated iron sequestration remains the central feature of Anemia of Chronic Disease (Anemia of Inflammation), modern hematology identifies a multi-front assault on erythropoiesis driven by inflammatory signaling. The pathophysiology can be distilled into four key mechanisms:
- Hepcidin-ERFE Dysregulation: In health, EPO stimulates the production of Erythroferrone (ERFE) from erythroblasts to suppress hepcidin and mobilize iron. In Anemia of Chronic Disease (Anemia of Inflammation), the overwhelming IL-6 inflammatory drive bypasses this feedback loop, keeping hepcidin levels high and rendering the "erythroid demand" signal ineffective.
- Erythropoietin (EPO) Resistance: Pro-inflammatory cytokines, specifically TNF-α and IL-1β, do more than just suppress EPO production; they induce peripheral resistance. These cytokines disrupt the JAK2/STAT5 signaling pathway within erythroid progenitor cells, essentially "silencing" the marrow's response to both endogenous and exogenous EPO.
- Accelerated Erythrophagocytosis: Inflammation triggers a hyperactive state in splenic and hepatic macrophages. This leads to the premature destruction of circulating red blood cells through increased phagocytic activity, significantly shortening the standard 120-day lifespan and increasing the demand on an already suppressed marrow.
- Marrow Microenvironment Stress: The accumulation of Reactive Oxygen Species (ROS) and the depletion of cellular antioxidants within the hematopoietic niche create a pro-apoptotic environment. This oxidative stress impairs the maturation of hematopoietic stem cells, leading to "ineffective erythropoiesis" where cells die before they can ever reach the reticulocyte stage.
While iron stores might appear normal, the body struggles to utilize it effectively for red blood cell production due to hepcidin's action.
Signs and Symptoms of Anemia of Chronic Disease (ACD)
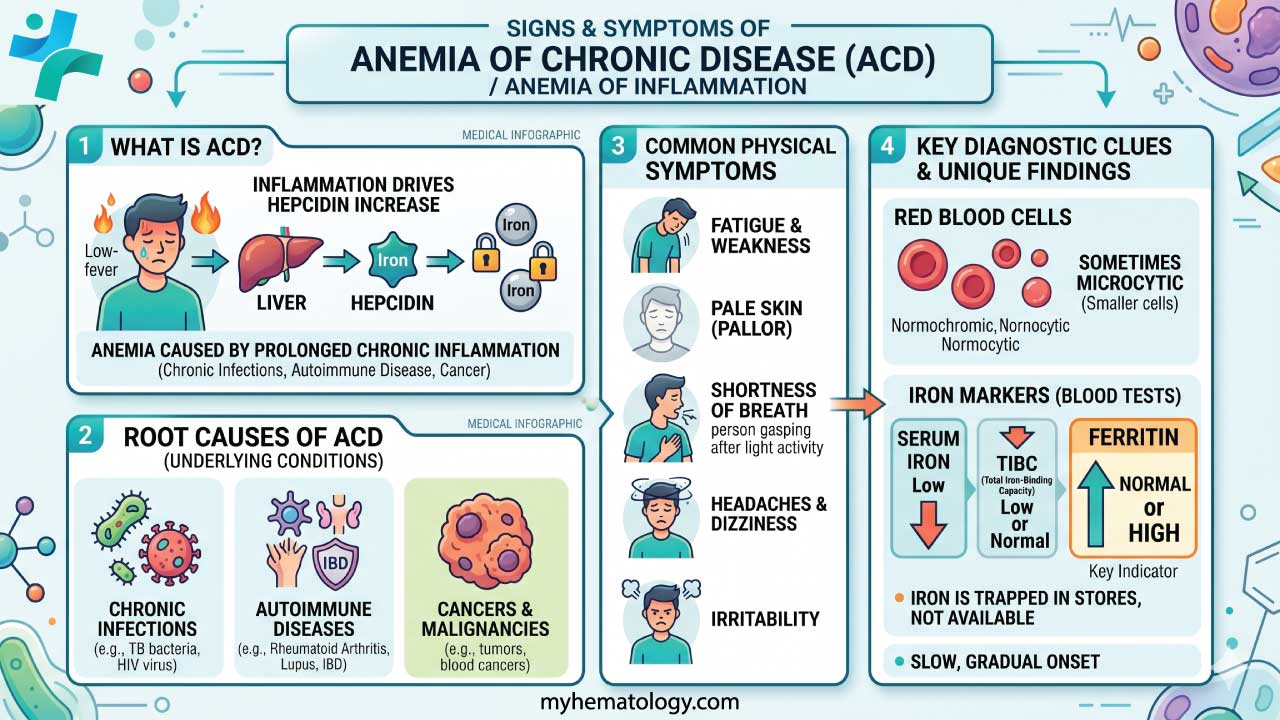
Anemia of chronic disease (ACD) is often a subtle condition, and its symptoms can be easily overlooked or attributed to the underlying chronic illness itself.
