Key Takeaways
Acute leukemia is a fast-progressing bone marrow cancer in which immature cells called blasts crowd out normal blood production, causing anemia, infections, and bleeding within days to weeks.
- AML and ALL differ by cell lineage: AML arises from myeloid precursors and is the most common acute leukemia in adults (~80% of adult cases), while ALL arises from lymphoid precursors and is the most common cancer in children.
- Risk Factors ▾: Major risk factors of acute leukemia include a history of Myelodysplastic Syndromes (MDS), certain genetic syndromes (like Down syndrome), prior exposure to intensive chemotherapy or radiation, and heavy tobacco use.
- Symptoms ▾: Clinical presentation of acute leukemia arises from bone marrow failure, resulting in non-specific symptoms such as fatigue (anemia), recurrent infections (neutropenia), and bleeding/bruising (thrombocytopenia), along with possible organ infiltration (ALL) and bone pain.
- Laboratory Investigations ▾: Diagnosis combines a CBC, peripheral smear, bone marrow biopsy, flow cytometry, and cytogenetic/molecular testing; under WHO 2022 and ICC 2022, defining genetic abnormalities can confirm AML even below the older 20% blast cutoff.
- Treatment ▾: Modern treatment of acute leukemia is highly individualized: "7+3" or CPX-351 induction in fit AML patients, ATRA + arsenic trioxide in APL, venetoclax + azacitidine in unfit older adults, and multi-phase chemotherapy plus immunotherapy (blinatumomab, CAR T-cell therapy) in ALL.
*Click ▾ for more information
Introduction
Acute leukemia is a fast-growing cancer that begins in the bone marrow. The marrow normally makes a steady supply of red blood cells, white blood cells, and platelets. In acute leukemia, that supply chain breaks down. Immature cells called blasts multiply quickly, fill the marrow, and crowd out healthy cells [1]. Within weeks, the body runs short of oxygen-carrying red cells, infection-fighting neutrophils, and clot-forming platelets.
Two main forms exist. Acute myeloid leukemia (AML) starts in myeloid precursor cells, which normally become red cells, platelets, and most white cells. Acute lymphoblastic leukemia (ALL) starts in lymphoid precursor cells, which become B and T lymphocytes [1,5]. AML is the most common acute leukemia in adults, accounting for about 80% of adult cases; ALL is the most common cancer in children.
The "acute" label matters. Chronic leukemias progress over years. Acute leukemia presents in days to weeks, and treatment is urgent. The good news: outcomes have improved sharply over the last two decades, especially in childhood ALL and in acute promyelocytic leukemia (APL) [3,5].
Pathophysiology of Acute Leukemia
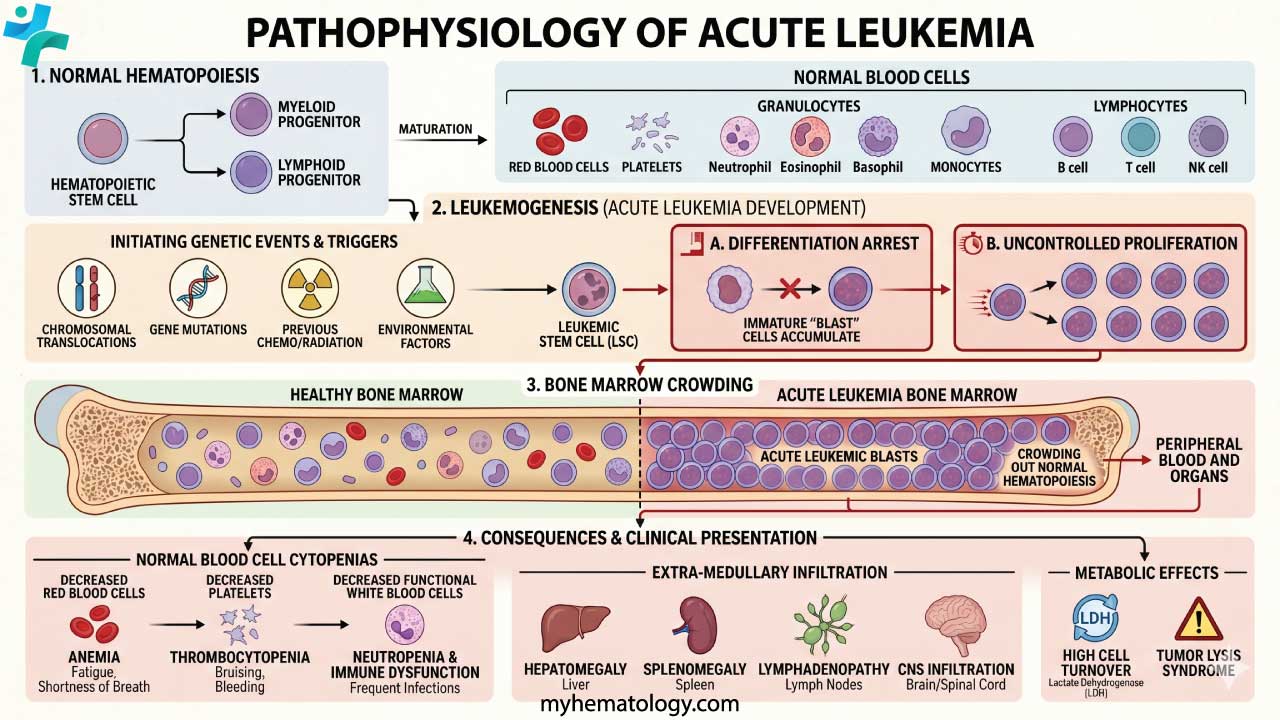
Both AML and ALL begin with acquired mutations in a single hematopoietic stem or progenitor cell. Two broad mutation types usually need to combine before frank leukemia develops:
- Class I mutations push the cell to grow and survive (examples: FLT3, KIT, RAS).
