Key Takeaways
Leukocytosis is an increase in white blood cell (WBC) count above the reference range.
- Significance ▾: Not a disease itself, but a marker of underlying infection, inflammation, or blood cancers.
- Causes ▾:
- Infections: Bacteria, viruses, fungi, or parasites battling your immune system.
- Inflammation: Chronic conditions like arthritis or injuries triggering immune response.
- Stress: Physical or emotional strain can temporarily elevate white blood cell count.
- Certain medications: Some drugs can stimulate white blood cell production.
- Spleen removal: This organ stores white blood cells, so its absence leads to a higher circulating count.
- Symptoms ▾: Fever, fatigue & malaise
- Diagnosis ▾: Requires a thorough history, physical exam, and complete blood count (CBC) with differential to identify the specific type of WBC elevated and potential causes.
- Treatment & Management ▾: Focuses on treating the underlying cause:
- Infections: Antibiotics, antifungals, or antivirals.
- Inflammation: Corticosteroids or immunosuppressants.
- Blood Cancers: Chemotherapy, radiation, or bone marrow transplant.
- Hyperleukocytosis Syndrome (HLS): Rare complication in some leukemias with extremely high WBC counts (>100,000/μL). Management involves:
- Hydration and urine alkalinization.
- Allopurinol or Rasburicase for uric acid control.
- Leukapheresis to remove excess WBCs (cautiously to avoid TLS).
- Hemodialysis in severe cases.
*Click ▾ for more information
Introduction
Leukocytosis can be defined as a condition where you have an increased white blood cell (WBC) count in the blood. White blood cells, also known as leukocytes, are a critical part of the body’s immune system and help fight infection and inflammation.
Normally, the white blood cell count falls within a specific range. Leukocytosis is diagnosed when the WBC count goes above the normal range.
Normal White Blood Cell (WBC) Count Ranges
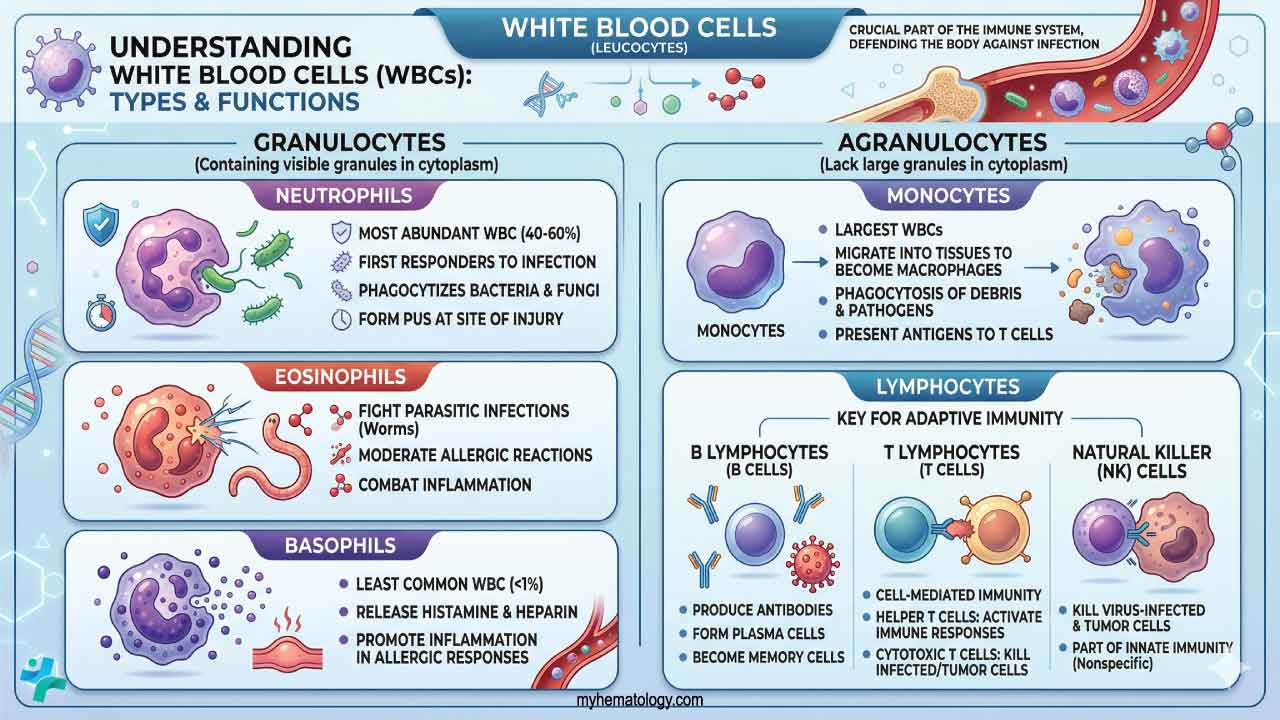
The white blood cell (WBC) count is part of a complete blood count (CBC) test, which measures various components of your blood. A CBC is frequently included in routine checkups to establish a baseline for your blood cell counts and potentially identify any underlying issues early on. Normal white blood cell (WBC) count and differential counts can vary depending on age.
