Key Takeaways
Megaloblastic anemia is caused by faulty DNA synthesis in red cell precursors, almost always due to vitamin B12 (cobalamin) or folate (vitamin B9) deficiency, producing oversized red cells (macrocytes) with a mean corpuscular volume typically above 100 fL.
Signs and symptoms ▾: Generally, megaloblastic anemia presents with symptoms of anemia. Megaloblastic anemia from B12 deficiency can cause irreversible neurological damage, including subacute combined degeneration of the spinal cord; folate deficiency does not cause these neurological signs.
Causes ▾: The most common cause of B12 deficiency in adults is autoimmune gastritis (pernicious anemia), in which immune destruction of stomach parietal cells eliminates intrinsic factor and blocks B12 absorption. In folate deficiency, poor intake of leafy greens, malabsorption and pregnancy are common causes.
Laboratory investigations & Diagnosis ▾: Diagnosis relies on a complete blood count and peripheral smear revealing macrocytosis (MCV >100 fL) and hypersegmented neutrophils, followed by measuring serum folate alongside total or active B12 levels. If initial results are borderline, metabolite testing (methylmalonic acid and homocysteine) and targeted antibody screens can definitively distinguish between B12 and folate deficiencies while identifying the underlying cause.
Treatment and management of megaloblastic anemia ▾: Standard treatment for B12 deficiency is hydroxocobalamin 1 mg intramuscularly with the dosing schedule depending on whether neurological symptoms are present, while folate deficiency is treated with oral folic acid 5 mg daily.
*Click ▾ for more information
What is Megaloblastic Anemia?
Megaloblastic anemia is a type of anemia caused by faulty DNA synthesis in the bone marrow. The cells that should grow up to become red blood cells cannot divide properly, so they end up oversized, immature, and short-lived. The trigger is almost always a deficiency of vitamin B12 (cobalamin) or folate (vitamin B9) [1,3].
This disorder is common, especially in older adults, vegans, pregnant women, and people with gut disorders. Recognizing it early matters because untreated B12 deficiency can cause permanent nerve damage [2,5].
This guide walks through what megaloblastic anemia is, why B12 and folate matter, how the deficiency causes the disease, and how clinicians diagnose and treat it.
Why Vitamin B12 and Folate Matter
Every dividing cell in your body needs DNA. Both B12 and folate are essential cofactors for building the building blocks of DNA. When either runs short, cell division stalls — red cell precursors keep growing in size because cytoplasm production continues, but they cannot divide because DNA synthesis is stuck. [1,3].
Many die in the marrow before they ever reach the bloodstream, a process called ineffective erythropoiesis (failed red cell production). Those that do escape are unusually large macrocytes. White cell and platelet production suffer too, which is why severe cases show low counts across all three blood lines [3,5].
Where We Get B12 and Folate
Vitamin B12 comes from animal foods: meat, fish, eggs, and dairy. Plants do not make B12, so vegans rely on fortified foods or supplements.
Folate is found in leafy greens, legumes, citrus fruits, liver, and fortified cereals and flour. Heat destroys folate, so heavily cooked food loses much of its content [1].