- Class II mutations stop the cell from maturing (examples: RUNX1, CEBPA, PML-RARA).
This "two-hit" pattern produces a clone of immature blasts that cannot mature normally. Over time, the clone takes over the marrow.
Acute Myeloid Leukemia (AML) Pathogenesis
AML is genetically diverse. Recurrent translocations define some subsets with distinct prognoses, including t(8;21), inv(16), and t(15;17) — the last of which defines APL. Mutations in NPM1 (the most common in AML), FLT3, and CEBPA drive risk stratification and treatment choice. FLT3-internal tandem duplications (FLT3-ITD), in particular, predict higher relapse risk [3].
Acute Lymphoblastic Leukemia (ALL) Pathogenesis
ALL is divided by the lymphoid lineage involved: B-ALL (about 80%) and T-ALL (about 20%). Key abnormalities include t(9;22), which produces the BCR::ABL1 fusion gene (the Philadelphia chromosome (Ph+)) and is most common in adult B-ALL. t(12;21) is favorable and most common in childhood B-ALL. KMT2A (formerly MLL) rearrangements vary in prognosis but tend to be aggressive in infants. A more recently recognized high-risk subgroup, Ph-like ALL, mimics the gene-expression profile of Ph+ ALL but is driven by other kinase-activating lesions such as CRLF2 overexpression [1].
Risk Factors
Most cases of acute leukemia have no clearly identifiable cause. Several factors do raise risk, though, and they fall into four groups.
Prior medical treatments and conditions
- Previous chemotherapy or radiation. Alkylating agents and topoisomerase II inhibitors used to treat earlier cancers can damage marrow DNA and lead to therapy-related AML or ALL years later.
- Pre-leukemic disorders. Myelodysplastic syndromes (MDS) frequently transform into AML. Myeloproliferative neoplasms such as polycythemia vera and essential thrombocythemia carry a smaller but real risk.
- Long-term immunosuppression. Solid organ transplant recipients have a slightly higher risk of certain leukemias.
Genetic syndromes and inherited factors
- Down syndrome (trisomy 21) is the most common syndrome linked to childhood ALL and AML.
- Rarer inherited disorders also raise risk: Fanconi anemia, Bloom syndrome, ataxia-telangiectasia, Li-Fraumeni syndrome, and Shwachman-Diamond syndrome.
- Most acute leukemia is not inherited. Having a first-degree relative with the disease — particularly an identical twin diagnosed in early childhood — modestly raises risk.
Environmental and lifestyle exposures
- Smoking is the only well-established lifestyle risk factor for AML. Benzene and other carcinogens in tobacco smoke damage marrow DNA.
- Industrial benzene exposure (in petrochemical and rubber industries) has long been linked to AML.
- High-dose ionizing radiation, such as from atomic bomb exposure or major nuclear accidents, raises both AML and ALL risk. Diagnostic-level radiation has not been clearly shown to do so.
Demographic factors
The age pattern is bimodal. ALL peaks in young children (under 5) and again in adults over 50. AML is mainly a disease of older adults, with a median age at diagnosis around 65–70 years. Most acute leukemias are slightly more common in males. ALL is somewhat more common in white than in Black populations.