| Age Group | Total WBC Count (x 109/L) | Neutrophils (%) | Lymphocytes (%) | Monocytes (%) | Eosinophils (%) | Basophils (%) |
| Newborn (0-1 day) | 9 – 34 | 30-60 | 20-60 | 3-10 | 1-6 | 0-2 |
| Infant (1 day – 1 month) | 5 – 19 | 40-70 | 20-50 | 3-10 | 1-6 | 0-2 |
| Child (1 month – 18 years) | 5 – 15 | 30-55 | 30-70 | 2-10 | 1-6 | 0-2 |
| Adult (>18 years) | 4.5 – 11 | 40-70 | 20-40 | 2-10 | 1-4 | 0-2 |
Significance of Leukocytosis
Leukocytosis, although not a specific diagnosis itself, holds significant importance as a diagnostic tool for several reasons:
- Alerts to Potential Problems: An elevated white blood cell count is a red flag that something is going on in the body. It indicates the immune system is activated, potentially fighting off an infection, inflammation, or other underlying issue.
- Guides Further Investigation: The presence of leukocytosis prompts further investigation to determine the cause. Doctors can consider the patient’s symptoms, medical history, and the specific type of leukocytosis (e.g., neutrophilia, lymphocytosis) to narrow down the possibilities. This can lead to a more efficient and targeted diagnostic approach.
- Helps Differentiate Conditions: Leukocytosis patterns can be helpful in differentiating between different conditions. For example, a high neutrophil count (neutrophilia) is more suggestive of a bacterial infection, while a high lymphocyte count (lymphocytosis) might point towards a viral infection or an autoimmune disorder.
- Monitors Treatment Response: In some cases, leukocytosis can be used to monitor how well a patient is responding to treatment. For instance, a decrease in white blood cell count after starting antibiotics for an infection might indicate the treatment is working.
Limitations to Consider
- Non-Specific: Leukocytosis can occur in various situations besides infection. Inflammation, tissue injury, and even some medications can cause a high WBC count.
- Variations: The normal white blood cell count can vary slightly depending on age, ethnicity, and even diurnal. It is lower in the morning and increases through the day.
- Severity Doesn’t Correlate: The degree of leukocytosis doesn’t necessarily reflect the severity of the underlying condition. Sometimes, severe infections might not cause a significant increase in WBCs.
- Can be Transient: Leukocytosis can sometimes be transient (short-lived) due to factors like stress, exercise, or certain medications.
- Can be Absent: Not all infections or inflammatory conditions lead to leukocytosis. Some individuals, especially those with compromised immune systems, might not experience a rise in WBC count despite an infection.
Physiological Leukocytosis
Leukocytosis can occur in various patient populations, but reference ranges for what constitutes “elevated” white blood cell (WBC) counts can differ depending on age and specific circumstances.
Newborns
- Higher Baseline WBC: Newborns naturally have higher baseline WBC counts compared to adults. This is due to their immature immune system and the stress of birth.
- Physiological Leukocytosis: In the first few days of life, newborns often experience a transient “physiological leukocytosis” with WBC counts reaching up to 30 x 109/L. This is normal and doesn’t necessarily indicate an infection.
- Leukocytosis in Newborns: However, a persistently high WBC count (above 30 x 109/L) or a rapid rise in WBC count can be a sign of infection, which needs prompt evaluation by a healthcare professional. The specific type of leukocytosis (neutrophilia, lymphocytosis, etc.) can offer clues about the potential type of infection.
Pregnant Women
- Physiological Leukocytosis: During pregnancy, women experience a physiological leukocytosis with WBC counts reaching up to 18 x 109/L. This is a normal adaptation to support the developing fetus and prepare for childbirth.
- Leukocytosis During Pregnancy: While a mild elevation in WBC count is expected, a significant or persistent increase can indicate an underlying infection, such as a urinary tract infection (UTI) or a vaginal infection. As with newborns, the specific type of leukocytosis can provide additional information.
- Considerations in Interpretation: When interpreting WBC counts in pregnant women, healthcare professionals consider factors like gestational age and potential sources of inflammation, such as labor or delivery.