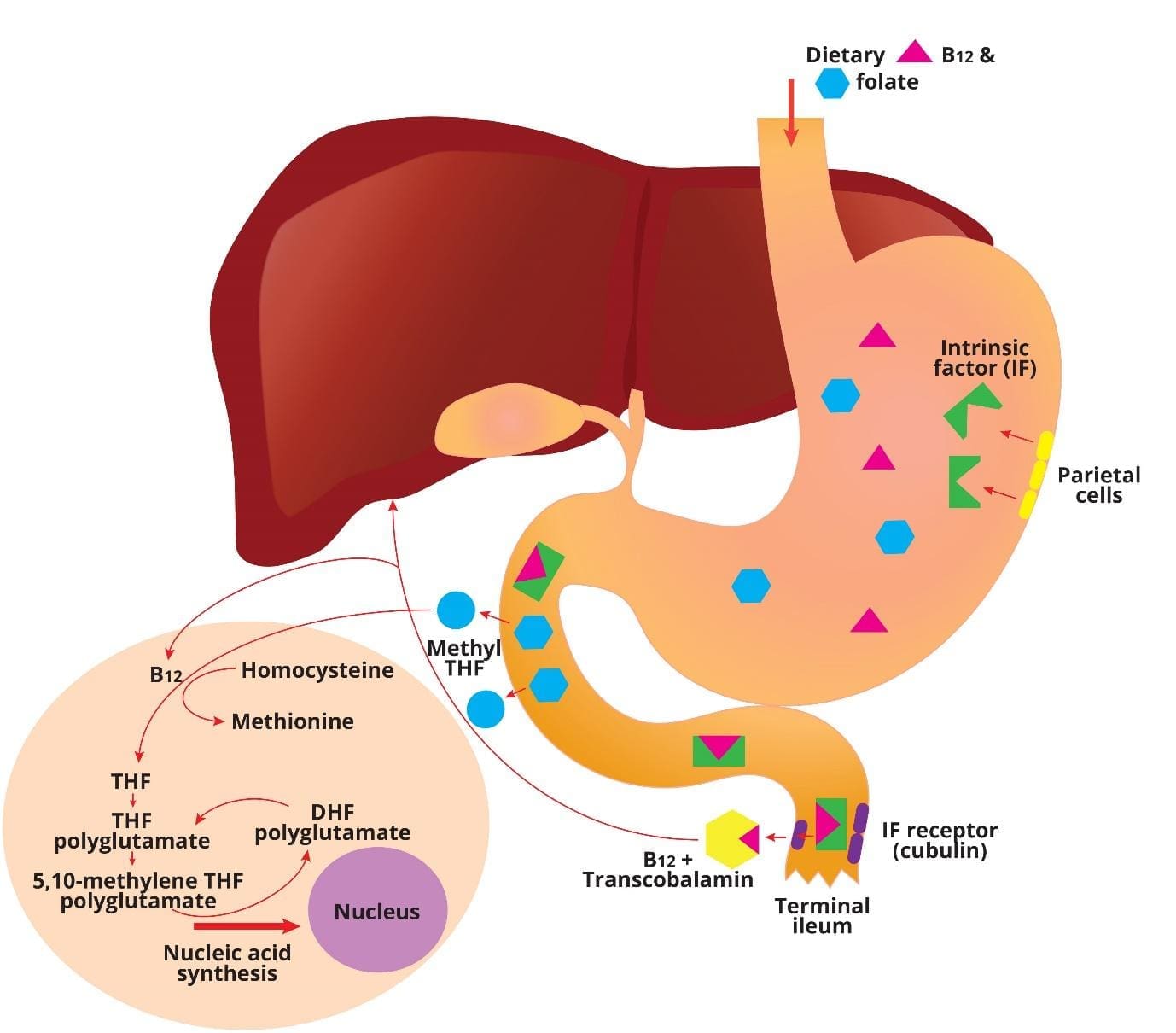
How the Body Absorbs B12 and Folate
Vitamin B12 Absorption
Vitamin B12 takes a long route through the gut before it ever enters the bloodstream. Each handover matters, and any of them can break.
In the mouth, B12 binds to a salivary protein called haptocorrin. In the stomach, acid releases B12 from food, and it stays bound to haptocorrin. Stomach cells (parietal cells) secrete intrinsic factor, a protein that will be needed downstream. In the duodenum, pancreatic enzymes free B12 from haptocorrin so it can bind to intrinsic factor instead. The B12–intrinsic factor complex then travels to the terminal ileum, where receptors called cubilin pull it into the gut wall. Finally, B12 enters the blood bound to transcobalamin, which delivers it to tissues [1,5].
If any link breaks — low stomach acid, missing intrinsic factor, ileal disease — absorption fails.
