Key Takeaways
A mast cell is a long-lived immune cell that lives in tissues like the skin, gut, and lungs, where it acts as a first responder against pathogens and allergens by releasing histamine and other chemicals.
- Function ▾: Mast cells are best known for driving allergic reactions and anaphylaxis, but they also help with wound healing, parasite defense, and venom detoxification.
- Related Disorders ▾: Disorders of mast cells fall into two main groups: mastocytosis (too many mast cells, often driven by the KIT D816V mutation) and mast cell activation syndrome (normal numbers but inappropriate release of mediators).
- Laboratory Investigation & Diagnosis ▾: The serum tryptase test is the key blood marker. A baseline above 20 ng/mL or a sharp rise during an attack signals abnormal mast cell activity. Diagnosis follows the World Health Organization (WHO) 2022 criteria and combines clinical features, tryptase levels, bone marrow findings, surface markers, and KIT mutation testing [2].
- Treatment ▾: Modern treatment ranges from antihistamines and epinephrine auto-injectors for symptoms, to targeted KIT inhibitors like avapritinib and midostaurin for advanced systemic mastocytosis [4,5]
*Click ▾ for more information
Why Mast Cells Matter
When someone breaks out in hives, struggles to breathe after a bee sting, or collapses from anaphylaxis, a mast cell is usually at the center of the story. Mast cells are tiny but powerful. They sit quietly in tissues until something — a pollen grain, a parasite, a drug, or a venom — sets them off. Within seconds they can flood the body with chemicals that change blood flow, tighten airways, and call in other immune cells.
The mast cell is a perfect example of how a single cell can link the innate and adaptive immune systems. Understanding the mast cell helps explain why symptoms come and go, what triggers to avoid, and how modern treatments work.
A Brief History
Paul Ehrlich first described mast cells in 1878 while still a medical student. He saw large connective-tissue cells that stained intensely with basic dyes and looked "stuffed" with granules. Thinking they had eaten extra nutrients from surrounding tissue, he named them Mastzellen, from the German word Mast meaning the fattening of livestock. We now know the granules are made by the cell, not eaten, but the name stuck.
What Is a Mast Cell?
A mast cell is a long-lived immune cell that lives in tissues rather than blood. It is packed with granules — small storage packets full of chemical messengers — that it can release within seconds.
- Where it comes from: Mast cells start as CD34+ stem cells in the bone marrow. Unlike most blood cells, they leave the marrow before they fully mature and finish growing up only after they reach a tissue.
- What it stores: Each cell carries large amounts of histamine, heparin (a natural blood thinner), and protein-cutting enzymes called tryptase and chymase.
- What it senses: The hallmark feature is FcεRI, a high-affinity receptor that binds IgE antibodies. This lets mast cells react to specific allergens with extreme sensitivity.
- How we identify it: Mature mast cells display CD117, also known as c-KIT. This receptor binds stem cell factor (SCF), the survival signal mast cells depend on for life.
Morphology: What a Mast Cell Looks Like
Under the Light Microscope
Mast cells are 10 to 20 micrometers across — larger than most white blood cells. They are round in the bone marrow but become spindle-shaped in connective tissue, especially near nerves and blood vessels.
The most striking feature is the dense cluster of granules. These granules show metachromasia, meaning they stain a different color than the dye itself. With toluidine blue, for example, they turn reddish-purple. This happens because the dye molecules clump together when they bind the highly acidic heparin inside the granules.
The nucleus is small, round, and sits in the center. It is mononuclear (a single shape), unlike the segmented, multi-lobed nucleus of a basophil. The granulation can be so heavy that the nucleus is hidden.
