Key Takeaways
Multiple myeloma is a cancer of plasma cells, a type of white blood cell that produces antibodies. In this condition, abnormal plasma cells multiply uncontrollably and accumulate in the bone marrow. It is slightly more common in males than females and most common between 50 – 80 years old.
- Pathophysiology ▾: Multiple Myeloma is characterized by the clonal proliferation of malignant plasma cells in the bone marrow, leading to the overproduction of a single, non-functional M-protein.
- Multiple myeloma symptoms ▾: Clinical presentation is defined by the CRAB criteria (HyperCalcemia, Renal failure, Anemia, Bone lesions), which signify disease-related end-organ damage.
- Laboratory investigations ▾: Diagnosis combines blood and urine tests for M-protein (SPEP, SFLC, UPEP), a bone marrow biopsy showing ≥10% clonal plasma cells, FISH cytogenetics for risk, and imaging — preferably low-dose whole-body CT, MRI, or PET-CT.
- Treatment and management ▾: The first-line treatment for multiple myeloma is a quadruplet regimen — most often daratumumab plus VRd (D-VRd) — based on the PERSEUS, CEPHEUS, and IMROZ trials, replacing the older triplet standard.
*Click ▾ for more information
Multiple myeloma is a cancer of plasma cells, the immune-system cells that make antibodies. In multiple myeloma, one plasma cell becomes abnormal and copies itself over and over inside the bone marrow, crowding out healthy blood cells and damaging bone, kidneys, and the immune system. It is the second most common blood cancer worldwide and is most often diagnosed in adults between 50 and 80 years old, with a median age around 69 [9].
This guide walks through what the disease is, how it shows up, how doctors diagnose and stage it, and how treatment has changed in the last few years.
What is multiple myeloma (MM)?
Multiple myeloma is a chronic blood cancer in which abnormal plasma cells multiply without control inside the bone marrow. It is currently treatable but not curable, and most modern treatment aims for long, deep remissions rather than eradication [9].
Who gets it
Multiple myeloma mostly affects older adults. It is slightly more common in men than in women. In the United States, people of Black African descent are roughly twice as likely as White individuals to develop multiple myeloma, a disparity thought to involve a mix of genetic, environmental, and healthcare-access factors [9].
A spectrum, not a single disease
Active multiple myeloma rarely appears out of nowhere. It usually develops from earlier, quieter stages.
- Monoclonal Gammopathy of Undetermined Significance (MGUS). The earliest and most common precursor. The patient has a small M-protein (under 3.0 g/dL) and fewer than 10% clonal plasma cells in the marrow, but no organ damage. MGUS is found in over 3% of people aged 50 and older, and it progresses to active myeloma at about 1% per year [9].
- Smoldering Multiple Myeloma (SMM). A higher-risk middle stage. M-protein is ≥3.0 g/dL or clonal plasma cells are ≥10%, but again, no organ damage. The risk of becoming active myeloma is highest in the first five years after diagnosis, so close monitoring (and sometimes early treatment in high-risk cases) is standard [6].
Understanding this spectrum matters because catching disease early — sometimes before symptoms appear — can change outcomes.
What are plasma cells?
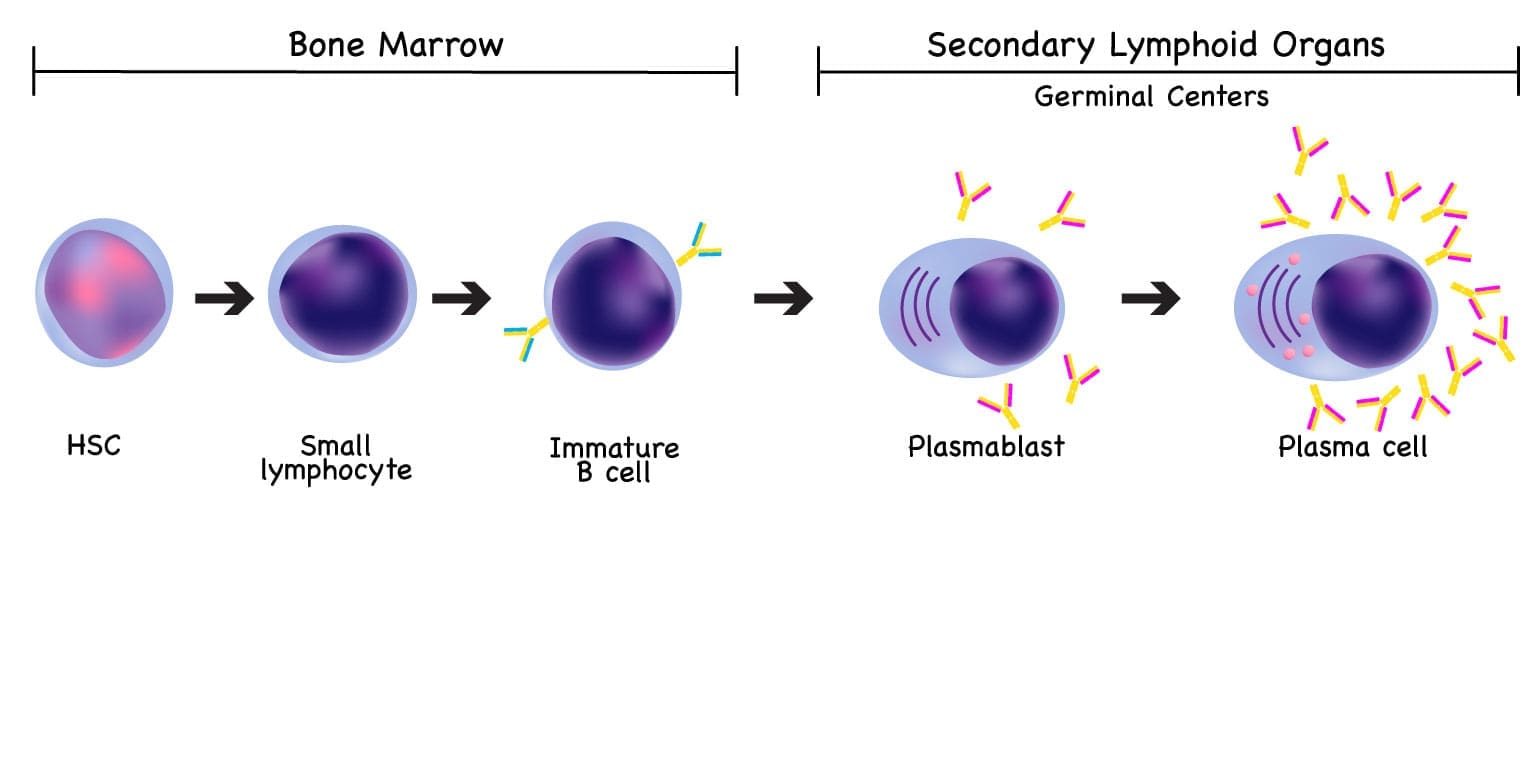
Plasma cells are the antibody factories of the immune system. They are the final, fully matured form of B lymphocytes, and their entire job is to produce immunoglobulins (antibodies) that fight infection.
