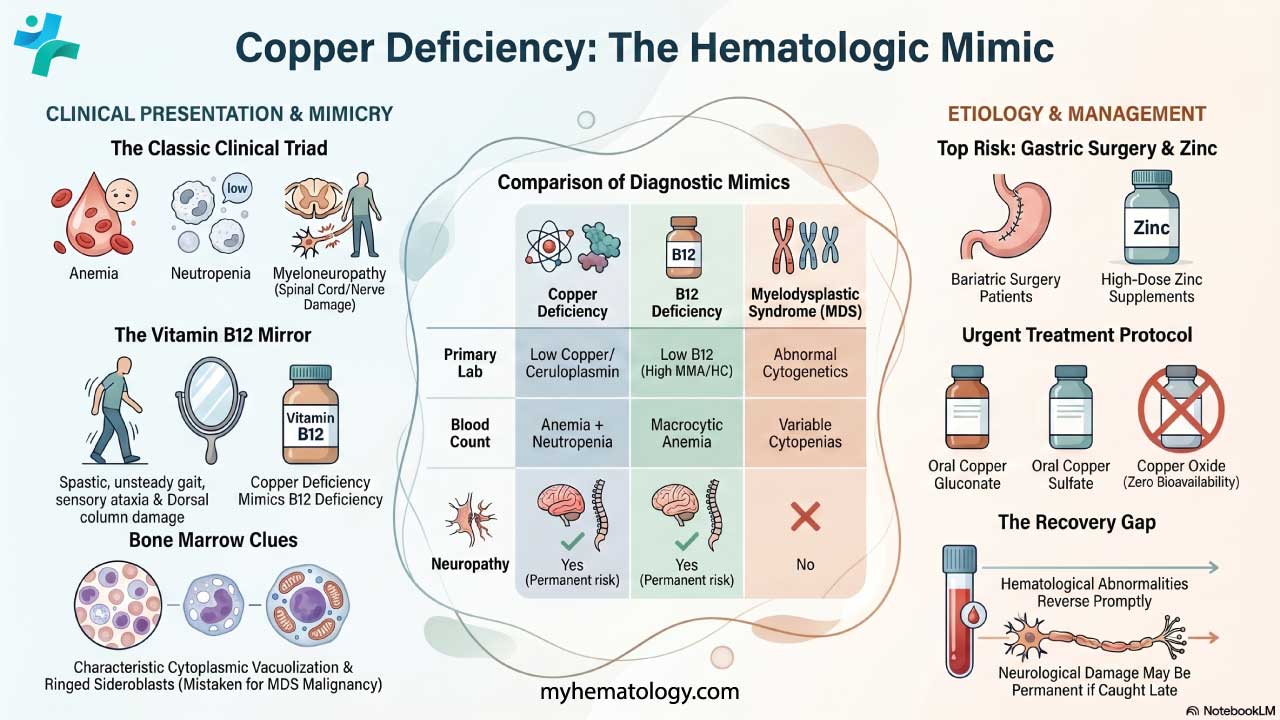
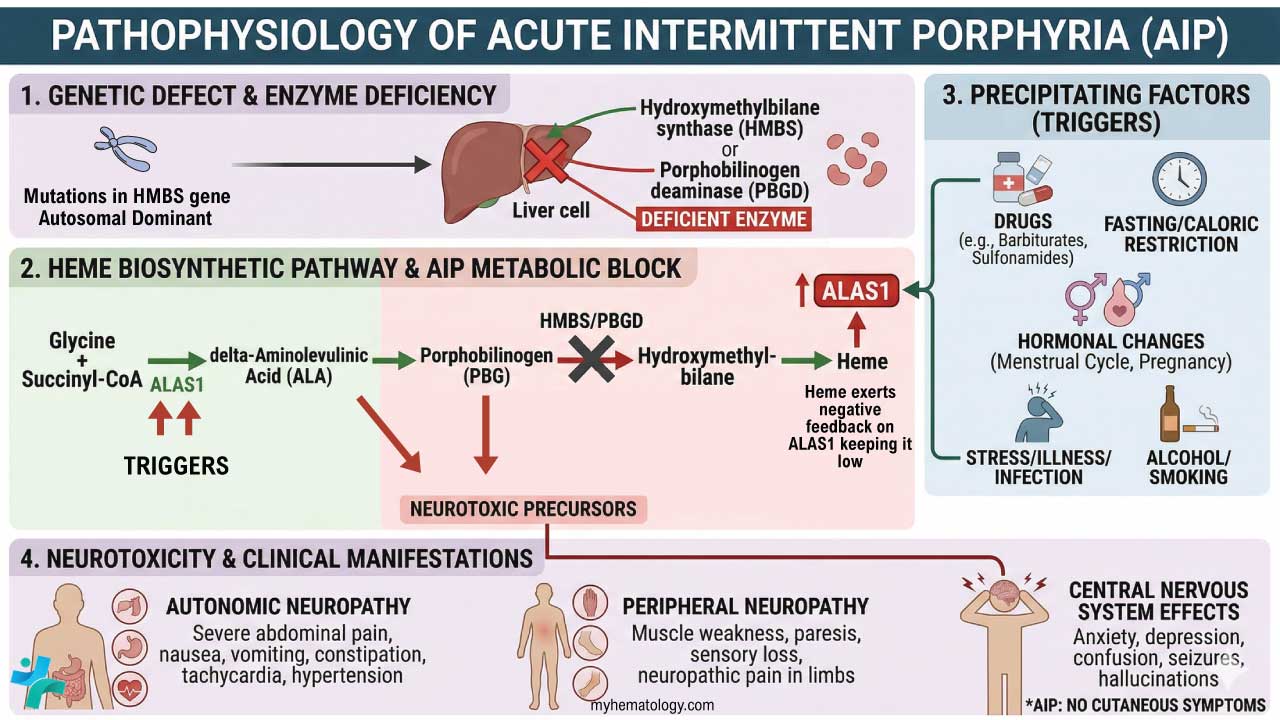
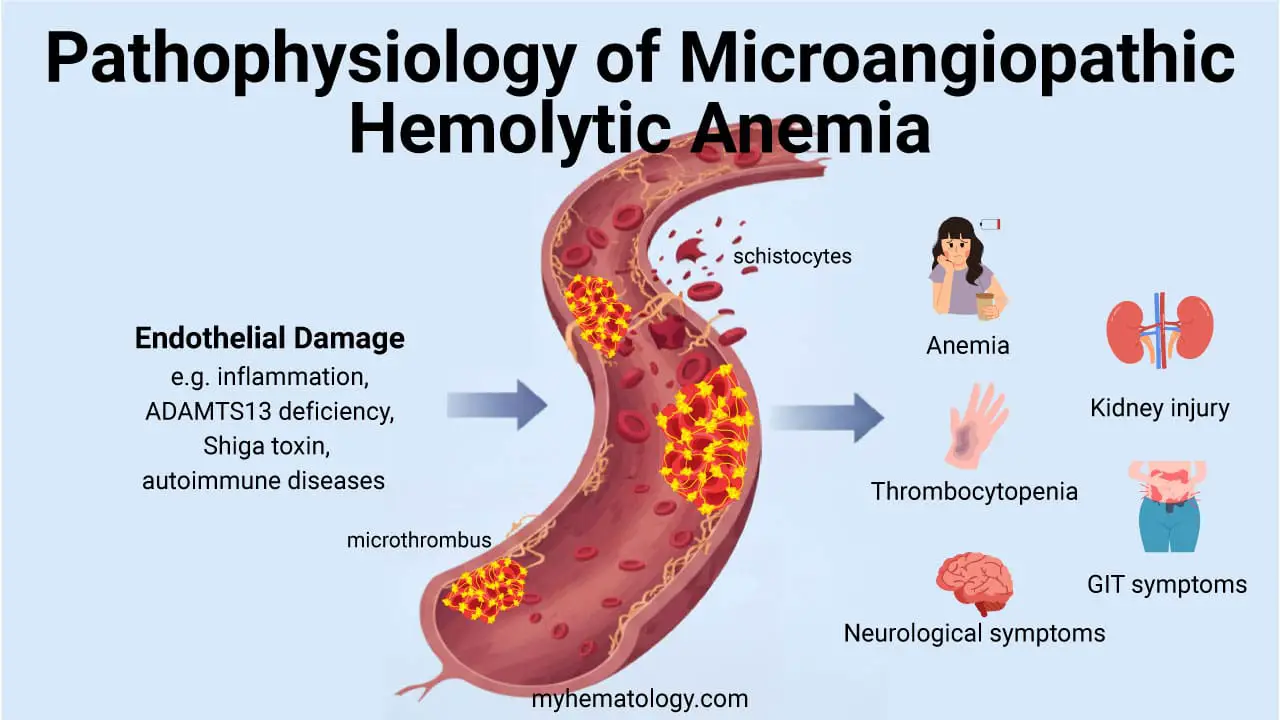
Procedure At-A-Glance
The Leishman stain is used in microscopy to differentiate types of blood cells and to detect blood parasites in peripheral blood smears.
- Prepare 0.066 M phosphate buffer at pH 6.8.
- Cover the smear with Leishman stain. Wait for 2-3 minutes.
- Add an equal volume of phosphate buffer (stain:buffer= 1:1).
- Stain for 15-20 minutes.
- Rinse with slow-running tap water.
- Air-dry or dry on low heat.