Types of Leukocytosis
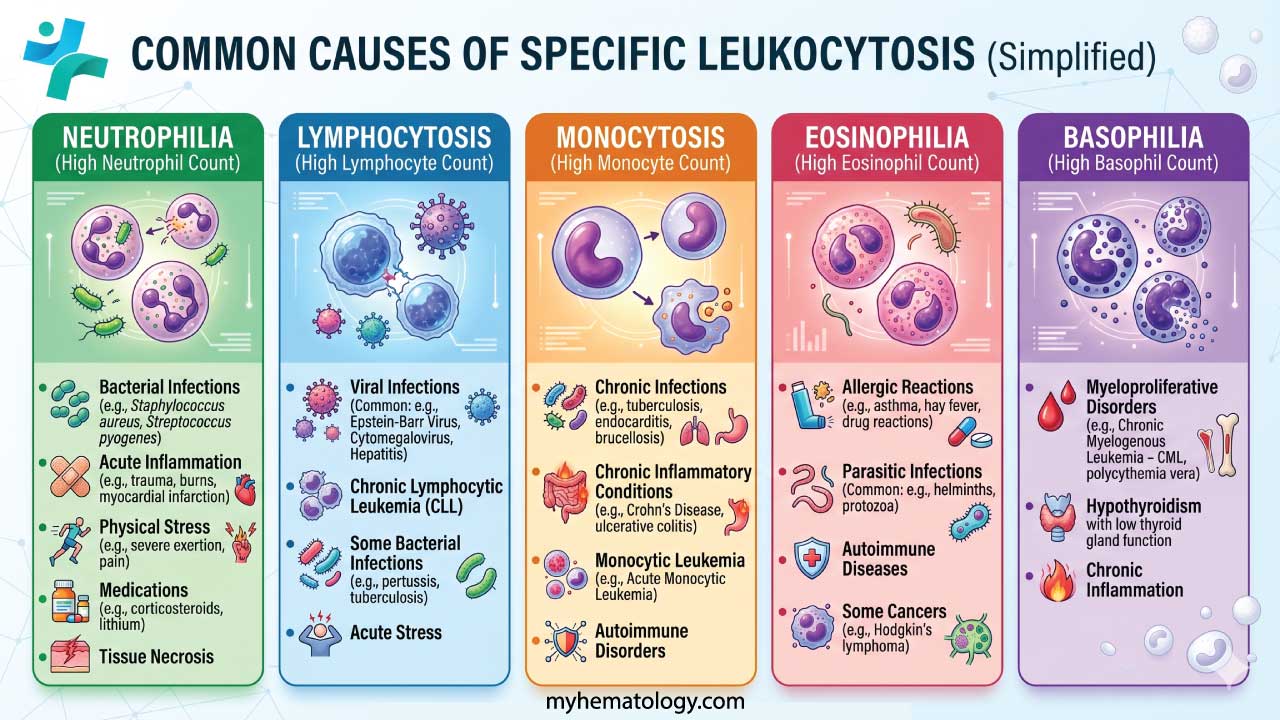
Predominant Cell Type
This classification focuses on the specific type of white blood cell that is predominantly elevated in the blood count.
- Neutrophilia: An increased number of neutrophils, the most common type of WBC. This often suggests a bacterial infection.
- Lymphocytosis: An elevated level of lymphocytes, which are involved in the immune response against viruses and other pathogens. This can be seen in viral infections, autoimmune diseases, or certain cancers.
- Monocytosis: An increase in monocytes, which are phagocytic WBCs and can indicate chronic infections, inflammatory conditions, or some cancers.
- Eosinophilia: An elevated number of eosinophils, which are involved in allergic reactions and parasite infections.
- Basophilia: The least common type of leukocytosis, characterized by an increase in basophils. This can be associated with allergic reactions, certain infections, or some bone marrow disorders.
Cell Maturity
This classification looks at the maturity of the white blood cells present in the blood.
- Shift to the Left (Leukocyte Left Shift): This indicates the presence of immature white blood cells (blasts and precursors) in the bloodstream. This can occur when the bone marrow is under stress and needs to rapidly increase WBC production, often seen in severe infections or chronic leukemia.
- Shift to the Right: A less common finding, a shift to the right indicates an increased number of mature white blood cells compared to immature ones. This is not typically associated with leukocytosis and might be seen in some vitamin B12 deficiencies.