Sign and Symptoms
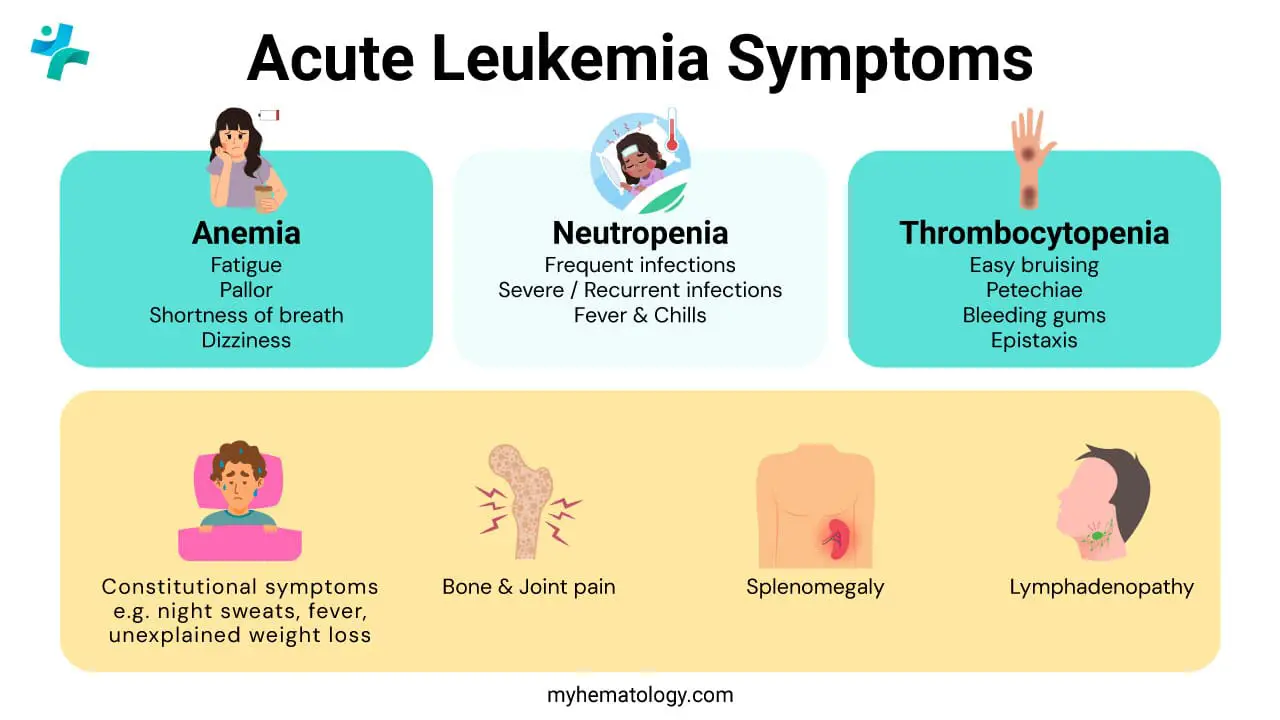
Symptoms appear quickly (typically over days to a few weeks) because blasts crowd out healthy blood cell production. The clinical picture is essentially the picture of bone marrow failure.
Symptoms from anemia (low red cells)
Fewer red cells means less oxygen delivery. Patients notice profound fatigue, pallor, shortness of breath on exertion, and sometimes dizziness or headaches.
Symptoms from thrombocytopenia (low platelets)
Without enough platelets, blood does not clot properly. Watch for easy bruising, petechiae (tiny red or purple skin spots, often on the lower legs), nosebleeds, bleeding gums, and heavy menstrual bleeding (menorrhagia).
Symptoms from neutropenia (low functional neutrophils)
The total white cell count can be low, normal, or sky-high — but the functional infection-fighting cells are usually depleted. The result: frequent or severe infections and fevers, sometimes from the leukemia itself rather than infection.
Other symptoms
- Bone or joint pain from marrow expansion, especially in children with ALL.
- Unexplained weight loss, night sweats, and fevers (the so-called B-symptoms).
- Lymphadenopathy — painless swollen lymph nodes in neck, armpit, or groin (more typical of ALL).
- Hepatosplenomegaly — an enlarged liver or spleen, causing abdominal fullness.
- CNS involvement (rare but serious): severe headaches, blurred vision, vomiting, or seizures.
- Leukostasis is a medical emergency in which a very high blast count thickens the blood, causing breathing problems and neurological symptoms.
Laboratory Investigations
Diagnosis rests on integrating several tests. Each one answers a different question.
Complete blood count (CBC)
The CBC usually shows anemia, thrombocytopenia, and either low or very high white cell counts. Functional neutrophils are typically reduced regardless of the total WBC.