Folate absorption
Folate is simpler. Dietary folates are converted to monoglutamate forms by gut enzymes, then absorbed mainly in the upper small intestine (jejunum) by a transporter called the proton-coupled folate transporter (PCFT). Once inside cells, folate is converted into its active forms and used for DNA synthesis [1,3].
How B12 and Folate Work Together
Inside the cell, B12 and folate are linked in one critical reaction. B12 is needed to convert methyl-tetrahydrofolate (methyl-THF) into the form of folate that builds DNA. Without B12, folate gets trapped as methyl-THF and cannot do its job — the so-called "methyl trap." This is why B12 deficiency and folate deficiency both produce the same red cell picture: the final pathway is the same blocked DNA synthesis [3,5].
Causes of Megaloblastic Anemia
Megaloblastic anemia comes down to two questions: is B12 low, or is folate low? Each has its own list of causes.
Causes of B12 Deficiency
Malabsorption (the most common cause)
- Autoimmune gastritis (pernicious anemia). The immune system attacks parietal cells, cutting off intrinsic factor. Most patients have antibodies to parietal cells, and about half have antibodies to intrinsic factor. The 2024 NICE guideline suggested using "autoimmune gastritis" in preference to the older term "pernicious anemia." Current consensus emphasizes that they are distinct stages of a disease continuum rather than strictly interchangeable terms. Autoimmune gastritis is the underlying root pathology, while pernicious anemia is the late-stage clinical manifestation resulting from severe intrinsic factor deficiency [2].
- Gastric surgery. Gastrectomy or bariatric surgery (gastric bypass, sleeve gastrectomy) removes or bypasses the stomach cells that make intrinsic factor [1,2].
- Crohn's disease, celiac disease, or ileal resection. These damage the terminal ileum where B12 is absorbed.
- Atrophic gastritis. Common in older adults. Reduced stomach acid means B12 stays stuck to food proteins and is never released [1,2].
- Small intestinal bacterial overgrowth (SIBO). Bacteria steal B12 before the gut can absorb it.
Dietary deficiency
The liver stores enough B12 for several years, so dietary deficiency takes time to appear. It eventually shows up in strict vegans who do not use fortified foods or supplements, in chronic alcohol use, and in severe malnutrition [1].
Drugs and other factors
- Proton pump inhibitors (PPIs) and H2 blockers reduce stomach acid and B12 release.
- Metformin interferes with B12 absorption in the ileum [2].
- Recreational nitrous oxide ("laughing gas," whippits) inactivates B12 inside the body, causing functional deficiency even when blood levels look normal. This is increasingly seen in younger adults [2].
- Aging reduces stomach acid and increases risk gradually.

Causes of Folate Deficiency
Folate stores last only a few months, so deficiency develops faster than with B12.
Inadequate intake
- Poor diet, especially low intake of leafy greens, legumes, and fortified grains.
- Chronic alcohol use, which both lowers intake and impairs absorption.
- Overcooking, which destroys folate [1].
Malabsorption
- Celiac disease, Crohn's disease, and tropical sprue.
- Bariatric surgery.
- Rare genetic transporter defects.
Increased need
- Pregnancy and breastfeeding.
- Chronic hemolytic anemia (such as sickle cell disease) where the marrow runs at full speed.
- Skin conditions with high cell turnover, such as severe psoriasis.
- Some myeloproliferative disorders.
Drugs and renal loss
- Methotrexate (a folate antagonist used for cancer and autoimmune disease).
- Some anticonvulsants and sulfasalazine.
- Hemodialysis, which removes folate from the blood [1].

Megaloblastic Anemia Symptoms and Signs
Symptoms develop slowly, often over months. The clinical picture has three layers: general anemia symptoms, mucosal and digestive signs shared by both deficiencies, and neurological signs that are specific to B12 deficiency.
General Anemia Symptoms (Both Deficiencies)
- Fatigue and weakness, usually the first complaint.
- Shortness of breath on exertion.
- Pale skin, sometimes with a mild yellow tint from low-grade hemolysis of fragile macrocytes.