General Symptoms
- Fatigue and weakness
- Shortness of breath, especially during exertion
- Pale skin
- Dizziness or lightheadedness
- Difficulty concentrating or thinking clearly (cognitive difficulties)
- Decreased exercise tolerance
Additional Symptoms
- Chest pain
- Headaches
- Restlessness or difficulty sleeping
- Cold hands and feet
- Loss of appetite
Complications of Anemia of Chronic Disease (Anemia of Inflammation)
While Anemia of Chronic Disease (Anemia of Inflammation) itself is rarely life-threatening, it can lead to various complications that can significantly impact a patient's quality of life.
- Worsened Heart Failure Symptoms: In patients with existing heart failure, Anemia of Chronic Disease (Anemia of Inflammation) can worsen symptoms like shortness of breath, fatigue, and exercise intolerance. This is because anemia reduces the oxygen-carrying capacity of the blood, making it even harder for the heart to pump blood effectively.
- Increased Risk of Infections: A well-functioning immune system relies on adequate oxygen delivery. Anemia of Chronic Disease (Anemia of Inflammation) can impair the immune system's ability to fight infections, potentially increasing the risk of developing infections or experiencing slower healing after an infection.
- Impaired Quality of Life: The fatigue, weakness, and shortness of breath associated with Anemia of Chronic Disease (Anemia of Inflammation) can significantly impact a person's daily activities, work performance, and overall well-being. This can lead to feelings of frustration, social isolation, and decreased quality of life.
- Increased Risk of Hospitalization: Patients with Anemia of Chronic Disease (Anemia of Inflammation) and its complications might require more frequent hospital admissions due to issues like worsening heart failure symptoms or infections.
- Cognitive Impairment (in some cases): Chronic anemia can affect oxygen delivery to the brain, potentially leading to problems with memory, concentration, and overall cognitive function.
Laboratory Investigations
To provide a comprehensive diagnostic profile of Anemia of Chronic Disease (Anemia of Inflammation), laboratory investigations must focus on identifying the "iron-block" mechanism and differentiating it from absolute iron deficiency.
Routine Hematology (CBC & PBF)
The initial workup usually reveals a mild-to-moderate anemia, typically with hemoglobin (Hb) levels between 8.0 and 9.5 g/dL.
- MCV and MCH: Generally normocytic and normochromic initially. However, in long-standing cases or severe inflammation, it can progress to a microcytic (MCV < 80 fL) pattern, mimicking iron deficiency.
- Reticulocyte Count: Characteristically low (low Reticulocyte Production Index). This reflects an "underproduction" anemia due to the bone marrow's inability to respond to EPO.
- Peripheral Blood Film (PBF): Often unremarkable, showing normocytic cells. If microcytosis is present, it is usually less pronounced than in severe Iron Deficiency Anemia (IDA).
Standard Iron Studies
This is the most critical area for clinical differentiation. In Anemia of Chronic Disease (Anemia of Inflammation), iron is present in the body but "locked away."
| Parameter | Result in ACD | Pathophysiological Reasoning |
| Serum Iron (Fe) | Low | Reduced release of iron from macrophages into the plasma. |
| Ferritin | Normal or High | Reflects adequate stores and its role as an acute-phase reactant. |
| TIBC | Low to Normal | Transferrin production is downregulated by inflammation. |
| Transferrin Saturation | Low | Typically 15 - 20%; low due to the drop in serum iron. |
Advanced Diagnostic Markers
For cases where Anemia of Chronic Disease (Anemia of Inflammation) and IDA coexist (a common clinical challenge), standard iron studies may be inconclusive.
- Soluble Transferrin Receptor (sTfR): Unlike ferritin, sTfR is not affected by inflammation. It is normal in Anemia of Chronic Disease (Anemia of Inflammation) but elevated in IDA.
- sTfR/log Ferritin Index: This ratio is the most accurate non-invasive method to identify "functional" iron deficiency.