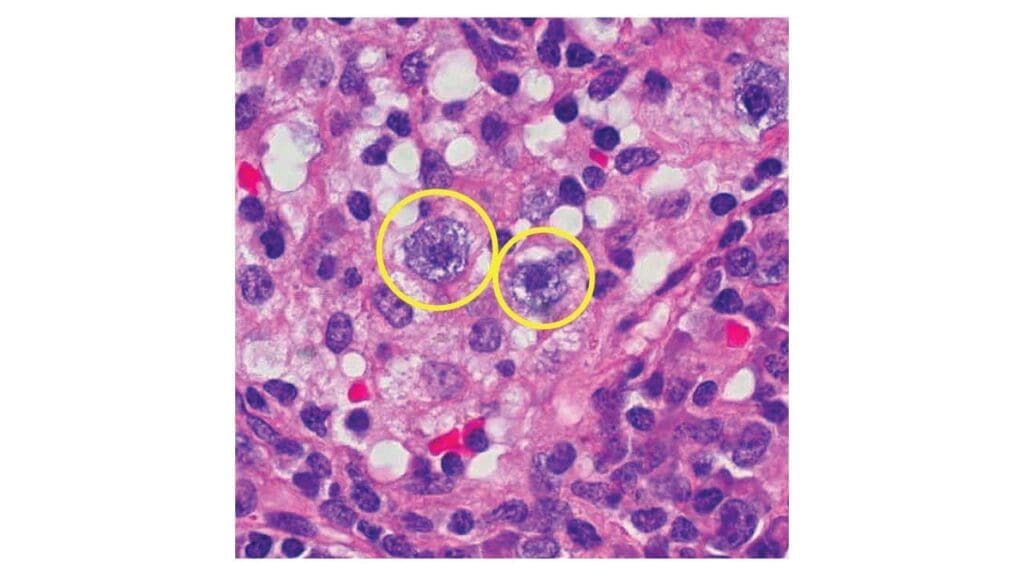
Under the Electron Microscope
At higher magnification, mast cells reveal:
- Secretory granules with distinctive internal patterns described as scrolls, crystals, or particles. These reflect tightly packed proteases and proteoglycans.
- Microvilli (finger-like surface projections) that increase surface area for sensing the environment.
- A well-developed Golgi apparatus and rough endoplasmic reticulum, showing the cell's high capacity to make and package mediators.
Mast Cell vs. Basophil
These two cells are easy to confuse. Here is how to tell them apart.
| Feature | Mast Cell | Basophil |
| Nucleus | Mononuclear, round or oval | Segmented (bi- or tri-lobed) |
| Size | Larger (10–20 µm) | Smaller (5–10 µm) |
| Granules | Many, small, uniform | Fewer, larger, variable |
| Granule pattern (EM) | Scrolls, crystals, particles | Coarse, particulate, "checkered" |
| Normal location | Tissues (skin, gut, lung) | Peripheral blood |
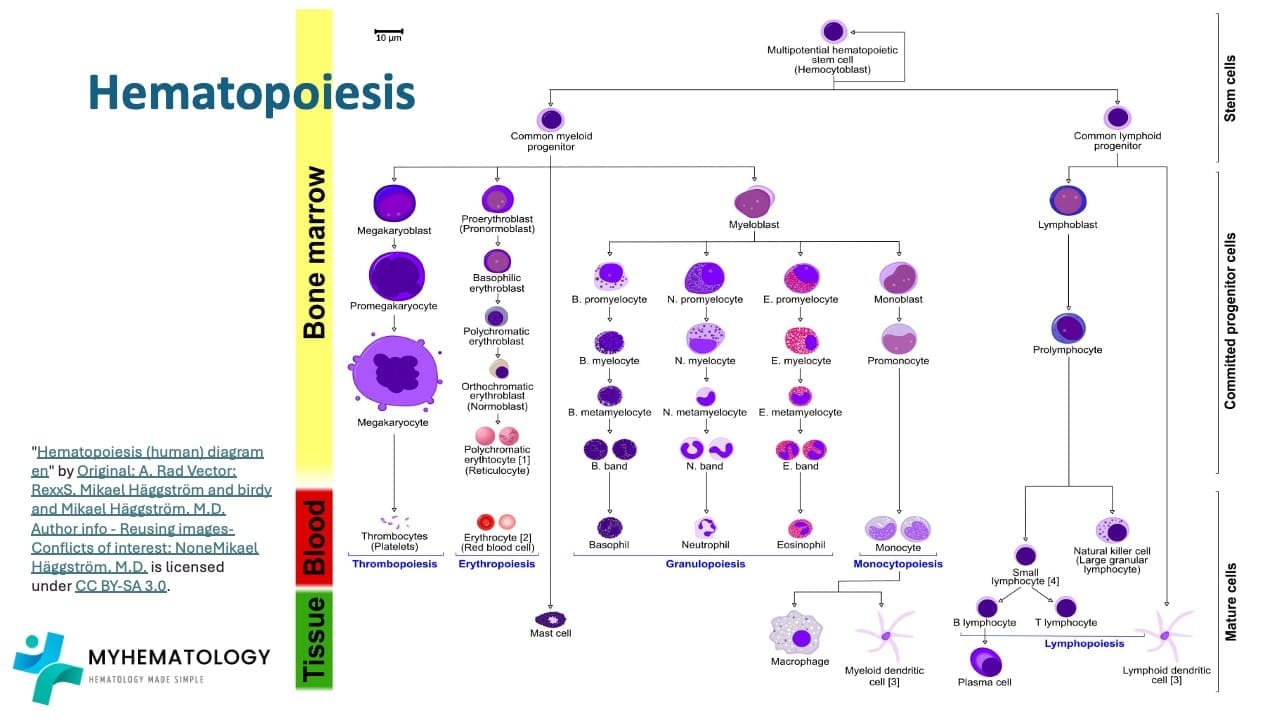
How Mast Cells Develop
Mast cells come from CD34+ stem cells in the bone marrow. They share a common myeloid ancestor with basophils and eosinophils, but their pathway diverges early.