- Fast heartbeat or palpitations.
- Headaches and dizziness [3].
Digestive Symptoms (Both Deficiencies)
- Glossitis — a red, sore, smooth tongue. A classic finding.
- Mouth ulcers and angular stomatitis (cracks at the corners of the mouth).
- Loss of appetite, sometimes with weight loss.
- Diarrhea or nausea [3].

Neurological Symptoms (B12 Deficiency only)
This is the critical distinguishing feature. Folate deficiency does not cause these problems, and giving folate alone can correct the anemia while nerve damage silently progresses [1,5].
- Paresthesia — pins and needles in the hands and feet, often symmetrical.
- Loss of balance, unsteady gait, clumsiness. These point toward subacute combined degeneration (SACD) of the spinal cord, where the dorsal columns and corticospinal tracts demyelinate [5].
- Cognitive changes: confusion, poor memory, difficulty concentrating.
- Mood changes: irritability, depression, and rarely psychosis or dementia-like symptoms.
- Visual disturbance.
The 2024 NICE guideline emphasizes that B12 deficiency can present with neurological or psychiatric symptoms even in the absence of anemia or macrocytosis. Do not rule it out just because the blood count looks normal.
How is Megaloblastic Anemia Diagnosed?
Diagnosis of megaloblastic anemia moves from broad to specific: confirm anemia, confirm it is macrocytic, confirm which vitamin is low, and find the underlying cause.
Clinical Testing Threshold
Current clinical guidelines caution against indiscriminate B12 testing. General fatigue or tiredness alone is no longer considered a valid clinical indication for a B12 assay unless it is accompanied by an abnormal complete blood count (such as unexplained macrocytosis) or clear neurological signs.
Initial Blood Tests
Complete Blood Count (CBC)
This test is the cornerstone of diagnosis. It will typically show:
- Low hemoglobin confirms anemia.
- High mean corpuscular volume (MCV), conventionally over 100 fL, identifies macrocytosis. Some references use a more sensitive cutoff of 95 fL, which catches earlier disease at the cost of more false positives [1,4].
- Pancytopenia — low red cells, white cells, and platelets — appears in severe cases.
- Reticulocyte count is low, reflecting failed red cell production.
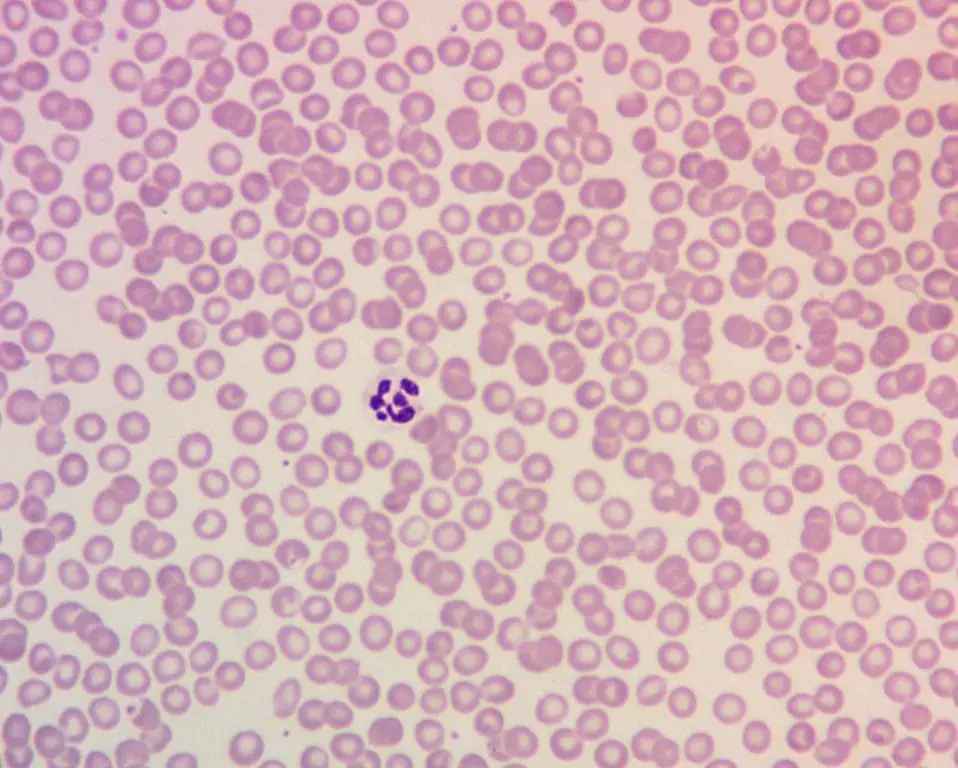
Peripheral Blood Smear
- Macro-ovalocytes — large, oval-shaped red blood cells.