Plasma cells start as B cells in the bone marrow. When a B cell encounters an antigen — say, a protein on a virus — it gets help from T cells and dendritic cells, multiplies, and differentiates into either a memory B cell (which remembers the threat for next time) or a plasma cell (which churns out antibodies). Most long-lived plasma cells return to the bone marrow, where they keep producing antibodies for months to years.
When this system goes wrong, it can produce several plasma cell diseases, including multiple myeloma, Waldenström's macroglobulinemia, AL amyloidosis, and Common Variable Immunodeficiency (CVID).
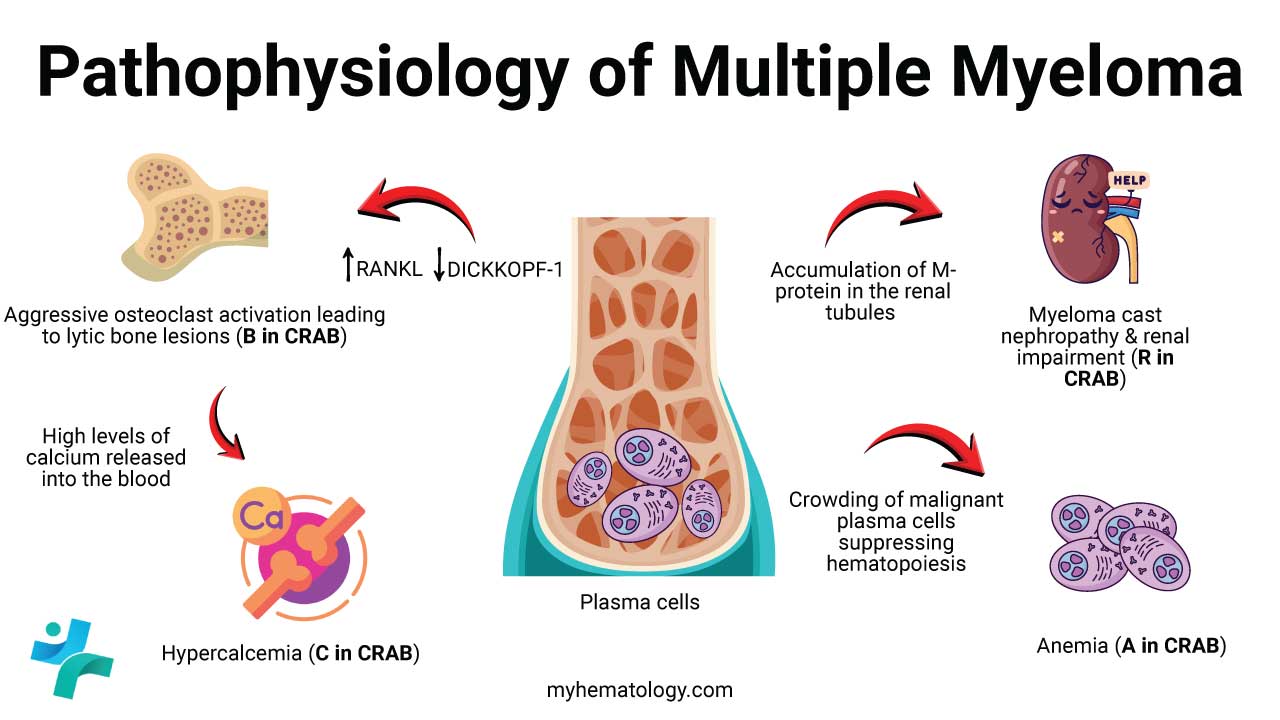
Pathophysiology of Multiple Myeloma
The disease has three intertwined drivers: a genetic problem inside the plasma cell, a supportive bone marrow environment that helps the cancer grow, and the resulting damage to specific organs.
The genetic spark
Multiple myeloma usually begins with a genetic accident in a B cell before it becomes a plasma cell. Two events are most common:
- Aneuploidy — an abnormal number of chromosomes, often extra copies of odd-numbered chromosomes.
- Immunoglobulin heavy chain (IgH) translocations — pieces of chromosome 14 swap places with other chromosomes (for example, t(4;14) or t(14;16)).
These early lesions push the cell toward malignancy. Later, secondary mutations stack up — loss of tumor-suppressor genes like TP53 (on chromosome 17p), or activation of oncogenes like MYC — and the disease grows more aggressive and harder to treat [9].
Why the bone marrow matters
Myeloma cells are not free agents. They depend on the bone marrow microenvironment (BMM) — the network of stromal cells, blood vessels, and signaling molecules around them. Stromal cells release interleukin-6 (IL-6), the most important survival signal for myeloma cells. The cancer cells, in turn, suppress local immune defenses and reprogram the marrow into a niche that protects them.
How damage happens: the CRAB picture
The pathology converges on four classic features, remembered as CRAB:
- C — Hypercalcemia from massive bone breakdown.
- R — Renal impairment from light chains clogging kidney tubules.
- A — Anemia from marrow crowding and inflammatory signaling.
- B — Bone disease from overactive osteoclasts and suppressed osteoblasts, producing the famous "punched-out" lytic lesions.
The bone changes deserve special attention because they are the most visually distinctive feature of myeloma. Normally, bone is constantly remodeled by osteoclasts (which break down bone) and osteoblasts (which build it back up). In myeloma, signaling molecules like RANKL drive osteoclasts into overdrive, while inhibitors such as DKK1 silence osteoblasts. The result is pure bone destruction with no repair — and as the bone dissolves, calcium spills into the bloodstream.