The progression looks like this:
Hematopoietic Stem Cell → Common Myeloid Progenitor → Mast Cell Progenitor → Tissue Mast Cell
The progenitor expresses CD34, CD117, and CD13 but has no granules yet.
Why c-KIT and SCF Matter
Mast cell life depends on the partnership between c-KIT (CD117) and its ligand, stem cell factor (SCF). Most other myeloid cells lose CD117 as they mature, but mast cells keep it for life. That is why CD117 is the most reliable marker to find mast cells in tissue samples.
Unique Trafficking
Unlike neutrophils or basophils, mast cells leave the bone marrow before they are fully formed. They travel briefly through the blood, then settle into tissues like skin, gut, or lung, where local cytokines guide their final maturation. This produces two main human subtypes:
- Mucosal Mast Cells (MCT): Found mainly in the intestinal and respiratory mucosa. They contain tryptase only.
- Connective Tissue Mast Cells (MCTC): Found in skin, lung interstitium, and intestinal submucosa. They contain tryptase, chymase, and carboxypeptidase.
Lifespan and Reference Range
Mast cells split their existence between two compartments:
- In blood: Mast cell progenitors pass through quickly. The bloodstream is just a highway to the tissues.
- In tissue: Mature mast cells live for weeks, months, or years. Unlike most mature white blood cells, they can also divide locally during inflammation.
In a normal peripheral blood smear, the expected count of mature mast cells is zero. Even one mast cell on a routine blood film is a red flag and should prompt investigation for a systemic mast cell disorder. In bone marrow, mast cells are present but rare — typically less than 0.1% of nucleated cells, usually clustered near bone spicules or small blood vessels.
What Mast Cells Do
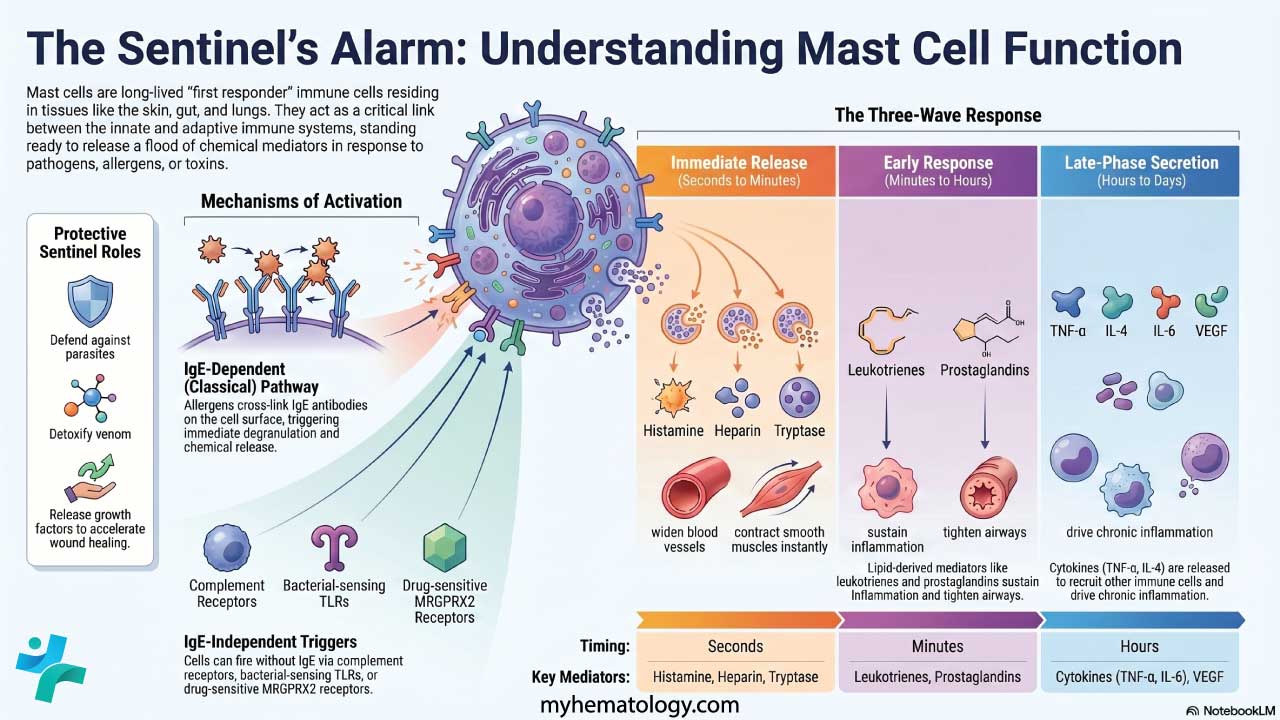
Mast cells link the innate and adaptive immune systems. Their job is to detect "danger signals" and start a fast, local inflammatory response.
How a Mast Cell Gets Activated
IgE-Dependent Activation (the Classical Allergy Pathway)
This is the basis of Type I hypersensitivity reactions like hay fever, food allergy, and anaphylaxis.