- Hypersegmented neutrophils with five or more nuclear lobes. Sensitive and reasonably specific for megaloblastic anemia.
- Anisopoikilocytosis (variation in size and shape).
Vitamin Level Testing
- Serum vitamin B12 below roughly 200 pg/mL suggests deficiency. Borderline values (200–400 pg/mL) need follow-up.
- Serum folate below roughly 4 ng/mL suggests deficiency. The 2014 BSH guideline considers serum folate equivalent to red cell folate as the first-line test [1].
- Active B12 (holotranscobalamin, holoTC) measures the fraction of B12 cells can actually use. NICE NG239 recognizes it as a first-line option alongside total B12 [2].
Confirmatory Tests
When vitamin levels are borderline, metabolite testing helps:
- Methylmalonic acid (MMA) rises in B12 deficiency and stays normal in folate deficiency.
- Homocysteine (HCY) rises in both deficiencies.
Summary
Elevated MMA + elevated HCY = B12 deficiency
Normal MMA + elevated HCY = folate deficiency
MMA also rises in kidney disease, which can confuse the picture [1,4].
Investigations for the Underlying Cause
Once B12 deficiency is confirmed, find out why:
- Intrinsic factor antibodies — highly specific for autoimmune gastritis (pernicious anemia), but only positive in roughly half of cases.
- Parietal cell antibodies — while traditionally included in the workup, recent diagnostic pathways explicitly advise against routine testing for gastric parietal cell antibodies due to their exceedingly low specificity. Diagnostic workups for malabsorption should focus primarily on intrinsic factor antibodies [9].
- Endoscopy and biopsy — used when autoimmune gastritis is suspected, partly to assess gastric cancer risk on long-term follow-up [2].
For folate deficiency, the workup focuses on diet, alcohol intake, malabsorption screens (celiac serology), and drug history.
How Is Megaloblastic Anemia Treated?
Treatment has three parts: replace the missing vitamin, fix the underlying cause where possible, and monitor the response.
Treatment for Vitamin B12 Deficiency
Standard practice in the UK and many other countries follows the BSH guideline regimen: hydroxocobalamin 1 mg intramuscularly (IM) [1]. A reticulocyte rise is expected within 7–10 days. No response means the diagnosis should be reviewed [1].
- Without neurological involvement: 1 mg IM three times a week for 2 weeks, then maintenance every 3 months.
- With neurological involvement: 1 mg IM on alternate days until no further improvement, then maintenance every 2 months.
The strict "every 2–3 months" maintenance schedule lacks a robust evidence base. Up to 40% of patients with true pernicious anemia report requiring more frequent injections to achieve adequate symptom relief; therefore, maintenance therapy intervals must be highly individualized based on the patient's clinical response [10,11].
High-dose oral cyanocobalamin (typically 1,000–2,000 mcg daily) is highly effective for dietary deficiencies and is recognized by NICE NG239 as an option for selected patients. However, intramuscular (IM) injections remain the definitive gold standard for malabsorptive conditions (like pernicious anemia), particularly to ensure rapid absorption and adequate tissue retention if any neurological symptoms are present. [1,2].