- An index < 1 suggests pure Anemia of Chronic Disease (Anemia of Inflammation).
- An index > 2 suggests IDA or Anemia of Chronic Disease (Anemia of Inflammation) with concurrent IDA.
- Reticulocyte Hemoglobin Content (CHr / RET-He): This measures the iron available to the red blood cells within the last 48–72 hours. It is a real-time snapshot of "iron-restricted erythropoiesis" and will be low even if ferritin is high.
- Serum Hepcidin: While not yet a routine bedside test, elevated hepcidin confirms the mechanistic cause of Anemia of Chronic Disease (Anemia of Inflammation).
Inflammatory Markers
To confirm the underlying "chronic" or "inflammatory" state, systemic markers are essential:
- C-Reactive Protein (CRP) & ESR: Usually significantly elevated. A normal CRP should prompt a search for alternative causes of anemia.
- Cytokine Profile: In research settings, IL-6 levels are measured, as IL-6 is the primary driver of hepcidin production.
Bone Marrow Examination (Rarely Required)
While rarely performed solely for Anemia of Chronic Disease (Anemia of Inflammation) it remains the gold standard for assessing iron distribution.
- In Anemia of Chronic Disease (Anemia of Inflammation): Iron is visible and often increased within macrophages (the storage safe), but notably absent or reduced within the developing erythroblasts (sideroblasts).
- In IDA: Iron is absent from both macrophages and erythroblasts.
Diagnostic Summary Table: ACD vs. IDA
| Marker | Anemia of Chronic Disease (ACD) | Iron Deficiency Anemia (IDA) |
| Serum Iron | Low | Low |
| Ferritin | Normal / High | Low |
| TIBC | Low / Normal | High |
| sTfR | Normal | High |
| Marrow Iron | Present (in macrophages) | Absent |
Differential Diagnosis
Distinguishing Anemia of Chronic Disease (Anemia of Inflammation) from other common anemias requires a systematic look at red cell indices, iron dynamics, and the involvement of other cell lines. While Anemia of Chronic Disease (Anemia of Inflammation) is often normocytic, its overlap with microcytic (IDA) and macrocytic (B12/Folate) categories makes a broad differential essential.
ACD vs. Common Anemias
| Feature | ACD (Anemia of Inflammation) | Iron Deficiency Anemia (IDA) | Megaloblastic Anemia (B12/Folate) | Aplastic Anemia |
| MCV (Size) | Normocytic (can become Microcytic) | Microcytic | Macrocytic (> 100 fL) | Normocytic (rarely mildly macrocytic) |
| Reticulocyte Count | Low (Inadequate production) | Low | Low (Ineffective erythropoiesis) | Critically Low (Aplastic) |
| Serum Iron | Low | Low | Normal to High | Normal to High |
| Serum Ferritin | Normal to High | Low | Normal to High | Normal to High |
| TIBC | Low to Normal | High | Normal | Normal |
| Blood Film | Usually Normocytic | Hypochromic, Pencil cells | Hypersegmented Neutrophils, Ovalocytes | Normocytic, Normochromic |
| WBC & Platelets | Usually Normal (or High if acute inflammation) | Usually Normal (Platelets may be High) | May be Low (Pancytopenia in severe cases) | Low (Pancytopenia) |
| Bone Marrow | Increased iron in macrophages; low sideroblasts | Absent iron stores | Megaloblastic changes | Hypocellular (replaced by fat) |
Key Differentiators
- ACD vs. IDA: The primary "tell" is the ferritin and TIBC. In Anemia of Chronic Disease (Anemia of Inflammation), the "body safe" (ferritin) is full but locked, so TIBC is low. In IDA, the safe is empty, so the body increases TIBC to "grab" any available iron.
- ACD vs. B12/Folate Deficiency: While both are "underproduction" anemias, B12 and Folate deficiencies are characterized by nuclear-cytoplasmic asynchrony. This results in large, fragile cells (MCV > 100 fL) and the classic finding of hypersegmented neutrophils (> 5 lobes).
- ACD vs. Aplastic Anemia: Anemia of Chronic Disease (Anemia of Inflammation) is typically a problem of iron restricted to the erythroid line. Aplastic Anemia is a primary bone marrow failure. If you see a low hemoglobin accompanied by low white cell and platelet counts (pancytopenia) without a clear inflammatory cause, a bone marrow biopsy is mandatory to rule out aplasia.