Multiple Myeloma (MM) Symptoms
The symptoms of active multiple myeloma trace back to that same CRAB framework, plus a few important extras. Because early symptoms are vague — fatigue, back pain, frequent infections — myeloma is often mistaken for arthritis, ordinary anemia, or simply aging.
Bone disease and pain
This is the most common reason patients seek help. Bone pain is typically located in the back, ribs, or pelvis, gets worse with movement, and does not ease with rest. Weakened bones can fracture from minor trauma or even spontaneously. If a vertebra collapses onto the spinal cord, the result — sudden severe back pain, leg weakness, numbness, or loss of bladder or bowel control — is a medical emergency requiring immediate care.
Anemia
Anemia is the most common laboratory finding at diagnosis. Patients describe extreme fatigue, breathlessness on exertion, pale skin, and a fast heartbeat as the body tries to compensate for low oxygen-carrying capacity.
Hypercalcemia
High blood calcium produces a memorable triad — stones, groans, and moans: kidney stones with extra thirst and urination; abdominal pain, nausea, and constipation; and confusion, depression, or muscle weakness.
Renal insufficiency
Kidney damage often starts silently. As it worsens, patients may notice fatigue, swelling in the legs, or reduced urine output. The main culprit is free light chains (Bence-Jones proteins) precipitating in the kidney tubules — a process called myeloma cast nephropathy.
Beyond CRAB
- Recurrent infections. Even though myeloma cells flood the body with M-protein, that protein is functionally useless. Meanwhile, normal antibody production drops — a state called functional hypogammaglobulinemia. Patients become unusually susceptible to bacterial infections, especially from encapsulated organisms like Streptococcus pneumoniae.
- Peripheral neuropathy. Tingling, numbness, or burning in the hands and feet, sometimes from the disease itself and sometimes from treatments such as bortezomib.
- Hyperviscosity syndrome. Uncommon in multiple myeloma (more typical of Waldenström's macroglobulinemia), but possible when IgM or IgA M-proteins reach very high levels. Symptoms include headaches, blurred vision, and abnormal bleeding.
- Constitutional symptoms. Weight loss, poor appetite, and general malaise as the disease and its inflammation progress.
Persistent unexplained back pain, recurrent infections, or unexplained anemia in someone over 50 deserves a workup that includes serum protein electrophoresis [10].
Investigation and Diagnosis
Diagnosing multiple myeloma takes a layered approach: blood tests, bone marrow sampling, and imaging. The goal is not just to find the cancer, but to determine whether it is causing damage and how high the risk is.
Blood and chemistry tests
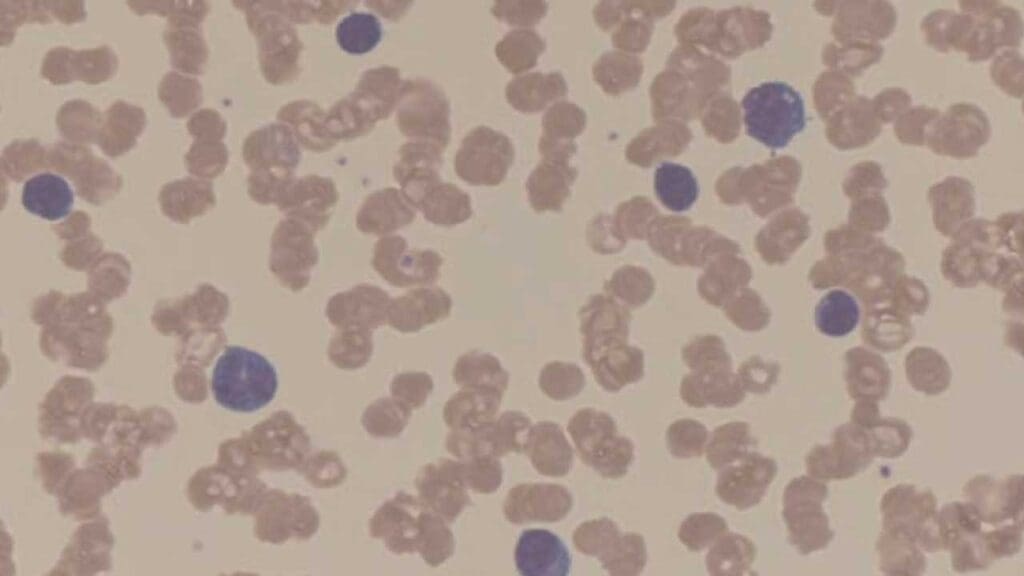
- Complete blood count (CBC). Anemia is the most common abnormality. Low white cell or platelet counts can also appear when marrow is crowded. The peripheral blood smear may show rouleaux formation, where red cells stack like coins because the M-protein coats them.
- Chemistry panel. Calcium (for hypercalcemia), creatinine and BUN (for kidney function), and lactate dehydrogenase (LDH) and beta-2 microglobulin (β2M) as prognostic markers.