Leukocytosis Causes
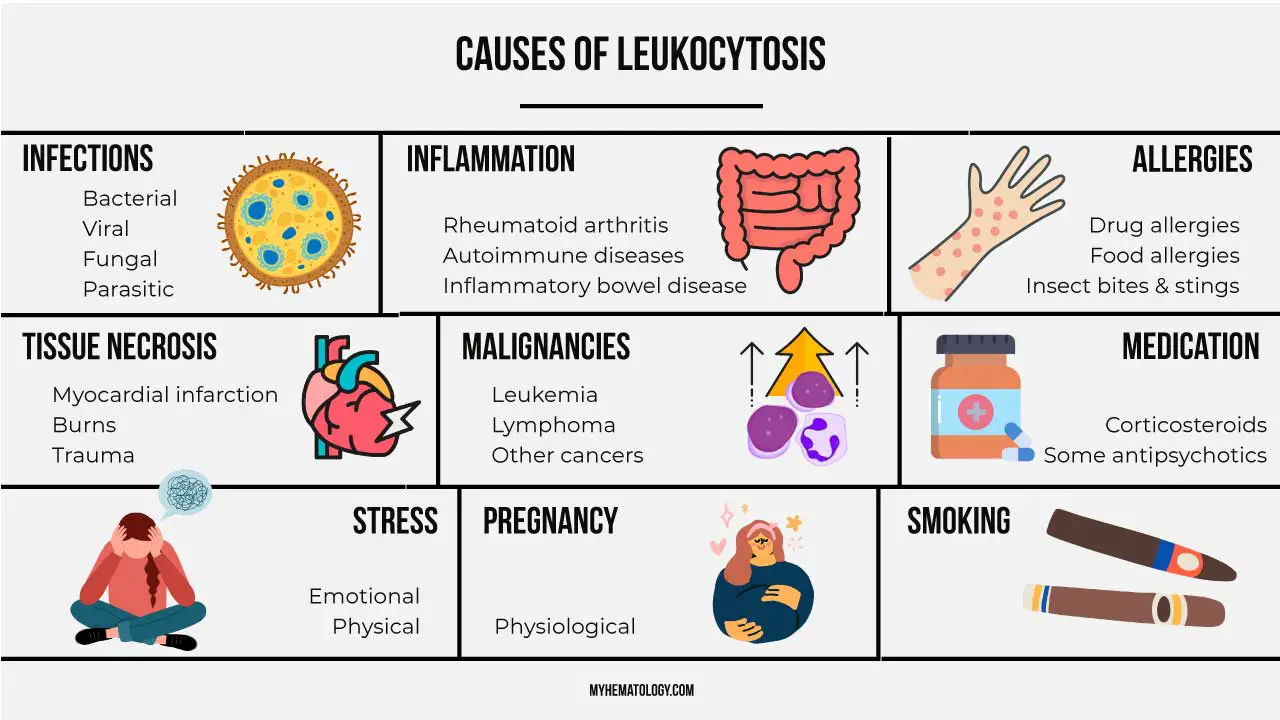
| Category | Examples |
| Infection | Bacterial infections (pneumonia, urinary tract infection) Viral infections (mononucleosis, cytomegalovirus) Parasitic infections (malaria) F ungal infections (aspergillosis) |
| Inflammation | Inflammatory bowel disease (IBD) Rheumatoid arthritis Systemic lupus erythematosus (SLE) Vasculitis |
| Autoimmune Diseases | Rheumatoid arthritis Systemic lupus erythematosus (SLE) Inflammatory bowel disease (IBD) Psoriasis |
| Tissue Injury | Burns Trauma Surgery |
| Neoplasms (Cancers) | Leukemias (acute and chronic) Lymphomas Multiple myeloma Solid tumors (in some cases) |
| Allergic Reactions | Drug allergies Food allergies Insect bites and stings Anaphylaxis |
| Stress Response | Major surgery Burns Severe emotional stress |
| Other Causes | Splenomegaly (enlarged spleen) Dehydration Smoking |
Leukocytosis Symptoms & Signs
Leukocytosis itself is often asymptomatic. However, the underlying cause of leukocytosis can manifest through various signs and symptoms.
General Symptoms
- Fever: An elevated body temperature is a common indicator of the body’s inflammatory response, potentially associated with infection.
- Fatigue: Increased metabolic demands during leukocytosis and the underlying condition can lead to tiredness and weakness.
- Malaise: A general feeling of discomfort, illness, or uneasiness can accompany leukocytosis.
Leukocytosis Symptoms Depending on the Cause
- Infection
- Localized pain, swelling, or redness at the infection site (e.g., sore throat with tonsillitis, urinary urgency with UTI).
- Cough, shortness of breath (respiratory infection).
- Diarrhea, nausea, vomiting (gastrointestinal infection).
- Inflammation
- Joint pain, swelling, and stiffness (rheumatoid arthritis).
- Skin rash (inflammatory skin conditions).
- Autoimmune Diseases
- Fatigue, weight loss, widespread pain (lupus).
- Cancer
- Unexplained weight loss, night sweats, easy bleeding or bruising (depending on the specific cancer).
Leukocytosis Physical Examination Findings
- Lymphadenopathy: Enlarged lymph nodes, which are part of the immune system and can become palpable during leukocytosis, especially when infection is the cause.
- Splenomegaly: An enlarged spleen, another immune organ, can sometimes be felt on physical examination in some cases of leukocytosis.
Approach to Leukocytosis
Points to consider when evaluating a patient with leukocytosis.
- Detailed History: A thorough history, including current symptoms, past medical history, medications, recent exposures (infectious agents, allergens), and social habits, is crucial.