- Sensitization: On first exposure to an allergen, B cells make IgE antibodies. These IgE molecules bind to FcεRI receptors on mast cells. The cell is now "loaded" but not yet firing.
- Cross-linking: On the next exposure, the allergen bridges two or more IgE molecules on the cell surface. This triggers a signaling cascade involving tyrosine kinases such as Syk.
- Degranulation: Calcium floods into the cell. Granules fuse with the cell membrane and dump their contents into the surrounding tissue.
IgE-Independent Activation
Mast cells can also fire without IgE, through:
- Complement receptors that bind C3a and C5a (anaphylatoxins).
- Toll-like receptors (TLRs) that recognize bacterial molecules like LPS.
- MRGPRX2, a receptor that responds to certain drugs (neuromuscular blockers, some opioids, fluoroquinolones) and neuropeptides like Substance P.
What Mast Cells Release
Mediators come out in three waves.
| Phase | Timing | Key Mediators | What They Do |
| Immediate (pre-formed) | Seconds to minutes | Histamine, heparin, tryptase, chymase | Widen blood vessels, leak fluid into tissues, contract smooth muscle, thin blood |
| Early (lipid-derived) | Minutes to hours | Leukotrienes, prostaglandins (PGD2), platelet-activating factor (PAF) | Tighten airways (bronchoconstriction), increase mucus, sustain vasodilation |
| Late (cytokines) | Hours to days | TNF-α, IL-4, IL-5, IL-6, VEGF | Bring in neutrophils and eosinophils, drive chronic inflammation, remodel tissue |
This sequence explains why an allergic reaction can have an immediate burst (hives, swelling, wheeze), then a delayed second wave hours later.
Common Triggers
In real-world practice, mast cells can be set off by foods (nuts, shellfish), insect stings, medications (NSAIDs, opioids, antibiotics, contrast dye, some anesthetics), heat, cold, exercise, alcohol, stress, and infections. Triggers vary widely between individuals, which is why identifying personal patterns is a core part of management.
Helpful Roles in Health
Even though mast cells get blamed for allergies, they protect the body in several ways:
- Defense against parasites: Mast cell mediators speed up gut motility and mucus production, helping "weep and sweep" worms out of the intestine.