Peripheral blood smear (PBS)
The critical finding in the blood smear is circulating blasts (immature cells that normally stay inside the marrow). Their presence in peripheral blood strongly suggests acute leukemia.
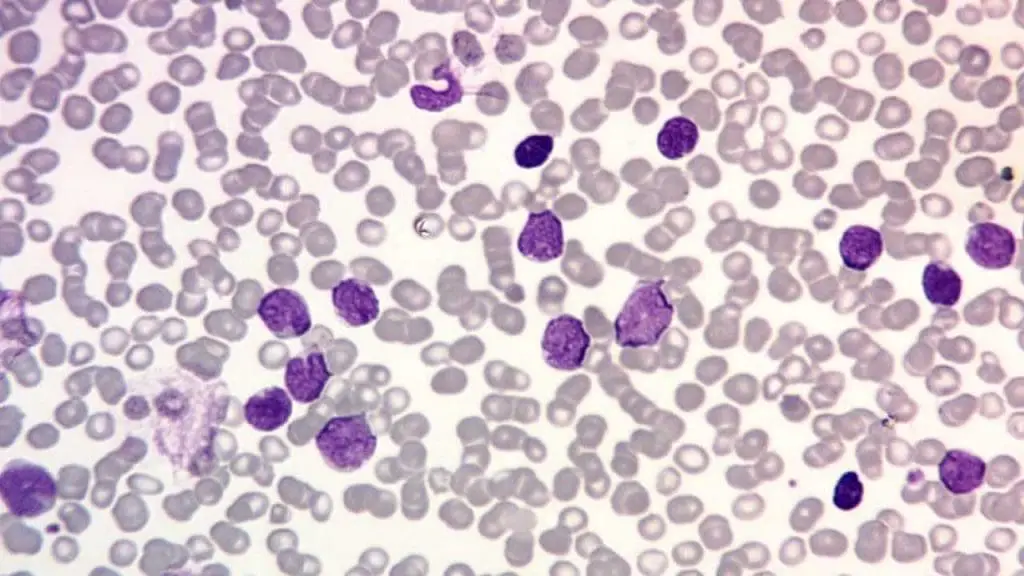
Bone marrow aspiration and biopsy
This is the definitive test. Historically, ≥20% blasts in marrow or blood confirmed acute leukemia under the WHO 2016 criteria. The WHO 2022 (5th edition) classification has changed this: AML can now be diagnosed below 20% blasts when certain defining genetic abnormalities are present (for example, PML::RARA, RUNX1::RUNX1T1, CBFB::MYH11, NPM1-mutated AML) [1].
The parallel International Consensus Classification (ICC) 2022 uses a 10% blast threshold for AML with recurrent genetic abnormalities, with a few exceptions including BCR::ABL1, where 20% is still required [2]. Both shifts reflect evidence that genetics, not blast percentage alone, drives outcome.
Immunophenotyping (flow cytometry)
This is the gold standard for assigning lineage:
- B-ALL typically expresses CD19, CD22, and CD10.
- T-ALL typically expresses CD3 and CD7.
- AML typically expresses CD13, CD33, CD117, and intracellular myeloperoxidase (MPO).
Flow cytometry is also used to track measurable residual disease (MRD) over time.
Cytogenetic and molecular analysis
Karyotyping, fluorescence in situ hybridization (FISH), and next-generation sequencing (NGS) detect chromosomal and molecular changes that drive risk stratification. In AML, the European LeukemiaNet (ELN) 2022 framework integrates these results into Favorable, Intermediate, and Adverse risk groups, which guide whether a patient should proceed to transplant in first remission [3].
The ELN 2022 classification was developed using patients receiving intensive chemotherapy, and recent analyses show it stratifies less reliably in older patients treated with venetoclax-based regimens [3,4].
NGS guides treatment
Rapid molecular profiling using NGS is now practically mandatory at diagnosis, as identifying mutations like FLT3 or IDH1/2 within a few days immediately dictates the choice of targeted drugs added to the first cycle of induction therapy.
Cytogenetics & Molecular Risk
Monitoring with MRD
After remission, doctors track MRD using flow cytometry, RT-PCR for fusion transcripts, or NGS, with sensitivities reaching 1 in 10,000 to 1 in 1,000,000 cells. MRD-negativity strongly predicts longer remission. MRD-positivity often triggers a change in therapy, including transplant [3,7].
MRD tracking differs by lineage. In ALL, MRD is typically tracked via flow cytometry or NGS (specifically tracking Ig/TCR gene rearrangements). In AML, flow cytometry or PCR for specific mutations (like NPM1or PML::RARA) is the standard; using NGS for AML MRD requires high caution due to 'clonal hematopoiesis of indeterminate potential' (CHIP), background mutations that can cause false positives [11].