- Physical Examination: A comprehensive physical exam can reveal signs suggestive of the underlying cause, such as fever, lymphadenopathy (swollen lymph nodes), rash, or organ-specific findings.
- Correlation with WBC Count and Type: The specific type of leukocytosis (neutrophilia, lymphocytosis, etc.) can offer clues about the potential cause. For example, neutrophilia is more suggestive of a bacterial infection, while lymphocytosis might point towards a viral infection.
- Other Laboratory Tests: Additional tests like blood cultures, urinalysis, chest X-ray, or other investigations might be necessary depending on the suspected cause.
Additional Considerations
- Shift to the Left: The presence of immature WBCs in the peripheral blood smear (shift to the left) can indicate a more severe infection and the body’s rapid attempt to produce more WBCs.
- Specific Leukocytosis Types: The type of leukocytosis (neutrophilia, lymphocytosis, etc.) can offer clues about the underlying cause.
Complications of Leukocytosis
Leukocytosis itself isn’t necessarily a disease, but rather an indicator of an underlying condition. However, depending on the severity and type of leukocytosis, there can be potential complications.
Vascular Occlusion
- Hyperleukocytosis: Extremely high WBC counts (>100 x 109/L) can lead to a condition called hyperleukocytosis.
- Increased Blood Viscosity: In hyperleukocytosis, the massive number of WBCs can thicken the blood, increasing its viscosity. This can impair blood flow and potentially lead to:
- Ischemic Organ Damage: Reduced blood flow to organs like the brain, lungs, kidneys, or heart can cause tissue damage and organ dysfunction.
- Stroke: If blood flow to the brain is compromised, a stroke can occur.
- Pulmonary Edema (Fluid in the Lungs): In severe cases, impaired blood flow to the lungs can lead to fluid buildup.
Disseminated Intravascular Coagulopathy (DIC)
This is a serious condition where abnormal blood clotting occurs throughout the bloodstream. Certain types of leukocytosis, particularly those associated with some cancers, can increase the risk of DIC. DIC can lead to:
- Bleeding complications
- Blockage of blood vessels by clots
Tumor Lysis Syndrome (TLS)
In certain cancers, particularly leukemias, aggressive treatment can lead to the rapid death of a large number of cancer cells. The breakdown products can overwhelm the body’s ability to handle them, leading to:
- Electrolyte imbalances (e.g., high potassium, low calcium)
- Kidney failure
- Heart rhythm disturbances
Increased Risk of Infection (Paradoxical)
While leukocytosis indicates the body’s attempt to fight infection, very high WBC counts can sometimes impair the function of certain white blood cells, making the body paradoxically more susceptible to opportunistic infections.
Organ Infiltration
In some cases of leukemias, the abnormal white blood cells can infiltrate organs like the liver, spleen, and lymph nodes, causing:
- Enlargement of these organs
- Disruption of their normal function
Laboratory Investigations for Leukocytosis
The diagnostic approach to leukocytosis requires a systematic progression from identifying the primary cell line involved to determining whether the elevation is a reactive (benign) response or a clonal (malignant) process.
Tier 1: The Initial Quantitative Assessment
The first step in any investigation of leukocytosis is a Complete Blood Count (CBC) with automated differential.
Absolute Counts vs. Percentages: Clinical decisions must be based on the absolute count of each cell type (Neutrophils, Lymphocytes, Monocytes, Eosinophils, and Basophils) rather than the percentage. For instance, a patient with a normal total WBC count may still have an absolute lymphocytosis masked by a low neutrophil count.
Total White Blood Cell (WBC) Count: Establishes the severity. While normal ranges vary, values > 11.0 x 109/L in adults typically trigger investigation. Extreme elevations (> 50.0 x 109/L) suggest either a leukemoid reaction or a myeloproliferative neoplasm.