Detecting the M-protein
These tests look for the abnormal monoclonal antibody made by the cancer:
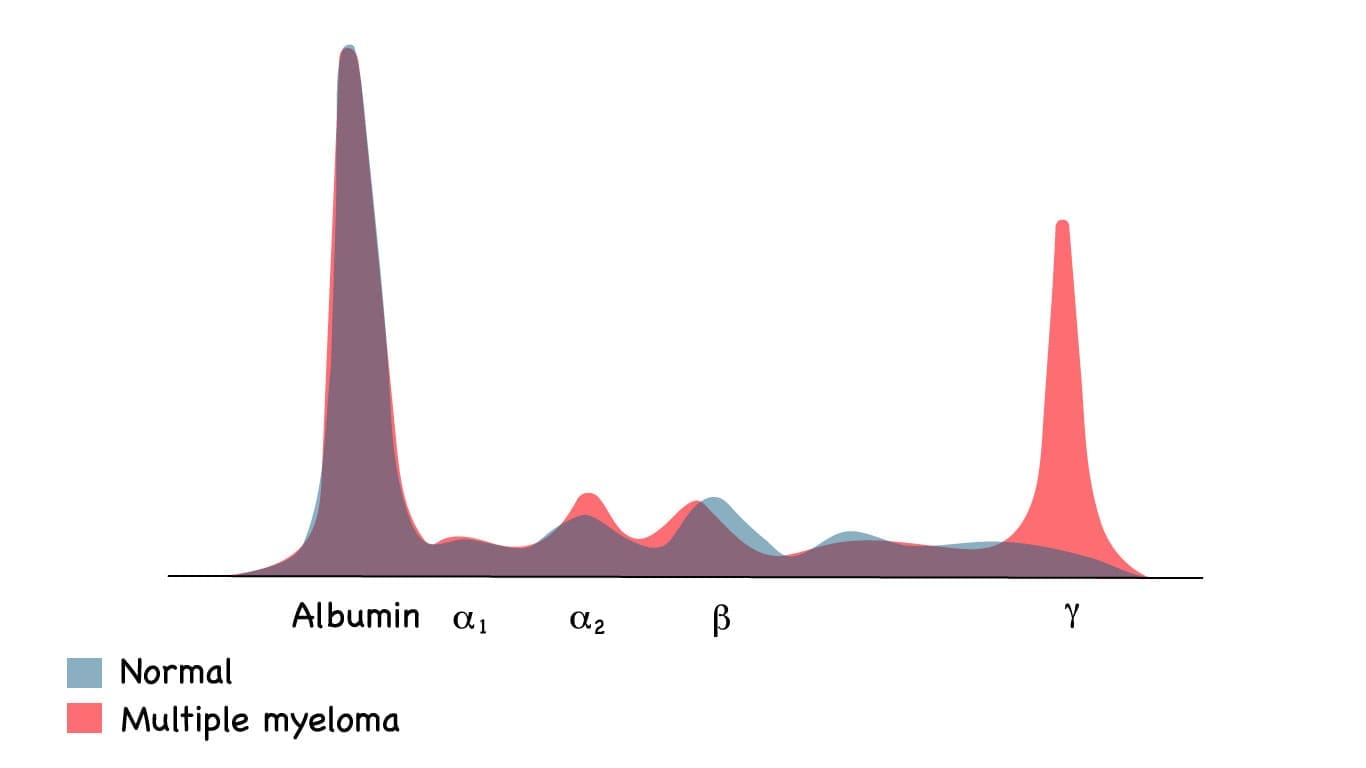
- Serum protein electrophoresis (SPEP) separates blood proteins and reveals the characteristic M-spike, usually in the gamma region.
- Serum free light chain (SFLC) assay measures unbound kappa and lambda light chains and calculates their ratio. An abnormal ratio can detect clonal disease even when the M-spike is small or absent.
- Urine protein electrophoresis (UPEP) detects Bence-Jones proteins, which are highly damaging to the kidneys.
Highly sensitive mass spectrometry (e.g., MALDI-TOF) is replacing SPEP in major centers, as it uniquely distinguishes therapeutic antibodies from actual M-proteins to prevent false positives. Furthermore, blood-based MRD testing via mass spectrometry and ctDNA now allows non-invasive remission tracking and actively guides treatment de-escalation in clinical trials [11].
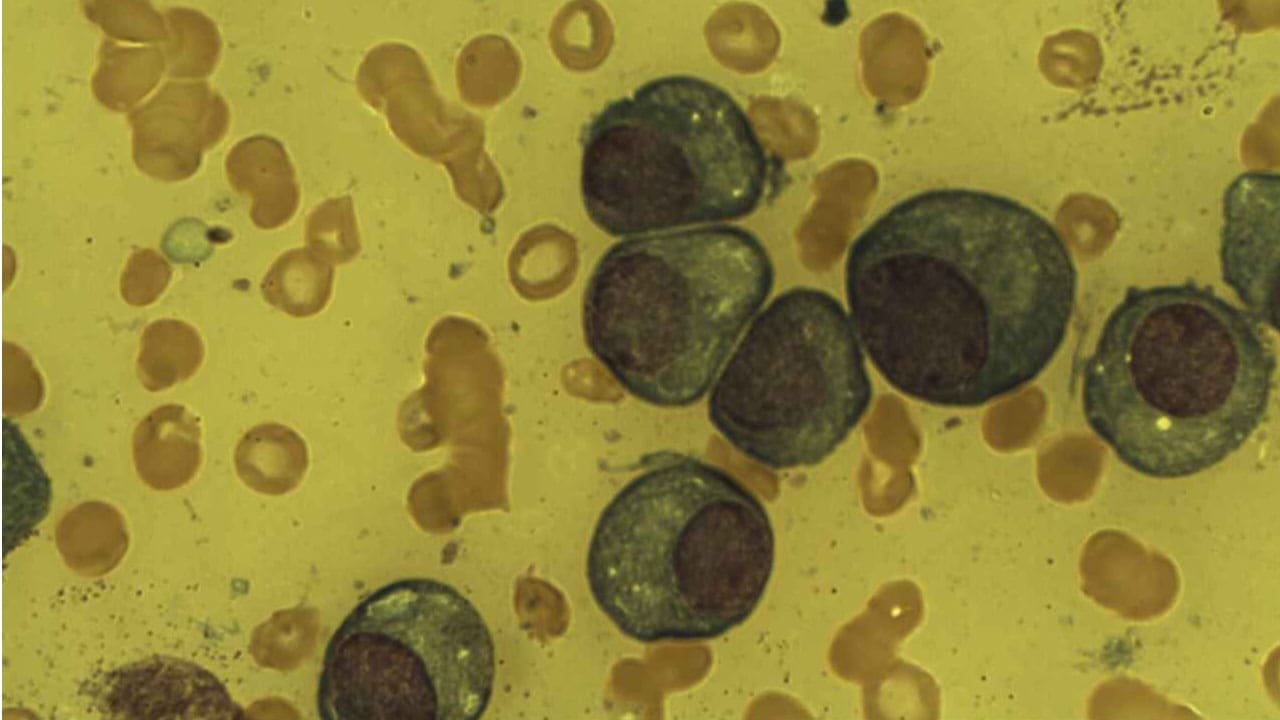
Bone Marrow Evaluation (The Definitive Test)
A bone marrow aspiration and biopsy is required for diagnosis. Two findings matter most:
- At least 10% clonal plasma cells in the marrow (or a biopsy-proven plasmacytoma).
- Confirmation that the cells are clonal, usually with flow cytometry showing surface markers like CD38 and CD138.
Cytogenetics and risk
Fluorescence in situ hybridization (FISH) on the marrow sample looks for high-risk genetic changes including del(17p), t(4;14), t(14;16), and gain or amplification of chromosome 1q. Any of these signals more aggressive disease and influences treatment intensity [7].
Imaging
Because myeloma destroys bone, imaging is essential.