Watch for hypokalemia in the first days of treating severe deficiency. Newly produced cells take up potassium rapidly, which can drop serum levels.
Lifelong therapy is required when the cause is not reversible — autoimmune gastritis, total gastrectomy, complete ileal resection.
Treatment for Folate Deficiency
- Oral folic acid 5 mg daily for several months until anemia resolves and stores are replenished [1].
- Address the cause: improve diet, treat celiac or Crohn's disease, review drugs.
- Pregnant women receive 400 mcg folic acid daily preconception and through the first trimester, with higher doses (5 mg) for those at increased risk of neural tube defects.
The Critical Rule
Never give folate alone to a patient with megaloblastic anemia until B12 deficiency has been ruled out. Folate will lift the hemoglobin and may make the patient feel better, but B12 nerve damage will keep progressing — sometimes irreversibly [1,5,6].
Monitoring
- Repeat CBC, B12, and folate levels to confirm response.
- Reticulocyte rise within 7–10 days.
- Long-term follow-up for autoimmune gastritis includes monitoring for gastric cancer, as recommended by NICE NG239 [2].
Public Health Note: Folate Fortification
Many countries — including the United States since 1998 and the United Kingdom since 2024 — now mandate folic acid fortification of flour or grain products. The goal is to prevent neural tube defects, but it has also reduced the prevalence of folate-deficiency megaloblastic anemia at population level [8].
Frequently Asked Questions (FAQs)
What causes megaloblastic anemia?
Megaloblastic anemia is caused by faulty DNA synthesis in developing red blood cells, almost always due to a deficiency of vitamin B12 (cobalamin) or folate (vitamin B9). Less common causes include inherited disorders of B12 metabolism, certain chemotherapy and immunosuppressant drugs (such as methotrexate), and recreational nitrous oxide use [1,2,7].
What is the difference between megaloblastic anemia and non-megaloblastic anemia?
Both megaloblastic and non-megaloblastic anemia are forms of macrocytic anemia, meaning they are characterized by abnormally large red blood cells (macrocytes). This is identified by a Mean Corpuscular Volume (MCV) greater than 100 fL. The key difference lies in the underlying cause and the specific appearance of the cells on a blood smear.
Why is vitamin B12 deficiency more dangerous than folate deficiency?
B12 deficiency can damage the spinal cord and peripheral nerves, causing numbness, balance problems, and cognitive changes. This neurological damage may not fully reverse if treatment is delayed. Folate alone can correct the anemia but does not protect the nervous system, so giving folate without ruling out B12 deficiency can mask the problem while nerve damage worsens [1,5,6].
What is the difference between the terms megaloblastic and macrocytic?
Megaloblastic refers to the type of cell development, while macrocytic refers to the size of the red blood cells. Megaloblastic anemia is a specific type of macrocytic anemia caused by abnormal precursor cells. Not all macrocytic anemias are megaloblastic, and having macrocytic red blood cells requires further investigation for diagnosis.
How does vitamin B12 deficiency cause macrocytic anemia?
When your body lacks vitamin B12, it struggles to properly utilize B9 (folate) for DNA synthesis in developing red blood cells. This disrupts their maturation process, leading to the formation of abnormally large but dysfunctional cells called megaloblasts. These giants can't properly divide or leave the bone marrow, resulting in fewer, oversized red blood cells in circulation – classic features of macrocytic anemia. This deficiency impacts oxygen delivery throughout your body, causing fatigue, weakness, and other symptoms.
Can megaloblastic anemia be treated with tablets, or are injections always needed?
It depends on the cause. Where absorption is intact (for example, dietary deficiency in vegans), oral B12 supplements work well. For absorption problems in megaloblastic anemia such as autoimmune gastritis (pernicious anemia), high-dose oral cyanocobalamin can also be effective in selected patients, but intramuscular hydroxocobalamin remains the standard, especially when there are neurological symptoms [1,2].
Is megaloblastic anemia preventable?