- Mount the slide with Depex and a cover slip for viewing.
Dip Slide Protocol with Coplin Jars
- Prepare the diluted stain. Let stand for 10 minutes. Fill jars.
- Fix the smear in methanol for 30 seconds.
- Dip in stain for 3 minutes.
- Transfer to buffer for 6 minutes.
- Rinse in deionized water for 10-15 seconds.
- Optional: Dip briefly in methanol for 1-2 seconds for contrast.
- Air dry, mount, and examine.
Introduction: Why the Leishman Stain Still matters
Modern automated cell counters can count blood cells in seconds, but they cannot always tell what kind of abnormal cell they are seeing. That is where the Leishman stain comes in. By coloring different parts of cells in different shades, it lets a human eye identify cell types, spot abnormal shapes, and find blood-borne parasites such as Plasmodium.
The Leishman stain is used in two broad fields:
- Hematology, to evaluate red blood cells, white blood cells, and platelets when a complete blood count flags an abnormality, when anemia or hemolysis is suspected, or when a leukemia is on the differential.
- Tropical medicine and parasitology, to identify malaria parasites, Babesia, trypanosomes, and Leishmania organisms in blood or tissue smears.
Once stained, a smear can reveal immature cells, abnormal nuclear shapes, or unusual cytoplasmic inclusions, all of which point toward conditions such as leukemia, lymphoma, or anemia [3].
If you are new to blood films, start with our companion article on Preparation of Peripheral Blood Smears. A poorly made smear cannot be rescued by even the best stain.
When Is a Peripheral Blood Smear Indicated?
Knowing when to make and stain a smear matters as much as knowing how. A peripheral smear is typically requested when:
- An automated complete blood count flags abnormal cells or platelets.
- A patient has unexplained anemia, jaundice, or a suspected hemolytic process.
- Blasts or atypical lymphocytes are reported by an analyzer.
- Malaria, babesiosis, or another blood-borne parasite is suspected.
- Schistocytes, sickle cells, or other diagnostic shapes need to be confirmed.
The smear adds a layer of human review the machine cannot fully replace.
Principle of Leishman Stain
The Leishman stain is a member of the Romanowsky family of blood stains. Its two active dye components are Azure B, a basic blue dye, and Eosin Y, an acidic orange dye [5]. Together they take advantage of one simple chemistry rule: opposite charges attract.
- Azure B is positively charged. It binds to negatively charged structures inside the cell, particularly the DNA in nuclei and the RNA in ribosomes. These structures end up shades of blue, violet, and purple.
- Eosin Y is negatively charged. It binds to positively charged proteins, especially hemoglobin in red blood cells. These appear pink to orange.
Because the two dyes work together on the same slide, every cell is colored in two contrasting tones at once. Nuclei stand out from cytoplasm, granules stand out from background, and parasites stand out from their host cells.
The dyes are dissolved in methanol, which has two advantages. First, methanol fixes the cells onto the slide as the stain hits them, so a separate fixation step is not required in the flood method. Second, methanol slowly oxidizes ("polychromes") methylene blue into a mixture of azure dyes, which is what gives a well-aged Leishman stain its rich color range.
A phosphate buffer at pH 6.8 is added during staining. The pH controls how strongly each dye binds, and even a small drift can give a smear that is "too blue" or "too pink" [2].
Method 1: Flood Slide Technique
This is the classic bench technique. It uses a small volume of stain poured directly onto each slide.
Materials for Flood Slide Method
- Leishman stain
- Phosphate buffer 0.066 M pH 6.8
- Solution A: KH2PO4 9.1 g/L
- Solution B: Na2HPO4 9.5 g/L
- Pasteur pipette
- Kim wipes
- Hair dryer (optional)
- Unstained peripheral blood smear slide
- Timer
- Cover slips (optional)
- Mounting medium (optional)
Protocol for Flood Slide Method
- Prepare 100 mL of phosphate buffer: mix 50.8 mL of Solution A with 49.2 mL of Solution B to obtain pH 6.8.
- Flood the slide with Leishman stain using a Pasteur pipette. About 3 mL covers a single smear. Wait 2–3 minutes. The methanol in the stain fixes the cells during this time.
- Add an equal volume of phosphate buffer so the stain : buffer ratio is 1:1. Gently blow with a Pasteur pipette to mix without touching the slide.
- Stain for 15–20 minutes.
- Rinse with slow-running tap water. Wipe the back and edges with a Kim wipe. Do not touch the smear surface.
- Dry the slide at room temperature or with a hair dryer on cool.
- Mount with Depex and a cover slip over the morphology zone.
- Examine under the microscope.
Method 2: Dip Slide Technique with Coplin Jars
This method is faster for batches and gives more consistent results across many slides.