- Low-dose whole-body CT (LD-WBCT) is now preferred over plain X-ray skeletal survey. It is far more sensitive at detecting lytic lesions [10].
- MRI and PET-CT detect soft-tissue involvement, extramedullary disease, and focal lesions that CT may miss. MRI of the spine is essential when spinal cord compression is suspected.
The International Myeloma Working Group (IMWG) Diagnostic Criteria
The International Myeloma Working Group (IMWG) criteria, last updated in 2014, define when a patient officially has active multiple myeloma and needs treatment [6].
Active multiple myeloma is diagnosed when all three of the following are present:
- Clonal plasma cells ≥10% in the bone marrow (or a biopsy-proven plasmacytoma).
- M-protein in serum or urine.
- At least one Myeloma Defining Event (MDE). MDEs include the four CRAB features or the SLiM biomarkers of high-risk disease, even in the absence of symptoms:
- S — Sixty: Bone marrow plasma cells ≥60%.
- Li — Light chain ratio: SFLC ratio ≥100 (with the involved chain ≥100 mg/L).
- M — MRI lesions: More than one focal lesion on MRI.
The SLiM additions allow physicians to treat patients with extremely high-risk disease before irreversible damage occurs.
| Feature | MGUS | Smoldering Multiple Myeloma | Active Multiple Myeloma |
| M-Protein | Low (< 3 g/dL) | Higher (≥ 3 g/dL) | Any level |
| Clonal Plasma Cells | < 10% | 10% - 60% | ≥ 10% |
| Myeloma Defining Event | Absent | Absent | Present (CRAB or SLiM criteria) |
Differential Diagnosis for Multiple Myeloma
Several conditions can look like multiple myeloma. The table below highlights what to look for.
| Condition | Underlying Pathology | Distinguishing Feature (vs. MM) |
| MGUS | Small, stable clone of abnormal plasma cells. | <10% clonal plasma cells; no MDEs |
| Smoldering Multiple Myeloma | Larger plasma cell clone | ≥10% clonal cells and/or M-protein ≥3 g/dL; no CRAB features |
| Systemic AL Amyloidosis | Misfolded light chains deposit as amyloid fibrils | Often <10% plasma cells; damage from protein deposition, not lytic lesions |
| Solitary Plasmacytoma | Localized plasma cell tumor | Single site; no systemic CRAB |
| Monoclonal Gammopathy of Renal Significance (MGRS) | Small clone; toxic M-protein damages kidneys | Kidney failure with low clonal burden and no other CRAB |
| Lymphoma / Leukemia | Lymphocytes, blasts, or myeloid cells | Cells are not terminally differentiated plasma cells; M-protein usually absent |
Staging multiple myeloma
Staging predicts how aggressive the disease will be and helps guide how intense treatment should be.
The Revised International Staging System (R-ISS) uses four pieces of information: serum albumin, β2-microglobulin, LDH, and high-risk cytogenetics from FISH (del(17p), t(4;14), or t(14;16)).
Revised International Staging System (R-ISS)
| Stage | Criteria | Prognosis |
|---|---|---|
| R-ISS I | β2-microglobulin < 3.5 mg/L Albumin ≥ 3.5 g/dL Standard-risk cytogenetics (absence of high-risk cytogenetic abnormalities) Normal LDH | Longest survival |
| R-ISS II | Not R-ISS stage I or III | Intermediate survival |
| R-ISS III | β2-microglobulin > 5.5 mg/L And either: High-risk cytogenetics (presence of del(17p) and/or t(4;14) and/or t(14;16)) or elevated LDH | Shortest survival |
A newer Second Revision (R2-ISS), published in 2022, refines R-ISS by also including chromosome 1q gain or amplification, which is now recognized as a significant adverse marker [7]. R2-ISS is increasingly used in clinical trials and is expected to replace R-ISS in routine practice.
When myeloma comes back after treatment, it is called relapsed myeloma; if it stops responding to current treatment, it is called refractory myeloma. Restaging often guides the next line of therapy.
Treatment and Management
The treatment of multiple myeloma has changed dramatically in the last few years. The big shift: triplet regimens have been replaced by quadruplet regimens as the preferred first-line treatment for most patients [1,2].
The goals of modern therapy are:
- Symptom control for CRAB features.
- Deep remission, increasingly measured by Minimal Residual Disease (MRD) negativity — the absence of detectable myeloma cells using highly sensitive lab tests. MRD-negative status is now a key endpoint in clinical trials and predicts longer remission [8].
- Prolonged survival with maintained quality of life.
Newly diagnosed multiple myeloma: who gets a transplant?
Treatment paths split based on whether a patient is fit enough for an autologous stem cell transplant (ASCT).
Transplant-eligible patients
The current preferred sequence is induction → transplant → maintenance.
- Induction. Per NCCN Version 1.2025 guidelines, the Category 1 preferred regimen is D-VRd: daratumumab (anti-CD38 antibody), bortezomib (proteasome inhibitor), lenalidomide (immunomodulatory drug), and dexamethasone (steroid). This quadruplet replaced the older VRd triplet after the PERSEUS trial showed significantly better progression-free survival and MRD negativity rates [1,2]. The isatuximab-based quadruplet (Isa-VRd) is also preferred and has Category 1 evidence for transplant-ineligible patients from the IMROZ trial [3].
- Consolidation with ASCT. After induction, patients receive high-dose melphalan followed by re-infusion of their own previously collected stem cells.
- Maintenance. Long-term lenalidomide (sometimes with daratumumab in high-risk disease) prevents relapse and extends remission [1].
Transplant-ineligible patients
For older or frail patients, treatment focuses on continuous, well-tolerated combinations. Preferred regimens now include D-VRd (subcutaneous daratumumab, FDA-approved in 2025 based on the CEPHEUS trial) and Isa-VRd. DRd (daratumumab + lenalidomide + dexamethasone) remains an option, particularly for patients who cannot tolerate bortezomib [1].
Relapsed and refractory multiple myeloma (RRMM)
When myeloma comes back, the strategy is to switch to drug classes the patient has not yet received or has not failed. The four main classes underpin nearly every regimen.