Many megaloblasctic anemia cases are preventable. Eating B12 from animal products or fortified foods, getting enough folate from leafy greens, legumes, and fortified grains, limiting alcohol, and taking supplements when at higher risk (such as during pregnancy or on a vegan diet) all help. National folic acid fortification programs have markedly reduced folate-deficiency anemia and neural tube defects worldwide [8].
What happens if megaloblastic anemia is left untreated?
Untreated megaloblastic anemia can lead to severe fatigue, heart strain, neurological damage (with B12 deficiency), pregnancy complications including neural tube defects in the baby, and rarely pancytopenia. Most cases respond well once the underlying deficiency is corrected, but long-standing nerve damage may not fully reverse [1,3,5].
What are the specific risks and considerations for pregnant women with megaloblastic anemia?
Pregnant women with megaloblastic anemia face unique risks and considerations compared to the general population.
Risks to the mother
- Increased risk of complications: Untreated megaloblastic anemia can lead to
- Preeclampsia: A serious pregnancy complication with high blood pressure and potential organ damage.
- Preterm birth: Delivering the baby before 37 weeks.
- Low birth weight: The baby being smaller than expected for gestational age.
- Postpartum hemorrhage: Excessive bleeding after childbirth.
- Neurological damage: Vitamin B12 deficiency, a major cause, can cause nerve damage if left untreated. This can lead to weakness, numbness, and cognitive problems.
Risks to the baby
- Neural tube defects: Folate deficiency, another cause, significantly increases the risk of these birth defects affecting the brain and spinal cord.
- Growth problems: The baby may not grow and develop as expected due to nutrient deficiencies.
- Increased risk of miscarriage: Studies suggest a potential link between untreated anemia and miscarriage, although more research is needed.
Considerations for management
- Early diagnosis and treatment are crucial: Prompt intervention minimizes risks to both mother and baby.
- Identifying the underlying cause: Addressing the root cause of the anemia, like B12 deficiency or dietary limitations, is essential.
- Prenatal vitamins: Folic acid supplementation is standard for all pregnant women, but those with anemia may need additional vitamin B12 supplements.
- Regular monitoring: Close monitoring of blood counts, vitamin levels, and fetal development is critical.
- Multidisciplinary care: Collaboration between obstetricians, hematologists, and nutritionists is often recommended for optimal management.
Are there any genetic factors that contribute to the development of megaloblastic anemia?
Inborn errors of metabolism: These are genetic mutations affecting enzymes involved in vitamin B12 or folate metabolism. This can lead to impaired absorption, utilization, or transport of these vitamins, ultimately causing megaloblastic anemia. Examples include:
- Megaloblastic anemia type I (cblC/cblD deficiency): Affects the conversion of vitamin B12 to its active form.
- Megaloblastic anemia type II (cblB deficiency): Affects the transport of vitamin B12 inside cells.
- Congenital folate malabsorption: Impairs the absorption of folate from the gut.
Fanconi anemia: This genetic disorder affects DNA repair mechanisms and can lead to bone marrow problems, including megaloblastic anemia initially before presenting with aplastic anemia.
Glossary of Related Medical Terms
- Anemia — Low hemoglobin in the blood, which reduces how much oxygen the blood can carry.
- Cobalamin — The chemical name for vitamin B12.
- Erythropoiesis — The process of making red blood cells in the bone marrow.
- Folate (vitamin B9) — A vitamin needed to build DNA; found in leafy greens, legumes, and fortified cereals.
- Folic acid — The synthetic, supplement form of folate.
- Glossitis — A red, sore, swollen tongue, often smooth in appearance.
- Holotranscobalamin (holoTC, "active B12") — The fraction of B12 available for cells to use; sometimes measured to assess B12 status.
- Hypersegmented neutrophil — A white blood cell whose nucleus has five or more lobes; a classic clue to megaloblastic anemia.
- Intrinsic factor — A protein made by stomach cells that allows vitamin B12 to be absorbed in the small intestine.