Materials for Dip Slide Method
- Leishman stain
- Phosphate buffer 0.066 M pH 6.8
- Unstained peripheral blood smear slide
- 4 Coplin jars
- Deionized water
- Methanol (for fixation, and optional differentiation)
- Forceps
- Timer
- Cover slips (optional)
- Mounting medium (optional)
Protocol for Dip Slide Method
- Dilute the stain concentrate per the manufacturer's instructions. Mix and let stand for 10 minutes.
- Fill the four Coplin jars: methanol, stain, buffer (or stain–buffer mix), and deionized water.
- Fix smears in methanol for 30 seconds.
- Transfer to the stain jar for 3 minutes using forceps. Submerge fully.
- Move to the buffer jar for 6 minutes.
- Rinse in deionized water for 10–15 seconds.
- Optional differentiation: dip in methanol for 1–2 seconds to sharpen contrast. Avoid longer exposure or the stain will wash off.
- Air-dry, mount with Depex, and examine.
Quality Control and Modern Practice
A good slide is the product of good practice, not luck. Routine QC steps include:
- Check buffer pH each day before staining. Even small drifts (above 7.0 or below 6.4) shift the color balance.
- Run a normal control smear alongside patient smears to confirm consistent staining.
- Filter the stain before use to prevent fine precipitates that can mimic bacteria on the slide.
- Replace working stain on a defined schedule and store stock in a tightly sealed, light-protected bottle.
Modern hematology laboratories increasingly use digital cell-imaging analyzers, which photograph stained smears and pre-classify cells using software. These systems still rely on Romanowsky stains such as Leishman, so consistent staining matters even more, because the algorithms are sensitive to color drift. The International Council for Standardization in Haematology (ICSH) publishes the reference Romanowsky method using Azure B and Eosin Y, along with standardized morphology nomenclature [3, 5]. A modified Leishman stain that adds phenol as an accentuator has also been described and can complete staining in roughly 3–5 minutes, which is useful in emergency settings.
Interpretation: Reading a Leishman-Stained Smear
In Hematology
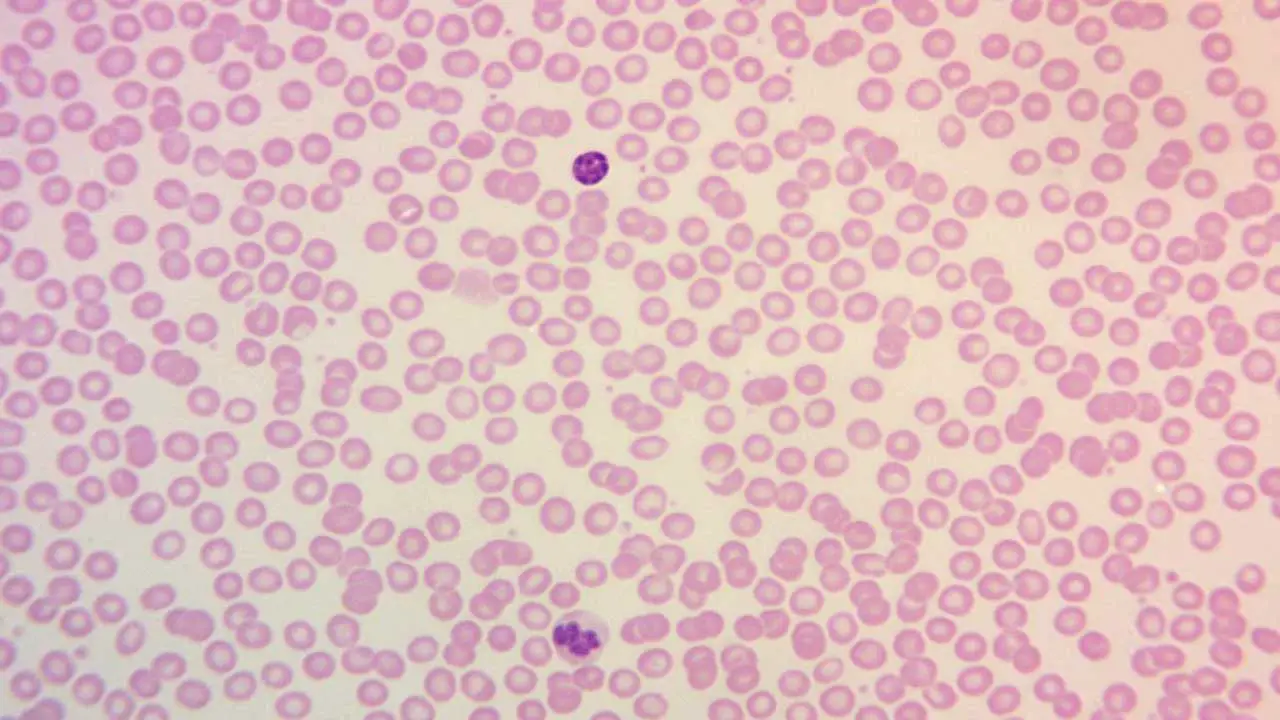
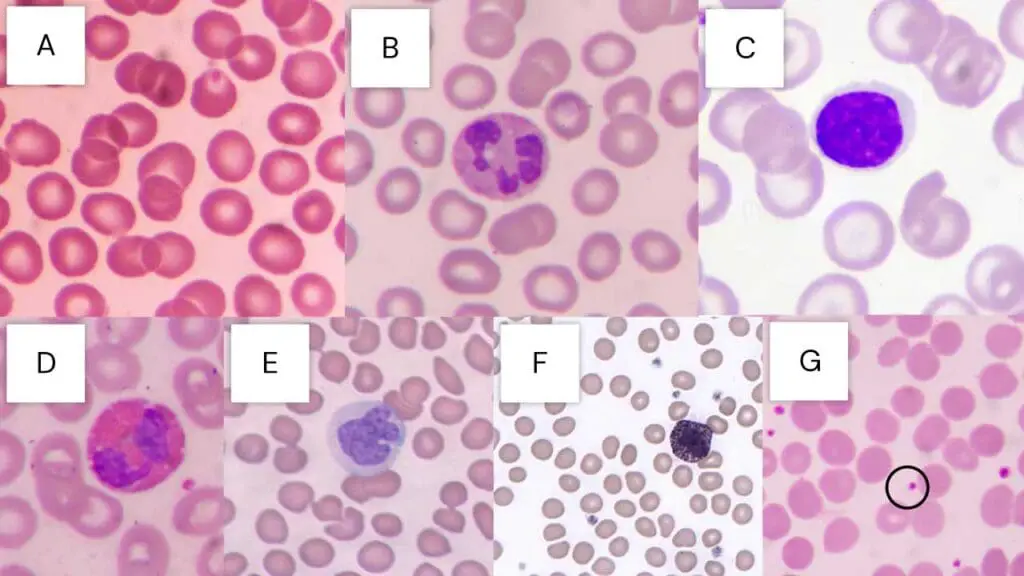
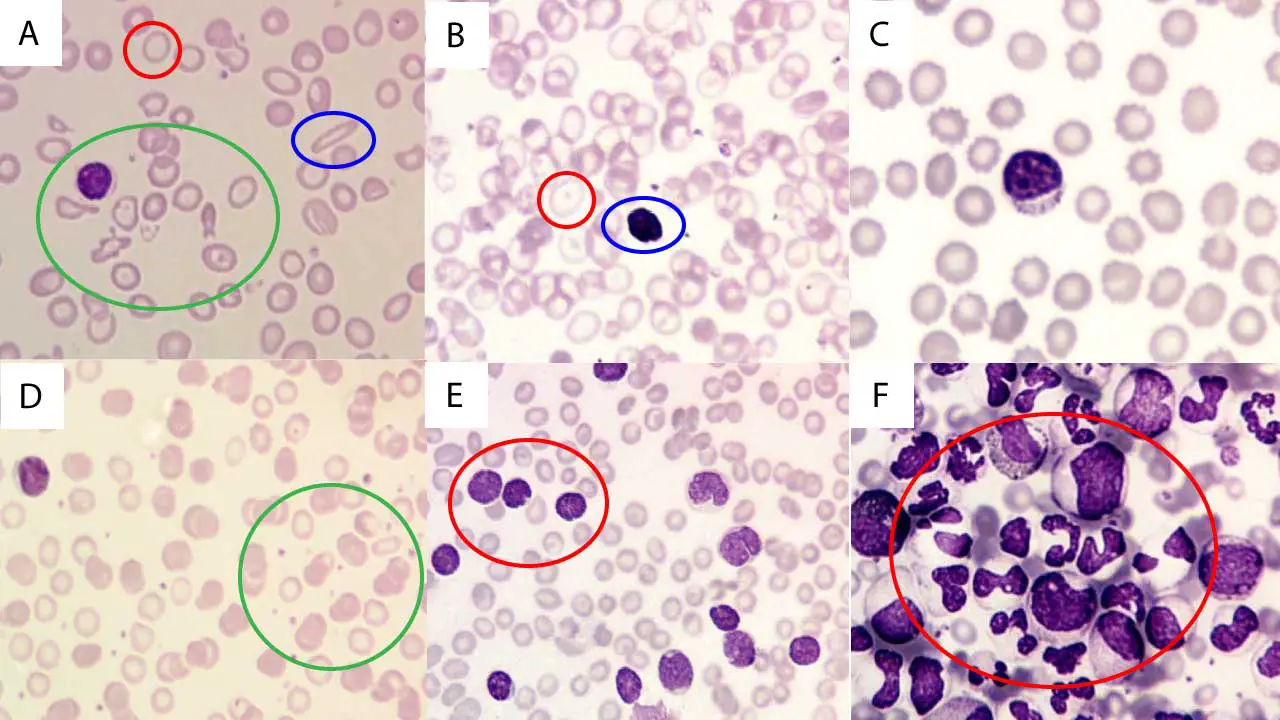
Abnormal findings like sickle cells, schistocytes, blasts, target cells and nucleated red cells are graded using ICSH-standardized terminology, which is how modern laboratories report results [3, 4].