Treatment and Management Approach
Treatment goals are remission first, then long-term cure. Modern care is highly stratified by patient fitness, age, and genetics.
AML treatment
1. Induction chemotherapy. The standard for fit patients remains the "7+3" regimen — cytarabine for 7 days plus an anthracycline (daunorubicin or idarubicin) for 3 days [3].
- APL is a separate emergency and is not treated with 7+3. The combination of all-trans retinoic acid (ATRA) and arsenic trioxide cures more than 90% of patients [5].
- CPX-351 (liposomal cytarabine and daunorubicin) is a standard option for fit older adults with therapy-related AML or AML with myelodysplasia-related changes [8].
2. Consolidation. This phase aims to eliminate residual disease. Options include high-dose cytarabine (HiDAC) or allogeneic hematopoietic stem cell transplantation (HSCT), which adds a "graft-versus-leukemia" effect [3].
3. Targeted therapies are now woven into AML care:
- FLT3 inhibitors — midostaurin or quizartinib (added to frontline induction [9]) and gilteritinib (relapsed/refractory) for FLT3-mutated AML.
- IDH1/IDH2 inhibitors — ivosidenib (which can also be combined with azacitidine for unfit frontline patients) and enasidenib for the corresponding mutations.
- Menin inhibitors — revumenib was recently approved for relapsed/refractory AML with KMT2A translocations or NPM1mutations, representing a major breakthrough in targeted therapy [12].
- Venetoclax + azacitidine is now the standard for older or unfit patients, improving survival over hypomethylating agents alone [4].
4. Maintenance therapy. Historically, AML had no standard maintenance phase unless a patient went to transplant. Today, oral azacitidine (CC-486) is the standard of care for maintenance in patients who achieve remission after intensive chemotherapy but are not candidates for a stem cell transplant [14].
ALL treatment
ALL therapy runs longer than AML therapy (typically 2 to 3 years) and uses several phases.
1. Induction, consolidation, and maintenance. Multi-agent chemotherapy forms the backbone (vincristine, corticosteroids, L-asparaginase, sometimes anthracyclines). Notably, Adolescent and Young Adult (AYA) patients (typically up to age 39) are now standardly treated with 'pediatric-inspired' intensive regimens, which have significantly improved survival rates compared to older adult protocols [13].
2. CNS prophylaxis. Because ALL frequently spreads to the central nervous system, intrathecal chemotherapy (commonly methotrexate) is mandatory.
3. Targeted therapy for Ph+ ALL. Adding tyrosine kinase inhibitors (TKIs) such as imatinib, dasatinib, or ponatinib has transformed outcomes. Recently, clinical trials have shown that combining a TKI with blinatumomab allows for highly effective, chemotherapy-free or chemotherapy-sparing regimens, drastically reducing toxicity while maintaining deep molecular responses [10].
4. Immunotherapies and novel agents.
- Tisagenlecleucel (CD19-directed CAR T-cell therapy) is approved for pediatric and young-adult relapsed/refractory B-ALL [6].
- Brexucabtagene autoleucel is approved for adult relapsed/refractory B-ALL.
- Blinatumomab (a CD19/CD3 bispecific T-cell engager) was originally used in MRD-positive or relapsed B-ALL; the ECOG-ACRIN E1910 trial has now extended its role into frontline consolidation, where it improves overall survival in MRD-negative B-ALL adults [7].
- Inotuzumab ozogamicin is an anti-CD22 antibody-drug conjugate used in relapsed/refractory B-ALL.
Side-by-side treatment overview
Prognosis at a glance
Numbers vary by subtype, age, and biology, but broad ranges help orient the reader:
- Childhood ALL: 5-year overall survival is now around 90% in high-income countries.
- Adult ALL: 5-year survival is roughly 40–50%, improving in Ph+ disease with TKIs.
- APL: cure rates exceed 90% with ATRA and arsenic trioxide [5].
- AML overall: 5-year survival is about 30%, sharply lower in adults over 65 and substantially better in favorable-risk subsets [3].
Supportive Care
Supportive care runs in parallel with definitive treatment. It covers transfusions for anemia and thrombocytopenia, prompt antibiotics for febrile neutropenia, antifungal and antiviral prophylaxis, tumor lysis syndrome prevention, nutritional support, and pain management.
The emotional load matters too. An acute leukemia diagnosis is sudden, and treatment can stretch over months to years. For patients and caregivers, access to social work, psychology, and peer support is part of standard care. Asking the team early for a written fever-and-symptom plan helps caregivers know exactly when to call the hospital.