Novel Agents (The Pillars of Modern MM Therapy)
| Class | How it works | Examples |
|---|---|---|
| Proteasome Inhibitors (PIs) | Block the cell's protein-recycling machinery, killing plasma cells that depend on it | Bortezomib, carfilzomib, ixazomib |
| Immunomodulatory Drugs (IMiDs) | Reshape the tumor environment and activate immune cells | Lenalidomide, pomalidomide |
| Monoclonal Antibodies (MoAbs) | Mark plasma cells for immune destruction | Daratumumab, isatuximab |
| ADC (Antibody-Drug Conjugate) | Deliver a toxin directly to BCMA-expressing plasma cells | Belantamab mafodotin* |
| CELMoDs (Cereblon E3 Ligase Modulators) | Next-generation oral agents that degrade transcription factors; highly effective even in lenalidomide/pomalidomide-refractory disease. | Iberdomide, mezigdomide [12]. |
*Though previously withdrawn as monotherapy, belantamab mafodotin is now a standard-of-care for early relapse. Phase 3 trials (DREAMM-7/8) demonstrated profound survival benefits when combined with bortezomib/dexamethasone (BVd) or pomalidomide/dexamethasone (BPd) compared to standard triplets [13,14].
Immunotherapy: CAR T-cells and bispecific antibodies
This is where myeloma treatment has moved fastest [4,5].
CAR T-cell therapy. A patient's own T-cells are collected, genetically engineered to recognize BCMA on plasma cells, and re-infused. Two products are FDA-approved:
- Idecabtagene vicleucel (ide-cel) — approved after ≥2 prior lines of therapy.
- Ciltacabtagene autoleucel (cilta-cel) — approved after ≥1 prior line in lenalidomide-refractory patients . Cilta-cel produces a median progression-free survival of around 35 months in this setting, with about a third of patients in ongoing remission beyond five years [4,5].
Bispecific T-cell engagers. These off-the-shelf antibodies have two arms — one grabs a T-cell, the other grabs a myeloma cell — physically forcing the immune system to engage. Four are FDA-approved:
- Teclistamab, elranatamab, and linvoseltamab (all anti-BCMA).
- Talquetamab (anti-GPRC5D — the first non-BCMA target).
Both CAR-T and bispecifics carry significant short-term risks, including cytokine release syndrome and neurotoxicity, and are given only at specialized centers [5].
Targeted therapy for t(11;14)
About 15–20% of multiple myeloma cases carry the t(11;14) translocation. These cells often overexpress BCL-2, making them especially sensitive to venetoclax, a BCL-2 inhibitor used (off-label or investigationally) in this subgroup at relapse.
Supportive care
Supportive care is not optional — it is central to quality of life and survival.
- Bone health. Intravenous bisphosphonates (zoledronic acid) or denosumab reduce fractures and bone pain. Localized radiation therapy can relieve severe focal bone pain.
- Infection prevention. Standard care includes appropriate vaccinations (influenza, pneumococcal, COVID-19, zoster) and antiviral prophylaxis. Because BCMA-targeted therapies (bispecifics/CAR-T) cause profound hypogammaglobulinemia, prophylactic Intravenous Immunoglobulin (IVIG) is now standard for patients with IgG <400 mg/dL, alongside strict PJP (Pneumocystis jirovecii pneumonia) and viral prophylaxis, to prevent fatal opportunistic infections [15].
- Kidney protection. Aggressive hydration, prompt treatment of hypercalcemia, and avoidance of nephrotoxic drugs help preserve kidney function.
- Pain management. A combination of medications and, when needed, focal radiation.
Living with multiple myeloma
Beyond the medical regimens, life with myeloma is shaped by fatigue, the constant threat of infection, and the long arc of a relapsing disease. Caregivers help by tracking medications and side effects, watching for warning signs (sudden severe back pain with leg weakness, fever, new confusion), and supporting infection precautions during intensive treatment. Most cancer centers offer social workers, palliative care, and patient support groups; using these resources early — not just at end stages — is consistently associated with better quality of life.
Frequently Asked Questions (FAQs)
What are the early signs of multiple myeloma?
Multiple myeloma often starts quietly. The most common early signs are persistent fatigue, unexplained bone pain (especially in the back, ribs, or hips), frequent infections, and abnormal blood test results such as anemia, high blood calcium, or kidney problems. Because these signs overlap with many ordinary conditions, myeloma is sometimes diagnosed only after months of symptoms. Anyone over 50 with persistent unexplained bone pain or recurring infections should ask their doctor about a blood test for monoclonal protein.
Is multiple myeloma curable?
Multiple myeloma is currently considered treatable but not curable. Modern therapies — including quadruplet drug regimens, autologous stem cell transplant, CAR T-cell therapy, and bispecific antibodies — can produce deep, long-lasting remissions, and some patients live more than a decade after diagnosis. Five-year relative survival in the United States is now about 60%, up from roughly 35% two decades ago. A small number of patients reach prolonged minimal residual disease (MRD) negativity, which some specialists describe as a "functional cure," though formal cure remains an active research goal.
What is the difference between MGUS, smoldering myeloma, and active multiple myeloma?
These three conditions sit on the same spectrum of plasma cell disorders. MGUS has a low M-protein (under 3 g/dL), fewer than 10% clonal plasma cells in the bone marrow, and no organ damage; it progresses to myeloma at about 1% per year. Smoldering myeloma has a higher M-protein (≥3 g/dL) or ≥10% clonal plasma cells but still no organ damage. Active multiple myeloma is diagnosed when there is at least one Myeloma Defining Event — either a CRAB feature (high calcium, kidney failure, anemia, bone lesion) or a SLiM biomarker (≥60% plasma cells, light-chain ratio ≥100, or more than one MRI focal lesion).
What is the standard first treatment for newly diagnosed multiple myeloma?
For most patients newly diagnosed with multiple myeloma, the current preferred first-line treatment is a four-drug (quadruplet) regimen. The most widely used is D-VRd: daratumumab (an anti-CD38 antibody), bortezomib (a proteasome inhibitor), lenalidomide (an immunomodulatory drug), and dexamethasone (a steroid). This is now the NCCN Category 1 preferred induction regimen for both transplant-eligible and many transplant-ineligible patients, based on the PERSEUS and CEPHEUS trials. Patients fit enough for an autologous stem cell transplant typically receive induction, then transplant, then long-term maintenance with lenalidomide.
What is CAR T-cell therapy for myeloma, and who can receive it?