In Parasitology
The Leishman stain is a frontline tool for diagnosing blood-borne parasitic infections.
- Malaria. The stain reveals Plasmodium parasites inside red blood cells. Parasite chromatin appears red and cytoplasm blue. A 2014 study comparing Leishman and Giemsa across 1,180 fever patients in India found near-perfect agreement (kappa = 0.995) for parasite detection and quantification, with Leishman offering clearer host-cell morphology [1].
- Leishmaniasis. Intracellular amastigotes appear as small ovoid bodies (1–5 µm) with a nucleus and a prominent kinetoplast. Although Giemsa and H&E are more common, Leishman stain works for tissue smears.
- Trypanosomiasis. Trypanosomes show a spindle shape (12–42 by 1.5–3.5 µm), red chromatin, a flagellum, and a subterminal kinetoplast. They are best identified in fresh blood or cerebrospinal fluid.
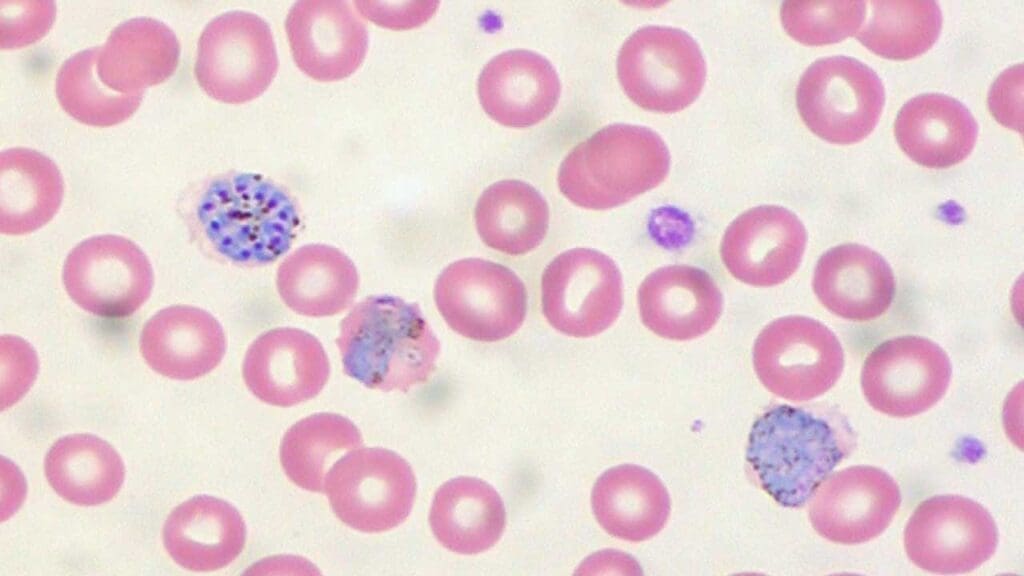
Troubleshooting
| Staining Problem | Common Causes | Solutions / Corrective Actions |
|---|---|---|
| Overall Faulty Stain |
|
|
| Too Blue (Excessive Basophilia) |
|
|
| Too Pink (Excessive Eosinophilia) |
|
|
| Unstained Neutrophil Granules |
|
|
| Pseudo-toxic Granules |
|
|
| Precipitates / Stain Deposit |
|
|
| Blue Background |
|
|
Stain deposits can mimic Gram-positive cocci and lead to false-positive infection reports. Always filter your stain.