Conclusion and Future Directions
Acute leukemia remains a serious illness, but the trajectory is hopeful. Updated WHO 2022 and ICC 2022 classifications integrate genetics into diagnosis [1,2]. ELN 2022 risk stratification refines treatment choice in AML, while newer frameworks are emerging for older patients on lower-intensity therapy [3]. Targeted agents, BiTEs, and CAR T-cell therapy are moving earlier in the treatment pathway [6,7].
Frequently Asked Questions (FAQs)
What is the primary difference between AML and ALL based on cell lineage?
AML originates from myeloid precursors that normally become red cells, platelets, and most white cells. ALL originates from lymphoid precursors that become B and T lymphocytes. This lineage difference shapes the immunophenotyping markers used at diagnosis and the chemotherapy backbones chosen for each disease.
What is measurable residual disease (MRD), and why is it important?
MRD is the small population of leukemia cells that may remain after a patient achieves morphologic complete remission. It is detected by flow cytometry, PCR, or next-generation sequencing at sensitivities of 1 in 10,000 to 1 in 1,000,000 cells. MRD-negative patients have markedly better outcomes, and MRD status often determines whether further therapy or a stem cell transplant is needed.
When is allogeneic stem cell transplantation indicated?
Allogeneic HSCT is generally offered to AML patients in the intermediate- or adverse-risk groups (per ELN 2022) during first remission, and to ALL patients with high-risk features or persistent MRD. It is also used for patients who have relapsed. It requires a suitable donor and a patient fit enough to tolerate it.
Doesn't BCR-ABL translocation cause Chronic Myeloid Leukemia (CML)?
Yes. The BCR::ABL1 fusion gene (Philadelphia chromosome) is the defining lesion of chronic myeloid leukemia (CML). The same translocation can also drive a high-risk subset of acute lymphoblastic leukemia known as Ph+ ALL, which makes up about 25–30% of adult B-ALL and 3–5% of pediatric B-ALL. CML usually involves a longer p210 fusion protein arising in a multipotent stem cell, while Ph+ ALL is more often associated with the shorter, more active p190 protein in a lymphoid-committed progenitor. Both respond to TKIs, but Ph+ ALL still requires intensive chemotherapy alongside.
Is acute leukemia curable?
Many cases are. Childhood ALL has 5-year survival above 85–90% in high-income countries. APL, once one of the most lethal leukemias, is now cured in over 90% of patients with ATRA plus arsenic trioxide [5]. Adult AML is harder, but venetoclax-based regimens, FLT3 and IDH inhibitors, and improved transplant outcomes are steadily raising the bar [3,4].
What can a caregiver do during treatment?
Caregivers help with infection prevention (hand hygiene, fever monitoring, avoiding crowded spaces during neutropenia), nutrition, transport to clinic visits, medication tracking, and emotional support. ALL therapy can run 2–3 years and AML often involves long inpatient stays. Asking the medical team for a written symptom-and-fever action plan, and using hospital social work and counseling services early, makes the marathon more manageable.
Glossary of Related Medical Terms
- Blast — an immature blood-forming cell. In acute leukemia, blasts pile up because they fail to mature.
- Bone marrow — the spongy tissue inside large bones where blood cells are made.
- Hematopoiesis — the process of making new blood cells.
- Pancytopenia — a drop in all three main blood cell lines (red cells, white cells, platelets) at the same time.
- Anemia — too few red blood cells or hemoglobin, leading to fatigue and pallor.
- Thrombocytopenia — too few platelets, which raises bleeding and bruising risk.
- Neutropenia — too few neutrophils (the main bacteria-fighting white blood cells), which raises infection risk.
- Petechiae — tiny pinpoint red or purple spots from small skin-vessel bleeds.
- Cytogenetics — the study of chromosomes; used to detect abnormalities like translocations.
- Immunophenotyping — labeling cell-surface markers (using flow cytometry) to identify whether a leukemia is myeloid or lymphoid.
- MRD (measurable / minimal residual disease) — small numbers of leukemia cells that remain after treatment, detected by sensitive techniques even when standard tests look clear.
- ELN (European LeukemiaNet) — the expert group that publishes widely used AML diagnosis and risk-stratification guidelines.
- Philadelphia chromosome (Ph+) — an abnormal chromosome made by a swap between chromosomes 9 and 22, producing the BCR::ABL1 fusion gene.
- TKI (tyrosine kinase inhibitor) — a targeted drug (e.g., imatinib) that blocks the abnormal BCR-ABL1 protein.
- HSCT (hematopoietic stem cell transplantation) — replacing the patient's bone marrow with healthy stem cells, usually from a donor (allogeneic).