- Macrocytic — Describes red blood cells that are larger than normal.
- Megaloblast — An abnormally large, immature red cell precursor seen in the bone marrow.
- Mean corpuscular volume (MCV) — The average size of a red blood cell, reported on a complete blood count.
- Methylmalonic acid (MMA) — A substance that builds up in the blood when B12 is deficient.
- Pernicious anemia / autoimmune gastritis — An autoimmune attack on stomach cells that cuts off intrinsic factor and causes B12 deficiency.
- Reticulocyte — A young red blood cell; counts rise when the bone marrow responds to treatment.
- Subacute combined degeneration (SACD) — Damage to the spinal cord caused by long-standing B12 deficiency, leading to numbness, weakness, and balance problems.
- Thrombocytopenia — Low platelet count.
Disclaimer: This article is intended for informational purposes only and is specifically targeted towards medical students. It is not intended to be a substitute for informed professional medical advice, diagnosis, or treatment. While the information presented here is derived from credible medical sources and is believed to be accurate and up-to-date, it is not guaranteed to be complete or error-free. See additional information.
References
- Devalia, V., Hamilton, M. S., Molloy, A. M., & British Committee for Standards in Haematology (2014). Guidelines for the diagnosis and treatment of cobalamin and folate disorders. British journal of haematology, 166(4), 496–513. https://doi.org/10.1111/bjh.12959.
- National Institute for Health and Care Excellence. (2024). Vitamin B12 deficiency in over 16s: diagnosis and management (NICE guideline NG239).
- Hariz A, Bhattacharya PT. Megaloblastic Anemia(Archived) [Updated 2023 Apr 3]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2026 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK537254/
- Aslinia, F., Mazza, J. J., & Yale, S. H. (2006). Megaloblastic anemia and other causes of macrocytosis. Clinical medicine & research, 4(3), 236–241. https://doi.org/10.3121/cmr.4.3.236
- Green, R., Allen, L. H., Bjørke-Monsen, A. L., Brito, A., Guéant, J. L., Miller, J. W., Molloy, A. M., Nexo, E., Stabler, S., Toh, B. H., Ueland, P. M., & Yajnik, C. (2017). Vitamin B12 deficiency. Nature reviews. Disease primers, 3, 17040. https://doi.org/10.1038/nrdp.2017.40
- Stabler S. P. (2013). Vitamin B12 deficiency. The New England journal of medicine, 368(21), 2041–2042. https://doi.org/10.1056/NEJMc1304350
- Campdesuner, V., Teklie, Y., Alkayali, T., Pierce, D., & George, J. (2020). Nitrous Oxide-Induced Vitamin B12 Deficiency Resulting in Myelopathy. Cureus, 12(7), e9088. https://doi.org/10.7759/cureus.9088
- Crider, K. S., Bailey, L. B., & Berry, R. J. (2011). Folic acid food fortification-its history, effect, concerns, and future directions. Nutrients, 3(3), 370–384. https://doi.org/10.3390/nu3030370
- Panozzo, M. P., Antico, A., & Bizzaro, N. (2025). Monitoring the follow-up of autoimmune chronic atrophic gastritis using parietal cell antibodies and markers of gastric function. Journal of translational autoimmunity, 10, 100273. https://doi.org/10.1016/j.jtauto.2025.100273
- Staley, K., Ahmadi, K. R., Carter, K., Cowan, K., Seage, H., Visser, P., Ward, N., & Hooper, M. (2022). Research priorities in pernicious anaemia: James Lind Alliance Priority Setting Partnership. BMJ open, 12(8), e065166. https://doi.org/10.1136/bmjopen-2022-065166
- Tyler, N., Hodkinson, A., Ahlam, N., Giles, S., Zhou, A., & Panagioti, M. (2022). Patient safety, self-injection, and B12 deficiency: a UK cross-sectional survey. The British journal of general practice : the journal of the Royal College of General Practitioners, 72(725), e891–e898. https://doi.org/10.3399/BJGP.2021.0711