Summary of Pathophysiological Mechanisms
- ACD: Cytokine-driven (IL-6) iron sequestration and EPO resistance.
- IDA: Absolute lack of systemic iron due to blood loss or poor intake.
- B12/Folate: Impaired DNA synthesis leading to megaloblastic maturation.
- Aplastic Anemia: Stem cell destruction (often T-cell mediated) leading to a "dry" marrow.
Additional Considerations
In some cases, a combination of anemia types might be present. For example, a patient with chronic inflammatory bowel disease (CIBD) might have Anemia of Chronic Disease (Anemia of Inflammation) along with iron deficiency due to blood loss. A thorough medical history, physical examination, and appropriate laboratory tests are essential for accurate diagnosis.
Treatment and Management
The management of Anemia of Chronic Disease (Anemia of Inflammation) is inherently complex because the anemia is a symptom of systemic inflammation rather than a primary bone marrow disorder. Effective treatment requires a balanced approach between addressing the underlying cause and managing iron-restricted erythropoiesis.
Primary Strategy: Treating the Underlying Condition
The most effective "cure" for Anemia of Chronic Disease (Anemia of Inflammation) is the resolution of the inflammatory state.
- Infection: Appropriate antibiotic, antiviral, or antifungal therapy.
- Autoimmune Disease: Use of corticosteroids, DMARDs, or biologics (e.g., TNF-alpha inhibitors) to lower the systemic cytokine load.
- Malignancy: Surgical resection, chemotherapy, or radiotherapy to reduce tumor-induced inflammation.
- Outcome: As the C-reactive protein (CRP) and IL-6 levels fall, hepcidin production decreases, allowing sequestered iron to be released from macrophages and normal erythropoiesis to resume.
Iron Supplementation (Oral vs. IV)
Iron therapy in Anemia of Chronic Disease (Anemia of Inflammation) is controversial because the "iron block" (hepcidin) prevents effective utilization.
- Oral Iron: Generally ineffective in active Anemia of Chronic Disease (Anemia of Inflammation). High hepcidin levels degrade ferroportin in the gut, leading to poor absorption and significant gastrointestinal side effects.
- Intravenous (IV) Iron: Preferred when iron therapy is indicated. IV iron bypasses the gut absorption barrier and "overwhelms" the macrophage sequestration, delivering iron directly to the bone marrow. Used when there is a mixed anemia (ACD + Absolute IDA) or a functional iron deficiency where ferritin is normal but transferrin saturation is < 20%.
Erythropoiesis-Stimulating Agents (ESAs)
ESAs (e.g., Epoetin alfa, Darbepoetin alfa) are used to overcome the EPO resistance and relative EPO deficiency seen in chronic inflammation.
- Clinical Use: Most common in Anemia of Chronic Kidney Disease (CKD) and chemotherapy-induced anemia.
- The "Iron Requirement": ESAs are significantly more effective when paired with IV iron. Without adequate available iron, ESAs will lead to "iron-restricted erythropoiesis."
- Risks: High doses of ESAs are associated with an increased risk of thromboembolic events and potential tumor progression in certain cancers. The goal is typically to maintain Hemoglobin (Hb) around 10 - 12 g/dL, rather than normalizing it.
Emerging & Novel Therapies
Recent understanding of the IL-6/hepcidin axis has led to new pharmacological targets:
- HIF-PH Inhibitors (e.g., Roxadustat): These oral agents stabilize Hypoxia-Inducible Factors, which naturally increase EPO production and, crucially, downregulate hepcidin. This makes them highly effective in inflammatory states where traditional EPO might fail.
- Anti-IL-6 Therapy (e.g., Tocilizumab): By blocking the IL-6 receptor, these drugs stop the primary signal for hepcidin production at the source.
- Hepcidin Antagonists: Direct hepcidin ferroportin-stabilizers and hepcidin-neutralizing antibodies are currently in various stages of clinical trials.
Red Blood Cell (RBC) Transfusions
Transfusions are reserved for severe, symptomatic anemia (Hb < 7 - 8 g/dL) or patients with underlying cardiovascular compromise.
- Pros: Immediate symptomatic relief.
- Cons: Risk of iron overload (especially since Anemia of Chronic Disease (Anemia of Inflammation) patients already have high iron stores), transfusion reactions, and alloimmunization. It does not treat the underlying inflammatory pathophysiology.