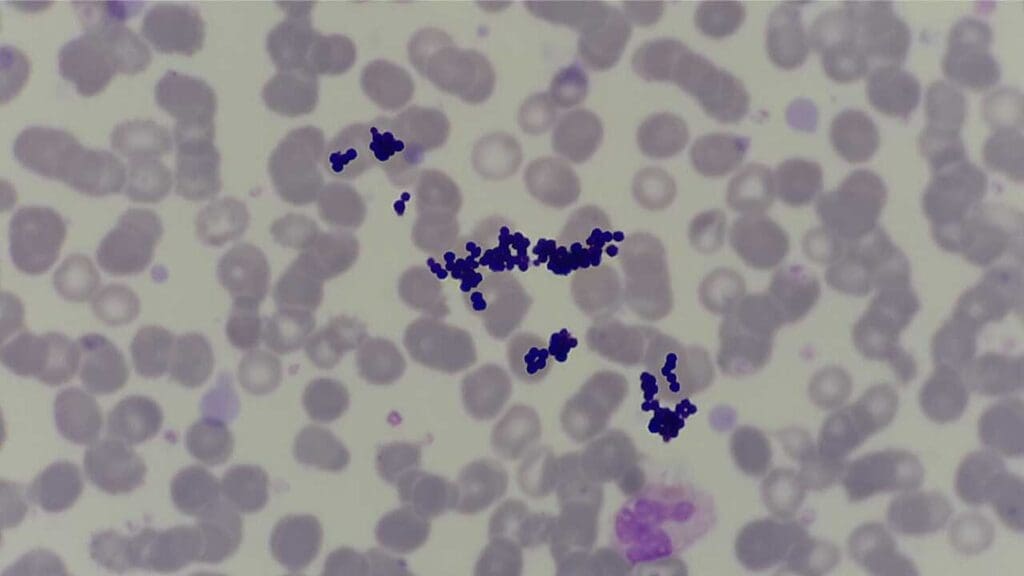
Comparison with Other Romanowsky Stains
The Leishman Advantage
For most undergraduate teaching laboratories, the Leishman stain offers the best balance of speed, cost, and diagnostic clarity, which is one reason it has remained popular for over a century.
Safety and Waste Disposal
Leishman stain contains methanol, which is flammable and toxic if inhaled, swallowed, or absorbed through the skin. Practical precautions:
- Wear gloves and eye protection. Work in a ventilated space or fume hood.
- Keep stain bottles away from open flames and sparks.
- Collect used stain and rinses in labeled flammable hazardous-waste containers. Do not pour stain waste down the drain or evaporate it.
- Follow your institution's chemical hygiene plan for disposal documentation.
Modern guidance does not recommend bench-side neutralization of methanol. Treat it as flammable hazardous waste and let your institutional waste service handle it.
Frequently Asked Questions (FAQs)
What is Leishman stain used for?
Leishman stain is a Romanowsky-type blood stain used to color a peripheral blood smear so that red cells, white cells, platelets, and blood-borne parasites can be examined under a microscope. It is commonly used to identify malaria parasites and to evaluate cells in suspected anemia, leukemia, and other blood disorders.
How long does it take to stain a slide with Leishman stain?
A standard flood-slide method takes around 18–25 minutes from start to dry slide. The dip-slide method using Coplin jars typically takes 10–15 minutes. A modified Leishman stain that uses phenol as an accentuator can complete in roughly 3–5 minutes and is sometimes used in emergency hematology
How to prepare Leishman stain from powder?
It is possible but generally not recommended. Ready-made commercial solutions are more reliable. If you must:
- Read the methanol SDS and work in a fume hood with PPE.
- Weigh 0.12 g of Leishman powder (e.g., Sigma-Aldrich 67690).
- Dissolve in 100 mL of methanol with stirring.
- Stand for 5 days at room temperature for full dye extraction.
- Filter through Whatman No. 1 paper.
- Optionally dilute with deionized water and 0.066 M phosphate buffer (pH 6.8).
- Adjust pH if needed.
- Store in a tightly sealed, light-protected, labeled bottle.
How does Leishman stain work?
t uses two oppositely charged dyes that bind opposite cell components. Azure B (basic, blue) binds DNA, RNA, and acidic structures, staining nuclei blue-violet. Eosin Y (acidic, orange) binds basic proteins like hemoglobin, staining red cells pink. The methanol solvent fixes cells and helps the dyes penetrate. The buffer pH (6.8) fine-tunes the final color balance.
What is the difference between Giemsa and Leishman staining?
Both are Romanowsky stains. Leishman is faster and self-fixing, with sharper host-cell detail in thin smears. Giemsa, applied after a separate methanol fixation, is preferred for thick smears and bone marrow morphology. A direct head-to-head study showed near-identical accuracy for malaria parasite quantification, with Leishman giving clearer red and white cell morphology for picking up alternative diagnoses [1].
Why alcohol is used in Leishman stain?
Methanol acts as solvent, fixative, and differentiator. It dissolves the dyes, fixes cells onto the slide, and can be used briefly at the end to remove excess stain and sharpen contrast. Excess methanol exposure, however, will over-destain the smear.
What are the advantages and disadvantages of Leishman stain?
Advantages: simple, affordable, fast, good differentiation of white cells, good for malaria, stable when stored well.
Disadvantages: less vivid than Giemsa, sensitive to technical variation, methanol-based safety considerations, requires training to interpret.
Can Leishman stain be used for other cell types besides blood cells?