- APL (acute promyelocytic leukemia) — a distinct AML subtype driven by t(15;17), curable with ATRA and arsenic trioxide.
- Induction / consolidation / maintenance — the three phases of leukemia chemotherapy: knock the disease down, mop up the rest, then keep it from returning.
Disclaimer: This article is intended for educational and informational purposes only. It is not intended to be a substitute for informed professional medical advice, diagnosis, or treatment. While the information presented here is derived from credible medical sources and is believed to be accurate and up-to-date, it is not guaranteed to be complete or error-free. See additional information.
References
- Khoury, J.D., Solary, E., Abla, O. et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia 36, 1703–1719 (2022). https://doi.org/10.1038/s41375-022-01613-1
- Arber, D. A., Orazi, A., Hasserjian, R. P., Borowitz, M. J., Calvo, K. R., Kvasnicka, H. M., Wang, S. A., Bagg, A., Barbui, T., Branford, S., Bueso-Ramos, C. E., Cortes, J. E., Dal Cin, P., DiNardo, C. D., Dombret, H., Duncavage, E. J., Ebert, B. L., Estey, E. H., Facchetti, F., Foucar, K., … Tefferi, A. (2022). International Consensus Classification of Myeloid Neoplasms and Acute Leukemias: integrating morphologic, clinical, and genomic data. Blood, 140(11), 1200–1228. https://doi.org/10.1182/blood.2022015850
- Döhner, H., Wei, A. H., Appelbaum, F. R., Craddock, C., DiNardo, C. D., Dombret, H., Ebert, B. L., Fenaux, P., Godley, L. A., Hasserjian, R. P., Larson, R. A., Levine, R. L., Miyazaki, Y., Niederwieser, D., Ossenkoppele, G., Röllig, C., Sierra, J., Stein, E. M., Tallman, M. S., Tien, H. F., … Löwenberg, B. (2022). Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood, 140(12), 1345–1377. https://doi.org/10.1182/blood.2022016867
- DiNardo, C. D., Jonas, B. A., Pullarkat, V., Thirman, M. J., Garcia, J. S., Wei, A. H., Konopleva, M., Döhner, H., Letai, A., Fenaux, P., Koller, E., Havelange, V., Leber, B., Esteve, J., Wang, J., Pejsa, V., Hájek, R., Porkka, K., Illés, Á., Lavie, D., … Pratz, K. W. (2020). Azacitidine and Venetoclax in Previously Untreated Acute Myeloid Leukemia. The New England journal of medicine, 383(7), 617–629. https://doi.org/10.1056/NEJMoa2012971
- Lo-Coco, F., Avvisati, G., Vignetti, M., Thiede, C., Orlando, S. M., Iacobelli, S., Ferrara, F., Fazi, P., Cicconi, L., Di Bona, E., Specchia, G., Sica, S., Divona, M., Levis, A., Fiedler, W., Cerqui, E., Breccia, M., Fioritoni, G., Salih, H. R., Cazzola, M., … Study Alliance Leukemia (2013). Retinoic acid and arsenic trioxide for acute promyelocytic leukemia. The New England journal of medicine, 369(2), 111–121. https://doi.org/10.1056/NEJMoa1300874
- Maude, S. L., Laetsch, T. W., Buechner, J., Rives, S., Boyer, M., Bittencourt, H., Bader, P., Verneris, M. R., Stefanski, H. E., Myers, G. D., Qayed, M., De Moerloose, B., Hiramatsu, H., Schlis, K., Davis, K. L., Martin, P. L., Nemecek, E. R., Yanik, G. A., Peters, C., Baruchel, A., … Grupp, S. A. (2018). Tisagenlecleucel in Children and Young Adults with B-Cell Lymphoblastic Leukemia. The New England journal of medicine, 378(5), 439–448. https://doi.org/10.1056/NEJMoa1709866
- Litzow, M. R., Sun, Z., Mattison, R. J., Paietta, E. M., Roberts, K. G., Zhang, Y., Racevskis, J., Lazarus, H. M., Rowe, J. M., Arber, D. A., Wieduwilt, M. J., Liedtke, M., Bergeron, J., Wood, B. L., Zhao, Y., Wu, G., Chang, T. C., Zhang, W., Pratz, K. W., Dinner, S. N., … Tallman, M. S. (2024). Blinatumomab for MRD-Negative Acute Lymphoblastic Leukemia in Adults. The New England journal of medicine, 391(4), 320–333. https://doi.org/10.1056/NEJMoa2312948