CAR T-cell therapy is a one-time treatment that genetically modifies a patient's own T-cells to recognize and kill myeloma cells. Two products are FDA-approved for multiple myeloma: idecabtagene vicleucel (ide-cel) and ciltacabtagene autoleucel (cilta-cel). Both target BCMA, a protein found on plasma cells. Following 2024 label expansions, cilta-cel is now approved after as few as one prior line of therapy in lenalidomide-refractory patients, and ide-cel is approved after two prior lines. CAR T-cell therapy can produce remissions lasting years but carries serious short-term risks, including cytokine release syndrome and neurotoxicity, so it is given only at specialized centers.
How can caregivers help someone living with multiple myeloma?
Caregivers play a central role. Practical support includes helping track appointments, medications, and side effects; recognizing warning signs that need urgent attention (sudden severe back pain with leg weakness, signs of infection like fever, confusion, or new bone pain); supporting infection precautions (vaccinations, hand hygiene, avoiding sick contacts during intensive treatment); and watching for fatigue and mood changes. Emotional support matters as much as logistics — myeloma is a long disease, and the emotional load on patients and families is often underestimated. Most cancer centers have social workers, palliative care teams, and patient support groups; using them early is associated with better quality of life.
Glossary of Related Medical Terms
- Plasma cell — A mature B lymphocyte whose job is to make antibodies. In myeloma, a single plasma cell becomes cancerous and copies itself.
- Monoclonal protein (M-protein, paraprotein) — An abnormal antibody produced in large amounts by myeloma cells. It looks like a sharp spike on a lab test called electrophoresis.
- Clonal — All coming from the same original cell. In myeloma, the abnormal plasma cells are clones of one ancestor.
- CRAB criteria — The four classic signs of active myeloma: high Calcium, Renal (kidney) failure, Anemia, and Bone lesions.
- MGUS (Monoclonal Gammopathy of Undetermined Significance) — A symptom-free precursor state with a small M-protein. About 1% of cases progress to myeloma each year.
- Smoldering myeloma (SMM) — A higher-risk in-between stage. More plasma cells or M-protein than MGUS, but no organ damage yet.
- Bence-Jones proteins — Free immunoglobulin light chains found in urine. They can clog and damage the kidneys.
- Lytic lesion — A "punched-out" hole in bone where myeloma cells have triggered destruction.
- Osteoclast — The bone cell that breaks bone down. Overactive in myeloma.
- Osteoblast — The bone cell that builds bone. Suppressed in myeloma.
- Bone marrow microenvironment (BMM) — The supportive cells, blood vessels, and signaling molecules around plasma cells in the marrow. Myeloma cells depend on it.
- Autologous stem cell transplant (ASCT) — A treatment where a patient's own stem cells are collected, the marrow is wiped out with high-dose chemotherapy, and the cells are returned to rebuild it.
- Induction therapy — The first intensive course of drugs given to bring the disease under control.
- Maintenance therapy — Lower-intensity treatment given long-term to keep the disease in remission.
- Proteasome inhibitor (PI) — A drug class (e.g., bortezomib) that blocks the cell's protein-recycling system, killing plasma cells that depend on it.
- Immunomodulatory drug (IMiD) — A drug class (e.g., lenalidomide) that reshapes the tumor environment and activates immune cells.
- Monoclonal antibody (MoAb) — A lab-made antibody that targets a specific protein on a myeloma cell. Examples: daratumumab, isatuximab (both anti-CD38).
- BCMA (B-cell Maturation Antigen) — A protein on the surface of plasma cells. It's a key target for newer myeloma immunotherapies.
- CAR T-cell therapy — A treatment where a patient's T-cells are engineered to recognize and kill myeloma cells.
- Bispecific antibody — A lab-made antibody with two arms: one grabs a T-cell, the other grabs a myeloma cell, forcing them together so the T-cell kills the cancer.
- Minimal residual disease (MRD) — Very small amounts of cancer left after treatment, detectable only by sensitive lab methods. MRD-negative status predicts longer remission.
- R-ISS / R2-ISS — Staging systems that combine blood markers and genetics to predict prognosis and guide therapy.
Disclaimer: This article is intended for informational purposes only and is specifically targeted towards medical students. It is not intended to be a substitute for informed professional medical advice, diagnosis, or treatment. While the information presented here is derived from credible medical sources and is believed to be accurate and up-to-date, it is not guaranteed to be complete or error-free. See additional information.
References
- Kumar, S. K., Callander, N. S., Adekola, K., Anderson, L. D., Jr, Baljevic, M., Baz, R., Campagnaro, E., Costello, C., D’Angelo, C., Derman, B., Devarakonda, S., Elsedawy, N., Godara, A., Godby, K., Hillengass, J., Holmberg, L., Htut, M., Huff, C. A., Hultcrantz, M., Kang, Y., Larson, S., Lee, H. C., Liedtke, M., Martin, T., Omel, J., Robinson, T., Rosenberg, A., Schroeder, M. A., Sherbenou, D., Suvannasankha, A., Valent, J., Varshavsky-Yanovsky, A. N., Vogl, D., Kovach, E., & Kumar, R. (2025). NCCN Guidelines® Insights: Multiple Myeloma, Version 1.2025: Featured Updates to the NCCN Guidelines®. Journal of the National Comprehensive Cancer Network, 23(5), 132-140. Retrieved May 20, 2026, from https://doi.org/10.6004/jnccn.2025.0023