Management Summary Table
| Intervention | Goal | Best For... |
| Treat Underlying Cause | Eliminate the inflammatory stimulus. | First-line for all patients. |
| IV Iron | Bypass hepcidin block in the gut. | Mixed ACD/IDA or functional deficiency. |
| ESAs | Overcome marrow resistance/low EPO. | CKD, Malignancy, or severe ACD. |
| HIF-PH Inhibitors | Stimulate EPO and lower hepcidin. | CKD (emerging as a major oral alternative). |
| RBC Transfusion | Rapid Hb increase. | Acute symptoms or hemodynamic instability. |
Always check the sTfR/log Ferritin Index before starting iron.
If the index is low (< 1), the patient has pure ACD, and giving iron (especially oral) is unlikely to help and may cause harm through oxidative stress. Focus on the inflammation first!
Frequently Asked Questions (FAQs)
What are the diagnostic criteria for Anemia of Chronic Disease (Anemia of Inflammation)?
There aren't strictly defined diagnostic criteria for Anemia of Chronic Disease (Anemia of Inflammation) because there's no single test that definitively diagnoses it. Diagnosis relies on a combination of factors:
Clinical Features: Presence of a chronic inflammatory condition (infection, autoimmune disease, etc.) lasting for at least 3 months.
Laboratory Findings
- Anemia: Microcytic (small red blood cells) or normocytic (normal-sized red blood cells) anemia based on a complete blood count (CBC).
- Iron Paradox:
- Low serum iron levels.
- Normal or elevated ferritin levels (indicating iron stores but limited availability due to hepcidin).
- Low Transferrin Saturation: Reflects low percentage of transferrin carrying iron.
- Low Reticulocyte Count: Suggests decreased red blood cell production in the bone marrow.
Additional Considerations
- While not routinely used for diagnosis, elevated inflammatory markers (C-reactive protein or erythrocyte sedimentation rate) might be present due to the underlying chronic condition.
- In some complex cases, a bone marrow examination might be necessary to rule out other causes of anemia.
How long does it take to recover from Anemia of Chronic Disease (Anemia of Inflammation)?
Unfortunately, there's no one-size-fits-all answer to how long it takes to recover from Anemia of Chronic Disease (Anemia of Inflammation) because the recovery timeline depends on several factors:
- Severity of the Underlying Condition: Effectively managing the chronic inflammatory disease that's causing Anemia of Chronic Disease (Anemia of Inflammation) is essential for improvement. The speed of recovery hinges on controlling the underlying condition and reducing inflammation.
- Degree of Anemia: The severity of the anemia itself also influences recovery time. More severe anemia might take longer to improve compared to mild cases.
- Treatment Approach: The specific treatment approach used, such as addressing nutritional deficiencies or using medications like ESAs (in specific cases), can impact the recovery rate.
- Individual Factors: Individual factors like overall health status and response to treatment can also influence how quickly red blood cell counts normalize.
Here's a general timeframe to consider
- With effective treatment of the underlying condition: In some cases, with successful management of the chronic inflammatory condition, noticeable improvement in anemia can occur within weeks to months.
- More complex cases: In situations with severe underlying conditions or where controlling inflammation is challenging, recovery might take several months or even longer.
Why is TIBC low in Anemia of Chronic Disease (Anemia of Inflammation)?
In Anemia of Chronic Disease (Anemia of Inflammation), the total iron-binding capacity (TIBC) is typically low even though there might be adequate or even elevated iron stores in the body.
Understanding Transferrin and TIBC
- Transferrin: This is a protein in the blood that plays a critical role in iron transport. It acts like a carrier, binding to iron and shuttling it throughout the body for various functions, including red blood cell production.
- TIBC: Total Iron-Binding Capacity is an indirect measurement of transferrin levels in the blood. The higher the TIBC, the more iron transferrin can potentially bind to.
The Role of Hepcidin in ACD
Chronic inflammation triggers the production of hepcidin, a regulatory protein produced by the liver. Hepcidin then binds to ferroportin, a protein on the surface of macrophages and intestinal enterocytes. These cells are responsible for iron release from stores and absorption from the diet, respectively. By binding to ferroportin, hepcidin essentially locks up iron within these cells, hindering its release into circulation even though iron stores might be normal or elevated.
How Hepcidin Affects TIBC
Since iron is trapped within cells due to hepcidin, less iron is available to bind to transferrin in the bloodstream. As a consequence, a significant portion of transferrin remains unbound. This translates to a lower TIBC because the total capacity for iron binding (transferrin) is not being fully utilized.