Yes, with limits. It can give a basic view of nuclei and cytoplasm in tissue sections, cytology specimens, or cultured cells, but stains tailored to those tissues usually give better detail.
What does a normal peripheral blood smear look like with Leishman stain?
Red cells appear pink-orange with a pale center taking up about one-third of their diameter. White cells show purple-blue nuclei with cytoplasm that varies by cell type. Platelets appear as small purple granular fragments scattered between cells. Cell sizes and shapes are mostly uniform.
Is Leishman stain safe to use?
The dye itself is generally safe in normal laboratory conditions, but methanol — its solvent — is flammable and toxic if inhaled, swallowed, or absorbed through skin. Always wear gloves and eye protection, work in a ventilated area, and dispose of waste through your institution's flammable hazardous-waste stream rather than down the drain.
How do you dispose of Leishman stain waste?
Collect it in labeled flammable hazardous-waste containers and route through your institution's chemical waste service. Never pour it down the drain, into regular trash, or evaporate it.
Glossary of Related Medical Terms
- Peripheral blood smear (PBS): A thin film of blood spread on a glass slide, stained, and examined under a microscope.
- Romanowsky stain: A family of blood stains that combine a basic dye (azure derivatives of methylene blue) with an acidic dye (eosin) to color cell components in contrasting shades. Leishman, Giemsa, and Wright stains are all Romanowsky types.
- Azure B: A basic dye derived from oxidized methylene blue. It binds DNA and other acidic structures, staining nuclei blue to violet.
- Eosin Y: An acidic dye that binds basic proteins such as hemoglobin, giving red blood cells their pink-orange color.
- Polychroming: The chemical process of oxidizing methylene blue into a mixture of azure dyes, which gives Romanowsky stains their characteristic color range.
- Phosphate buffer: A solution that holds the staining mixture at a stable pH (around 6.8) so the dyes bind correctly.
- Fixation: A step that preserves cell structure, usually with methanol, before staining.
- Anisocytosis: Variation in red cell size on a smear.
- Poikilocytosis: Variation in red cell shape.
- Hypochromia: Pale-staining red cells caused by reduced hemoglobin.
- Auer rods: Needle-shaped pink-staining structures in the cytoplasm of myeloblasts. A clue to acute myeloid leukemia.
- Trophozoite, schizont, gametocyte: Different life-cycle stages of the malaria parasite seen inside red blood cells.
- Kinetoplast: A dense DNA-containing organelle near the flagellum of trypanosomes and leishmania.
- Differentiation (staining): A controlled rinse step that removes excess dye to sharpen contrast.
- Thrombocytopenia: A low platelet count.
- Erythropoiesis: The process of red blood cell production in the bone marrow.
Disclaimer: This protocol is for educational purposes only. Local laboratory standard operating procedures take precedence. It is not intended to be a substitute for informed professional medical advice, diagnosis, or treatment. Always consult a qualified healthcare professional for clinical decision-making. While the information presented here is derived from credible medical sources and is believed to be accurate and up-to-date, it is not guaranteed to be complete or error-free. See additional information.
References
- Sathpathi, S., Mohanty, A. K., Satpathi, P., Mishra, S. K., Behera, P. K., Patel, G., & Dondorp, A. M. (2014). Comparing Leishman and Giemsa staining for the assessment of peripheral blood smear preparations in a malaria-endemic region in India. Malaria journal, 13, 512. https://doi.org/10.1186/1475-2875-13-512
- Sareen, R., Kapil, M., & Gupta, G. N. (2018). Incubation and its effect on Leishman stain. Journal of laboratory physicians, 10(3), 357–361. https://doi.org/10.4103/JLP.JLP_154_17
- Palmer, L., Briggs, C., McFadden, S., Zini, G., Burthem, J., Rozenberg, G., Proytcheva, M., & Machin, S. J. (2015). ICSH recommendations for the standardization of nomenclature and grading of peripheral blood cell morphological features. International journal of laboratory hematology, 37(3), 287–303. https://doi.org/10.1111/ijlh.12327
- Zini, G., d'Onofrio, G., Erber, W. N., Lee, S. H., Nagai, Y., Basak, G. W., Lesesve, J. F., & International Council for Standardization in Hematology (ICSH) (2021). 2021 update of the 2012 ICSH Recommendations for identification, diagnostic value, and quantitation of schistocytes: Impact and revisions. International journal of laboratory hematology, 43(6), 1264–1271. https://doi.org/10.1111/ijlh.13682
- ICSH reference method for staining of blood and bone marrow films by azure B and eosin Y (Romanowsky stain). International Committee for Standardization in Haematology. (1984). British journal of haematology, 57(4), 707–710. https://doi.org/10.1111/j.1365-2141.1984.tb02949.x