Tier 2: Qualitative Morphological Analysis
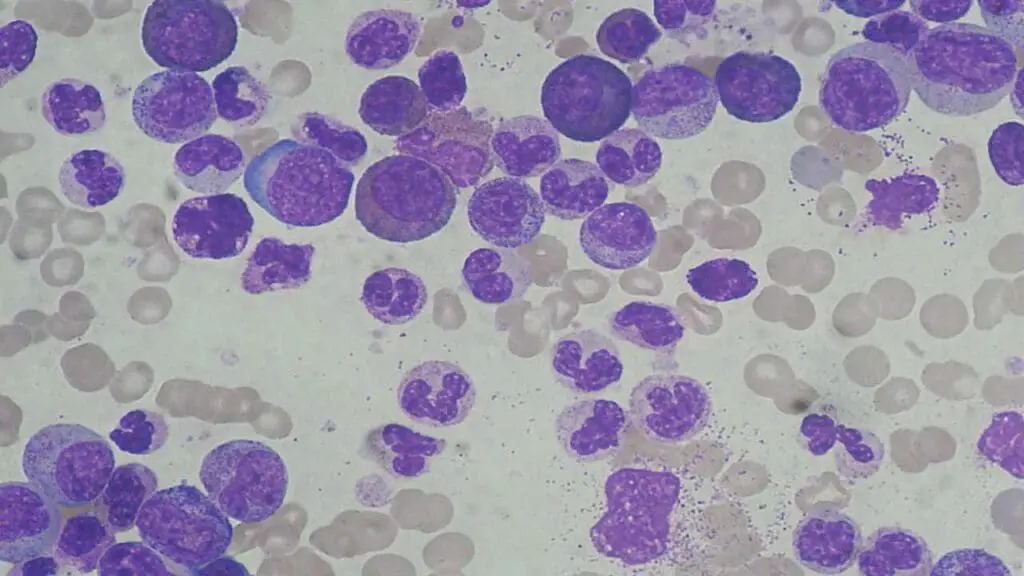
If the automated count is abnormal, a Peripheral Blood Smear (PBS) is the most high-yield manual investigation. It provides critical morphological clues:
- Left Shift: The presence of immature myeloid cells (bands, metamyelocytes, myelocytes). A “regenerative” left shift with mature cells dominating is usually reactive.
- Toxic Changes: Findings such as toxic granulation, Döhle bodies, and cytoplasmic vacuolization within neutrophils strongly point toward severe bacterial infection or inflammatory stress.
- Atypical Lymphocytes: Large, reactive lymphocytes with “scalloped” borders often indicate viral infections like Epstein-Barr Virus (EBV).
- Blast Cells: The presence of any blasts (immature precursors with high nuclear-to-cytoplasmic ratios and visible nucleoli) requires immediate hematology consultation to rule out acute leukemia.
Tier 3: Directed Clinical Investigations
Once the predominant cell type is identified, testing is directed toward the most likely etiology:
- Infectious Workup: Blood cultures, urinalysis, sputum culture, and site-specific imaging (e.g., Chest X-ray for pneumonia). Viral serologies (HIV, EBV, CMV) are indicated for lymphocytosis.
- Inflammatory Markers: Erythrocyte Sedimentation Rate (ESR) and C-Reactive Protein (CRP) help quantify the degree of systemic inflammation.
- Metabolic & Medication Review: Assessing for recent corticosteroid use (causing demargination) or physiological stressors like ketoacidosis, burns, or acute hemorrhage.
Tier 4: Advanced Hematologic Evaluation
In cases where a reactive cause cannot be found, or if the smear is suspicious for a primary bone marrow disorder, advanced testing is necessary:
- Bone Marrow Aspiration and Biopsy: The definitive investigation to assess marrow architecture, cellularity, and the presence of infiltrative diseases or fibrosis.istory, and the type of leukocytosis identified on the CBC. A targeted approach is crucial to ensure a cost-effective and efficient evaluation.
- Leukocyte Alkaline Phosphatase (LAP) Score: Historically used to differentiate Chronic Myeloid Leukemia (low LAP) from a leukemoid reaction (high LAP). While largely replaced by molecular testing, it remains a classic board exam differentiator.
- Flow Cytometry: Essential for lymphocytosis. It determines “clonality” by assessing cell surface markers (CD markers). If all elevated lymphocytes express the same markers (e.g., CD5, CD19, and CD23 in CLL), a malignancy is confirmed.
- Cytogenetics and Molecular Studies: Tests for the Philadelphia Chromosome t(9;22) or the BCR-ABL1 fusion gene are gold standards for diagnosing CML. JAK2 mutation testing is used for other myeloproliferative neoplasms.
Diagnostic Summary
| Finding on Smear | Likely Investigation Path |
| Toxic Granulations | Cultures, Procalcitonin, Imaging for infection. |
| Smudge Cells | Flow cytometry for Chronic Lymphocytic Leukemia (CLL). |
| Auer Rods | Urgent Bone Marrow Biopsy for Acute Myeloid Leukemia (AML). |
| Hypersegmented Neutrophils | Vitamin B12 and Folate levels (rule out megaloblastic changes). |
| Eosinophilia | Stool for ova/parasites, Allergy testing, or CT for occult malignancy. |
Leukocytosis Treatment and Management
Treatment of leukocytosis is rarely about “lowering the number” itself; rather, it is about identifying and addressing the physiological or pathological driver. The clinical urgency is determined by the presence of symptomatic leukostasis or the risk of Tumor Lysis Syndrome (TLS).
Management of Reactive Leukocytosis
In the vast majority of clinical encounters, leukocytosis is a secondary “reactive” phenomenon. Management is strictly etiological.
- Infectious Triggers: Administration of targeted antimicrobials. The WBC count is used here as a dynamic marker; a declining trend often validates the efficacy of the chosen antibiotic or antiviral regimen.