- Sonneveld, P., Dimopoulos, M. A., Boccadoro, M., Quach, H., Ho, P. J., Beksac, M., Hulin, C., Antonioli, E., Leleu, X., Mangiacavalli, S., Perrot, A., Cavo, M., Belotti, A., Broijl, A., Gay, F., Mina, R., Nijhof, I. S., van de Donk, N. W. C. J., Katodritou, E., Schjesvold, F., … PERSEUS Trial Investigators (2024). Daratumumab, Bortezomib, Lenalidomide, and Dexamethasone for Multiple Myeloma. The New England journal of medicine, 390(4), 301–313. https://doi.org/10.1056/NEJMoa2312054
- Facon, T., Dimopoulos, M. A., Leleu, X. P., Beksac, M., Pour, L., Hájek, R., Liu, Z., Minarik, J., Moreau, P., Romejko-Jarosinska, J., Spicka, I., Vorobyev, V. I., Besemer, B., Ishida, T., Janowski, W., Kalayoglu-Besisik, S., Parmar, G., Robak, P., Zamagni, E., Goldschmidt, H., … IMROZ Study Group (2024). Isatuximab, Bortezomib, Lenalidomide, and Dexamethasone for Multiple Myeloma. The New England journal of medicine, 391(17), 1597–1609. https://doi.org/10.1056/NEJMoa2400712
- San-Miguel, J., Dhakal, B., Yong, K., Spencer, A., Anguille, S., Mateos, M. V., Fernández de Larrea, C., Martínez-López, J., Moreau, P., Touzeau, C., Leleu, X., Avivi, I., Cavo, M., Ishida, T., Kim, S. J., Roeloffzen, W., van de Donk, N. W. C. J., Dytfeld, D., Sidana, S., Costa, L. J., … Einsele, H. (2023). Cilta-cel or Standard Care in Lenalidomide-Refractory Multiple Myeloma. The New England journal of medicine, 389(4), 335–347. https://doi.org/10.1056/NEJMoa2303379
- Dima, D., Banerjee, R., & Hansen, D. K. (2025). CAR T-cell therapy and bispecific antibodies in the management of multiple myeloma. Hematology. American Society of Hematology. Education Program, 2025(1), 324–333. https://doi.org/10.1182/hematology.2025000721
- Rajkumar, S. V., Dimopoulos, M. A., Palumbo, A., Blade, J., Merlini, G., Mateos, M. V., Kumar, S., Hillengass, J., Kastritis, E., Richardson, P., Landgren, O., Paiva, B., Dispenzieri, A., Weiss, B., LeLeu, X., Zweegman, S., Lonial, S., Rosinol, L., Zamagni, E., Jagannath, S., … Miguel, J. F. (2014). International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. The Lancet. Oncology, 15(12), e538–e548. https://doi.org/10.1016/S1470-2045(14)70442-5
- D'Agostino, M., Cairns, D. A., Lahuerta, J. J., Wester, R., Bertsch, U., Waage, A., Zamagni, E., Mateos, M. V., Dall'Olio, D., van de Donk, N. W. C. J., Jackson, G., Rocchi, S., Salwender, H., Bladé Creixenti, J., van der Holt, B., Castellani, G., Bonello, F., Capra, A., Mai, E. K., Dürig, J., … Sonneveld, P. (2022). Second Revision of the International Staging System (R2-ISS) for Overall Survival in Multiple Myeloma: A European Myeloma Network (EMN) Report Within the HARMONY Project. Journal of clinical oncology : official journal of the American Society of Clinical Oncology, 40(29), 3406–3418. https://doi.org/10.1200/JCO.21.02614
- Kumar, S., Paiva, B., Anderson, K. C., Durie, B., Landgren, O., Moreau, P., Munshi, N., Lonial, S., Bladé, J., Mateos, M. V., Dimopoulos, M., Kastritis, E., Boccadoro, M., Orlowski, R., Goldschmidt, H., Spencer, A., Hou, J., Chng, W. J., Usmani, S. Z., Zamagni, E., … Avet-Loiseau, H. (2016). International Myeloma Working Group consensus criteria for response and minimal residual disease assessment in multiple myeloma. The Lancet. Oncology, 17(8), e328–e346. https://doi.org/10.1016/S1470-2045(16)30206-6
- Kumar, S. K., Rajkumar, V., Kyle, R. A., van Duin, M., Sonneveld, P., Mateos, M. V., Gay, F., & Anderson, K. C. (2017). Multiple myeloma. Nature reviews. Disease primers, 3, 17046. https://doi.org/10.1038/nrdp.2017.46
- Cowan, A. J., Green, D. J., Kwok, M., Lee, S., Coffey, D. G., Holmberg, L. A., Tuazon, S., Gopal, A. K., & Libby, E. N. (2022). Diagnosis and Management of Multiple Myeloma: A Review. JAMA, 327(5), 464–477. https://doi.org/10.1001/jama.2022.0003
- Derman, B. A., & Yee, A. J. (2025). Clinical applications of mass spectrometry in multiple myeloma. Blood advances, 9(24), 6593–6603. https://doi.org/10.1182/bloodadvances.2024015685
- Richardson, P. G., Trudel, S., Popat, R., Mateos, M. V., Vangsted, A. J., Ramasamy, K., Martinez-Lopez, J., Quach, H., Orlowski, R. Z., Arnao, M., Lonial, S., Karanes, C., Pawlyn, C., Kim, K., Oriol, A., Berdeja, J. G., Rodríguez Otero, P., Casas-Avilés, I., Spirli, A., Poon, J., … CC-92480-MM-001 Study Investigators (2023). Mezigdomide plus Dexamethasone in Relapsed and Refractory Multiple Myeloma. The New England journal of medicine, 389(11), 1009–1022. https://doi.org/10.1056/NEJMoa2303194
- Hungria, V., Robak, P., Hus, M., Zherebtsova, V., Ward, C., Ho, P. J., Ribas de Almeida, A. C., Hajek, R., Kim, K., Grosicki, S., Sia, H., Bryant, A., Pitombeira de Lacerda, M., Aparecida Martinez, G., Sureda Balarí, A. M., Sandhu, I., Cerchione, C., Ganly, P., Dimopoulos, M., Fu, C., … DREAMM-7 Investigators (2024). Belantamab Mafodotin, Bortezomib, and Dexamethasone for Multiple Myeloma. The New England journal of medicine, 391(5), 393–407. https://doi.org/10.1056/NEJMoa2405090
- Dimopoulos, M. A., Beksac, M., Pour, L., Delimpasi, S., Vorobyev, V., Quach, H., Spicka, I., Radocha, J., Robak, P., Kim, K., Cavo, M., Suzuki, K., Morris, K., Pompilus, F., Phillips-Jones, A., Zhou, X. L., Fulci, G., Sule, N., Kremer, B. E., Opalinska, J., … DREAMM-8 Investigators (2024). Belantamab Mafodotin, Pomalidomide, and Dexamethasone in Multiple Myeloma. The New England journal of medicine, 391(5), 408–421. https://doi.org/10.1056/NEJMoa2403407
- Sheu, M., Molina Garcia, S., Patel, M., Mushtaq, A., Rust, T., Anns Asif, M., Anwer, F., & Majeed, A. (2025). Infection Prophylaxis with Intravenous Immunoglobulin in Multiple Myeloma Patients Treated with Teclistamab. Oncology, 1–5. Advance online publication. https://doi.org/10.1159/000548964