- Lancet, J. E., Uy, G. L., Newell, L. F., Lin, T. L., Ritchie, E. K., Stuart, R. K., Strickland, S. A., Hogge, D., Solomon, S. R., Bixby, D. L., Kolitz, J. E., Schiller, G. J., Wieduwilt, M. J., Ryan, D. H., Faderl, S., & Cortes, J. E. (2021). CPX-351 versus 7+3 cytarabine and daunorubicin chemotherapy in older adults with newly diagnosed high-risk or secondary acute myeloid leukaemia: 5-year results of a randomised, open-label, multicentre, phase 3 trial. The Lancet. Haematology, 8(7), e481–e491. https://doi.org/10.1016/S2352-3026(21)00134-4
- Erba, H. P., Montesinos, P., Kim, H. J., Patkowska, E., Vrhovac, R., Žák, P., Wang, P. N., Mitov, T., Hanyok, J., Kamel, Y. M., Rohrbach, J. E. C., Liu, L., Benzohra, A., Lesegretain, A., Cortes, J., Perl, A. E., Sekeres, M. A., Dombret, H., Amadori, S., Wang, J., … QuANTUM-First Study Group (2023). Quizartinib plus chemotherapy in newly diagnosed patients with FLT3-internal-tandem-duplication-positive acute myeloid leukaemia (QuANTUM-First): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet (London, England), 401(10388), 1571–1583. https://doi.org/10.1016/S0140-6736(23)00464-6
- Foà, R., Bassan, R., Vitale, A., Elia, L., Piciocchi, A., Puzzolo, M. C., Canichella, M., Viero, P., Ferrara, F., Lunghi, M., Fabbiano, F., Bonifacio, M., Fracchiolla, N., Di Bartolomeo, P., Mancino, A., De Propris, M. S., Vignetti, M., Guarini, A., Rambaldi, A., Chiaretti, S., … GIMEMA Investigators (2020). Dasatinib-Blinatumomab for Ph-Positive Acute Lymphoblastic Leukemia in Adults. The New England journal of medicine, 383(17), 1613–1623. https://doi.org/10.1056/NEJMoa2016272
- Heuser, M., Freeman, S. D., Ossenkoppele, G. J., Buccisano, F., Hourigan, C. S., Ngai, L. L., Tettero, J. M., Bachas, C., Baer, C., Béné, M. C., Bücklein, V., Czyz, A., Denys, B., Dillon, R., Feuring-Buske, M., Guzman, M. L., Haferlach, T., Han, L., Herzig, J. K., Jorgensen, J. L., … Cloos, J. (2021). 2021 Update on MRD in acute myeloid leukemia: a consensus document from the European LeukemiaNet MRD Working Party. Blood, 138(26), 2753–2767. https://doi.org/10.1182/blood.2021013626
- Issa, G. C., Aldoss, I., DiPersio, J., Cuglievan, B., Stone, R., Arellano, M., Thirman, M. J., Patel, M. R., Dickens, D. S., Shenoy, S., Shukla, N., Kantarjian, H., Armstrong, S. A., Perner, F., Perry, J. A., Rosen, G., Bagley, R. G., Meyers, M. L., Ordentlich, P., Gu, Y., … Stein, E. M. (2023). The menin inhibitor revumenib in KMT2A-rearranged or NPM1-mutant leukaemia. Nature, 615(7954), 920–924. https://doi.org/10.1038/s41586-023-05812-3
- Stock, W., Luger, S. M., Advani, A. S., Yin, J., Harvey, R. C., Mullighan, C. G., Willman, C. L., Fulton, N., Laumann, K. M., Malnassy, G., Paietta, E., Parker, E., Geyer, S., Mrózek, K., Bloomfield, C. D., Sanford, B., Marcucci, G., Liedtke, M., Claxton, D. F., Foster, M. C., … Larson, R. A. (2019). A pediatric regimen for older adolescents and young adults with acute lymphoblastic leukemia: results of CALGB 10403. Blood, 133(14), 1548–1559. https://doi.org/10.1182/blood-2018-10-881961
- Wei, A. H., Döhner, H., Pocock, C., Montesinos, P., Afanasyev, B., Dombret, H., Ravandi, F., Sayar, H., Jang, J. H., Porkka, K., Selleslag, D., Sandhu, I., Turgut, M., Giai, V., Ofran, Y., Kizil Çakar, M., Botelho de Sousa, A., Rybka, J., Frairia, C., Borin, L., … QUAZAR AML-001 Trial Investigators (2020). Oral Azacitidine Maintenance Therapy for Acute Myeloid Leukemia in First Remission. The New England journal of medicine, 383(26), 2526–2537. https://doi.org/10.1056/NEJMoa2004444