- Inflammatory Triggers: Use of NSAIDs, corticosteroids, or biologics for conditions like Rheumatoid Arthritis or IBD. Paradoxically, initiating high-dose corticosteroids can cause an immediate increase in the WBC count due to neutrophil demargination. Clinicians must distinguish this “steroid-jump” from a worsening infection.
- Stress-Induced: For leukocytosis following acute physical stress (seizures, myocardial infarction, or heavy exercise), management is purely observational as counts typically normalize within 24 hours.
Emergency Management: Hyperleukocytosis & Leukostasis
When the WBC count exceeds 100 x 109/L (especially in Acute Myeloid Leukemia), the blood becomes hyperviscous, leading to leukostasis, which is a medical emergency where white cells “plug” the microvasculature.
Immediate Interventions
- Hyperhydration: Aggressive intravenous fluids (e.g., 2.5 – 3 L/m² per day) to decrease blood viscosity and maintain renal perfusion.
- Cytoreduction:
- Hydroxyurea: Often used as a “holding measure” to rapidly reduce the counts while a definitive diagnosis or chemotherapy plan is finalized.
- Leukapheresis: The mechanical removal of WBCs via an apheresis machine. It is typically reserved for patients showing neurological or respiratory symptoms of leukostasis.
- TLS Prophylaxis: Massive cell turnover (spontaneous or treatment-induced) releases intracellular contents.
- Allopurinol: For low-to-intermediate risk.
- Rasburicase: For high-risk patients or those with pre-existing hyperuricemia to prevent uric acid nephropathy.
The “Leukostasis” Red Flags
If a patient has a WBC count > 100x 109/L, monitor for dyspnea (suggests pulmonary leukostasis), neurological deficits (confusion, headaches, or visual blurring) and priapism (sign of microvascular occlusion).
Management of Malignant Leukocytosis
If the investigation confirms a primary hematologic malignancy, treatment shifts to definitive oncology protocols.
- Chronic Myeloid Leukemia (CML): Targeted therapy with Tyrosine Kinase Inhibitors (TKIs) like Imatinib.
- Acute Leukemias (AML/ALL): Intensive induction chemotherapy and potentially hematopoietic stem cell transplantation.
- Chronic Lymphocytic Leukemia (CLL): Often follows a “watch and wait” approach unless the patient becomes symptomatic or the “lymphocyte doubling time” is less than 6 months.
Monitoring and Follow-Up
- Trend Analysis: A single WBC value is less informative than the trend. In a resolving infection, the “Left Shift” (immature cells) should disappear before the total WBC count fully normalizes.
- Post-Splenectomy: Patients with a history of splenectomy may maintain a “new normal” baseline of 12-15 x 109/L. Management here is education regarding the increased risk of Overwhelming Post-Splenectomy Infection (OPSI) rather than treating the count.
Frequently Asked Questions (FAQs)
How long does it take for WBC to return to normal?
The time it takes for your WBC to return to normal after leukocytosis depends on the underlying cause:
- Infections: For common bacterial infections, WBC count usually normalizes within 5 to 25 days after starting antibiotics and as the infection resolves.
- Viral infections: WBC can take longer to return to normal after viral illnesses, sometimes up to several weeks.
- Inflammation: If caused by an inflammatory condition, WBC will normalize as the inflammation is controlled with medication.
- Blood Cancers: In blood cancers, the timeframe for WBC normalization depends on the type and severity of the cancer and the treatment plan.
Why does leukocytosis occur after splenectomy?
Leukocytosis after a splenectomy is a well-documented phenomenon and is generally considered physiologic (normal response) rather than a sign of infection.
The Spleen’s Role
- A vital organ in the lymphatic system that acts as a filter for blood.
- It removes old or damaged blood cells, including some white blood cells.
- It also serves as a reservoir for white blood cells, storing them until they’re needed to fight infection.
Splenectomy and White Blood Cells
- After a splenectomy, this storage pool for white blood cells is removed. The body compensates by increasing production of white blood cells in the bone marrow to maintain adequate levels in circulation.
- This leads to a temporary elevation in circulating white blood cells, a condition known as post-splenectomy leukocytosis.
Types of Leukocytosis after Splenectomy
- Early: Immediately after surgery, there’s a transient rise in neutrophils (a type of white blood cell). This usually peaks within a day or two and gradually returns to near normal within a week.
- Long-term: A mild to moderate elevation in white blood cells, including lymphocytes and monocytes (other types of white blood cells), can persist for months or even years after a splenectomy. This is considered a normal physiological response.
What is the main cause of leukocytosis?
Leukocytosis itself doesn’t have a single main cause because it’s a response, not a specific disease. The most common triggers are infection and inflammation. Your body ramps up white blood cell production to fight off invaders or deal with inflamed tissues. There are other causes as well, but these are the most frequent culprits.