Low TIBC in Anemia of Chronic Disease (Anemia of Inflammation) reflects limited iron availability due to hepcidin, not a true lack of iron stores. It contrasts with iron deficiency anemia, where TIBC is high because there's insufficient iron to bind to all the available transferrin.
Imagine transferrin as taxis and iron as passengers. In a normal situation (without ACD), most taxis (transferrin) are carrying passengers (iron). TIBC would be high, reflecting a full capacity. In Anemia of Chronic Disease (Anemia of Inflammation), due to hepcidin, iron is stuck at the "depots" (macrophages and enterocytes). Even though there are plenty of taxis, most are empty, leading to a lower TIBC.
Why is ferritin high in Anemia of Chronic Disease (Anemia of Inflammation)?
In Anemia of Chronic Disease (Anemia of Inflammation), ferritin levels are often normal or elevated even though there's limited iron available for red blood cell production.
Ferritin is a protein found in cells throughout the body, particularly in the liver, spleen, and bone marrow. It acts as a storage unit for iron, binding to excess iron and keeping it safe for future use. A high ferritin level generally indicates sufficient or even overloaded iron stores in the body.
High ferritin in Anemia of Chronic Disease (Anemia of Inflammation) indicates iron stores are present, but they are not available for erythropoiesis (red blood cell production) due to hepcidin. This is a key difference from iron deficiency anemia (IDA) where ferritin levels are typically low because there's a genuine lack of iron stores in the body.
Imagine ferritin as a safe deposit box for iron. In a healthy situation, the safe deposit box (ferritin) has some iron stored. In ACD, the safe deposit box (ferritin) is full of iron, but it's locked shut (due to hepcidin) and inaccessible for red blood cell production. This leads to high ferritin but limited usable iron.
Can osteoarthritis cause Anemia of Chronic Disease (Anemia of Inflammation)?
Osteoarthritis is a degenerative joint disease primarily characterized by wear and tear of cartilage in the joints. While some low-grade inflammation might be present in OA, it's different from the systemic and persistent inflammation seen in conditions that cause Anemia of Chronic Disease (Anemia of Inflammation).
Can diabetes cause Anemia of Chronic Disease (Anemia of Inflammation)?
Diabetes itself does not directly cause Anemia of Chronic Disease (Anemia of Inflammation), but there can be a complex interplay between the two conditions. Chronic complications of diabetes, such as nephropathy (kidney disease) or neuropathy (nerve damage), can sometimes trigger secondary inflammatory processes. This could contribute to Anemia of Chronic Disease (Anemia of Inflammation) in some cases.
Can hypertension cause Anemia of Chronic Disease (Anemia of Inflammation)?
No, hypertension (high blood pressure) is not a direct cause of Anemia of Chronic Disease (Anemia of Inflammation). Anemia of Chronic Disease (Anemia of Inflammation) arises from chronic inflammatory conditions, whereas high blood pressure is a cardiovascular condition with distinct causes. However, there can be some indirect connections and considerations.
Who treats Anemia of Chronic Disease (Anemia of Inflammation)?
A patient with Anemia of Chronic Disease (Anemia of Inflammation) will typically be managed by a collaborative team of healthcare professionals including the primary care physician, the hematologist and the specialist for the underlying chronic condition.
How do you differentiate Anemia of Chronic Disease from Iron Deficiency Anemia?
While both present with low serum iron, Anemia of Chronic Disease (Anemia of Inflammation) is characterized by normal-to-elevated ferritin, a low Total Iron-Binding Capacity (TIBC), and normal sTfR. In contrast, IDA features low ferritin, a high TIBC, and an elevated sTfR. The sTfR/log ferritin index is the most reliable calculation for distinguishing the two.
Why is oral iron often ineffective in Anemia of Inflammation?
Chronic inflammation triggers the liver to produce high levels of hepcidin. Hepcidin binds to and degrades ferroportin in the intestinal lining. Without ferroportin, the dietary or supplemental iron absorbed by the gut cannot be transferred into the bloodstream, making oral iron largely ineffective.
When is intravenous (IV) iron indicated for Anemia of Chronic Disease?
IV iron is considered when patients have a concurrent absolute or functional iron deficiency, especially in settings like chronic kidney disease (CKD), inflammatory bowel disease (IBD), or chronic heart failure, where oral iron absorption is blocked by hepcidin or poorly tolerated.
What are HIF-PH inhibitors and how do they treat anemia?
HIF-PH inhibitors (like Roxadustat) are a newer class of oral medications used primarily for anemia of chronic kidney disease. They stabilize Hypoxia-Inducible Factors, which stimulates the body's natural production of EPO and helps lower hepcidin levels, improving overall iron utilization.
Glossary of Related Medical Terms
- Erythropoiesis-Stimulating Agents (ESAs): Synthetic hormones (like epoetin alfa) used to stimulate the bone marrow to produce more red blood cells.
- Ferroportin: A transmembrane protein responsible for transporting iron out of cells (such as enterocytes and macrophages) and into the bloodstream.
- Functional Iron Deficiency: A state where total body iron stores are normal or high, but the iron is locked within macrophages and unavailable for red blood cell production (erythropoiesis).
- Hepcidin: A peptide hormone produced by the liver that serves as the master regulator of iron homeostasis; it is upregulated by inflammation (IL-6) and inhibits iron transport by degrading ferroportin.
- Hypoxia-Inducible Factor (HIF): A transcription factor that responds to cellular oxygen levels, playing a key role in stimulating endogenous erythropoietin production.
- Soluble Transferrin Receptor (sTfR): A truncated fragment of the membrane transferrin receptor; elevated levels in the serum indicate cellular iron deficiency.
References
- Madu AJ, Ughasoro MD. Anaemia of Chronic Disease: An In-Depth Review. Med Princ Pract. 2017;26(1):1-9. doi: 10.1159/000452104. Epub 2016 Sep 28. PMID: 27756061; PMCID: PMC5588399.
- GBD 2021 Anaemia Collaborators. Prevalence, years lived with disability, and trends in anaemia burden by severity and cause, 1990-2021: findings from the Global Burden of Disease Study 2021. Lancet Haematol. 2023 Sep;10(9):e713-e734. doi: 10.1016/S2352-3026(23)00160-6. Epub 2023 Jul 31. Erratum in: Lancet Haematol. 2023 Oct;10(10):e796. Erratum in: Lancet Haematol. 2024 Jan;11(1):e10. PMID: 37536353; PMCID: PMC10465717.
- Svenson N, Bailey J, Durairaj S, Dempsey-Hibbert N. A simplified diagnostic pathway for the differential diagnosis of iron deficiency anaemia and anaemia of chronic disease. International Journal of Laboratory Hematology. 2021;43:1644–1652.
- Marques, O., Weiss, G., & Muckenthaler, M. U. (2022). The role of iron in chronic inflammatory diseases: from mechanisms to treatment options in anemia of inflammation. Blood, 140(19), 2011–2023. https://doi.org/10.1182/blood.2021013472
- Wiciński, M., Liczner, G., Cadelski, K., Kołnierzak, T., Nowaczewska, M., & Malinowski, B. (2020). Anemia of Chronic Diseases: Wider Diagnostics-Better Treatment?. Nutrients, 12(6), 1784. https://doi.org/10.3390/nu12061784
- Tussing-Humphreys, L., Pusatcioglu, C., Nemeth, E., & Braunschweig, C. (2012). Rethinking iron regulation and assessment in iron deficiency, anemia of chronic disease, and obesity: introducing hepcidin. Journal of the Academy of Nutrition and Dietetics, 112(3), 391–400. https://doi.org/10.1016/j.jada.2011.08.038
- Li, Z., Shen, L., Tu, Y., Lu, S., & Liu, B. (2025). Hypoxia-inducible factor-prolyl hydroxylase inhibitors in treatment of anemia with chronic disease. Chinese medical journal, 138(12), 1424–1432. https://doi.org/10.1097/CM9.0000000000003470
- Sun, C. C., Vaja, V., Babitt, J. L., & Lin, H. Y. (2012). Targeting the hepcidin-ferroportin axis to develop new treatment strategies for anemia of chronic disease and anemia of inflammation. American journal of hematology, 87(4), 392–400. https://doi.org/10.1002/ajh.23110
- Pasini, E., Corsetti, G., Romano, C., Aquilani, R., Scarabelli, T., Chen-Scarabelli, C., & Dioguardi, F. S. (2021). Management of Anaemia of Chronic Disease: Beyond Iron-Only Supplementation. Nutrients, 13(1), 237. https://doi.org/10.3390/nu13010237