Can leukocytosis be cured?
Leukocytosis isn’t actually cured because it’s not a disease itself. It’s a sign that your body’s immune system is activated. The focus is on treating the underlying cause that’s driving the high white blood cell count.
What level of WBC is alarming?
A white blood cell count is generally considered alarming when it falls outside the normal range, which can vary slightly between labs. Adults typically have a WBC between 4.5 – 11 x 109/L. A high WBC (leukocytosis) above 11 x 109/L might indicate infection or inflammation, and very high levels (hyperleukocytosis) over 50 x 109/L require urgent medical attention. However, the specific level that’s concerning can depend on factors like age and recent surgery.
Is leukocytosis life threatening?
Leukocytosis itself isn’t usually life-threatening, but the underlying cause can be. The severity depends on the cause – a common infection likely isn’t a threat, but very high WBC or blood cancers require prompt attention. Early diagnosis and treatment are key.
Can leukocytosis turn into leukemia?
Leukocytosis itself doesn’t directly turn into leukemia. It’s a general rise in white blood cells, while leukemia is a specific cancer of the blood cells. However, leukocytosis can be a sign of leukemia, especially if the white blood cell count is very high or there are abnormal types of white blood cells present.
Can medications like steroids cause a high WBC count?
Yes, corticosteroids are a very common cause of transient leukocytosis. They induce a process called demargination, preventing neutrophils from adhering to blood vessel walls and keeping them in the circulating bloodstream. This can elevate the WBC count without any underlying infection being present.
How do you differentiate a leukemoid reaction from leukemia?
Both conditions can present with a significantly elevated WBC count. A peripheral blood smear in a leukemoid reaction typically shows toxic granulation and Döhle bodies. Furthermore, a leukocyte alkaline phosphatase (LAP) score is usually elevated in a leukemoid reaction but decreased in chronic myeloid leukemia (CML). Definitive diagnosis often requires flow cytometry or molecular testing for markers like the BCR-ABL1 fusion gene, which is absent in reactive states.
Why is the Absolute Neutrophil Count (ANC) more important than the total WBC count?
The ANC provides a precise number of the neutrophils actively circulating in the blood, whereas the total WBC count lumps all cell types together. Calculating the ANC allows clinicians to accurately assess a patient’s true risk for bacterial infections or monitor their response to immunosuppressive therapies.
Glossary of Related Medical Terms
- Absolute Neutrophil Count (ANC): A measure of the exact number of neutrophil granulocytes present in the blood, providing a more accurate assessment of immune system status than total WBC percentage.
- Demargination: The process where white blood cells detach from the endothelial lining of blood vessels and re-enter the circulating blood pool, frequently induced by stress, epinephrine, or corticosteroids.
- Flow Cytometry: A biophysical laser-based technology used to analyze the physical and chemical characteristics of cells, vital for immunophenotyping and distinguishing reactive from malignant cells.
- Hyperleukocytosis: An extreme elevation in the white blood cell count (typically > 100.0 x 109/L) that can increase blood viscosity and lead to vascular stasis and tissue hypoxia.
- Left Shift: An increased proportion of younger, immature myeloid cells (such as band cells or metamyelocytes) in the peripheral blood, typically indicating an active response to an acute bacterial infection.
- Leukemoid Reaction: A profound, non-leukemic elevation in the white blood cell count (often > 50.0 x 109/L) driven by an exaggerated physiological response to severe infection, inflammation, or tissue necrosis.
Disclaimer: This article is intended for informational purposes only and is specifically targeted towards medical students. It is not intended to be a substitute for informed professional medical advice, diagnosis, or treatment. While the information presented here is derived from credible medical sources and is believed to be accurate and up-to-date, it is not guaranteed to be complete or error-free. See additional information.
References
- Mank V, Azhar W, Brown K. Leukocytosis. [Updated 2024 Feb 4]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-.
- George TI. Malignant or benign leukocytosis. Hematology Am Soc Hematol Educ Program (2012) 2012 (1): 475–484.
- Goldberg S, Hoffman J. Clinical Hematology Made Ridiculously Simple, 1st Edition: An Incredibly Easy Way to Learn for Medical, Nursing, PA Students, and General Practitioners (MedMaster Medical Books). 2021.
- Keohane EM, Otto CN, Walenga JM. Rodak’s Hematology 6th Edition (Saunders). 2019.
- Abramson, N., & Melton, B. (2000). Leukocytosis: basics of clinical assessment. American family physician, 62(9), 2053–2060.
- Wang, W., & Hu, Z. (2022). Leukocytosis with left-shifted myeloid maturation in a peripheral blood specimen: a clue to the lymphoid blast phase of CML. Blood, 139(2), 305. https://doi.org/10.1182/blood.2021013673