- Venom detox: Proteases like carboxypeptidase A3 break down toxins from snake and bee venom.
- Wound healing: By releasing VEGF and fibroblast growth factor, mast cells encourage new blood vessels and bring in fibroblasts to repair tissue.
- Local blood flow: Histamine release fine-tunes circulation in specific tissues.
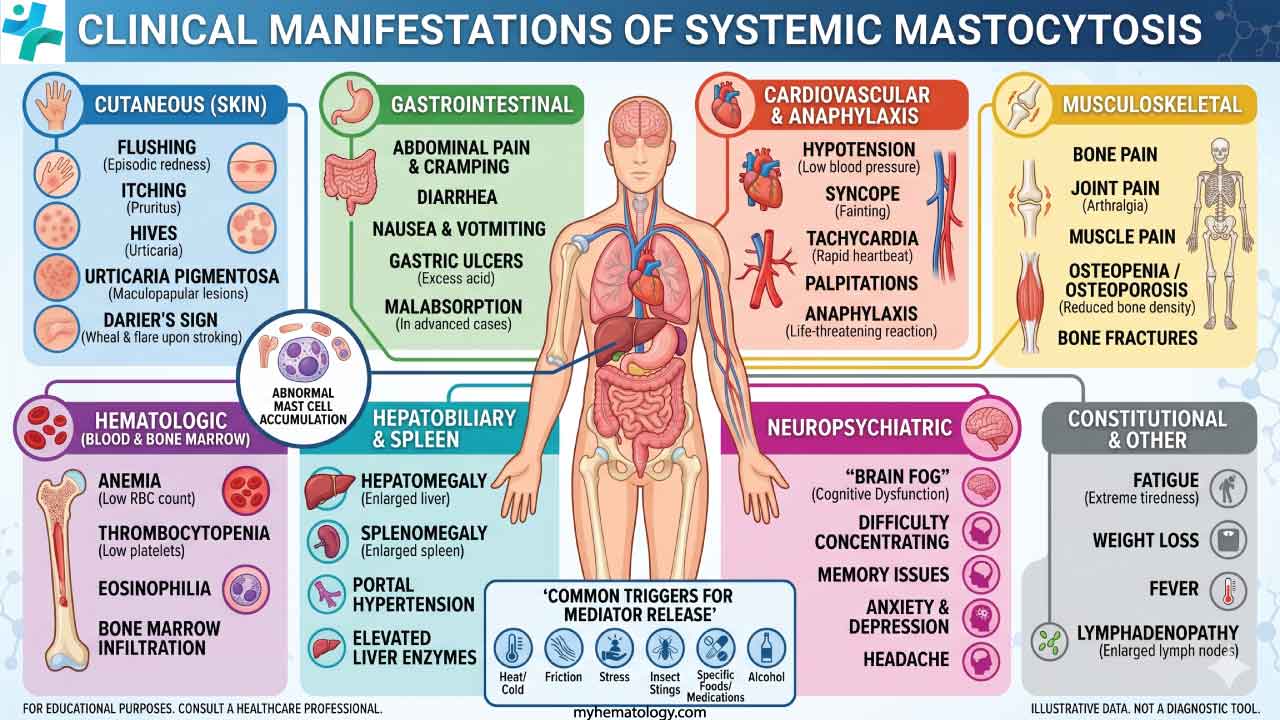
Mast Cell Disorders
In clinical practice, problems with mast cells are grouped under Mast Cell Activation Disease (MCAD). The spectrum runs from cells that are simply too sensitive to rare blood cancers where they multiply uncontrollably.
The most important distinction is between mastocytosis (a problem of quantity and clonality) and mast cell activation syndrome (a problem of quality and function).
Anaphylaxis: The Mast Cell Emergency
The most dangerous outcome of mast cell activation is anaphylaxis. This is a sudden, whole-body reaction with airway swelling, wheezing, low blood pressure, and shock. Treatment is intramuscular epinephrine — the only medication that reverses the reaction. Antihistamines and steroids help with skin and delayed symptoms but are never substitutes for epinephrine. Anyone with a history of anaphylaxis or systemic mastocytosis should carry an epinephrine auto-injector at all times.
Mastocytosis (Clonal Proliferation)
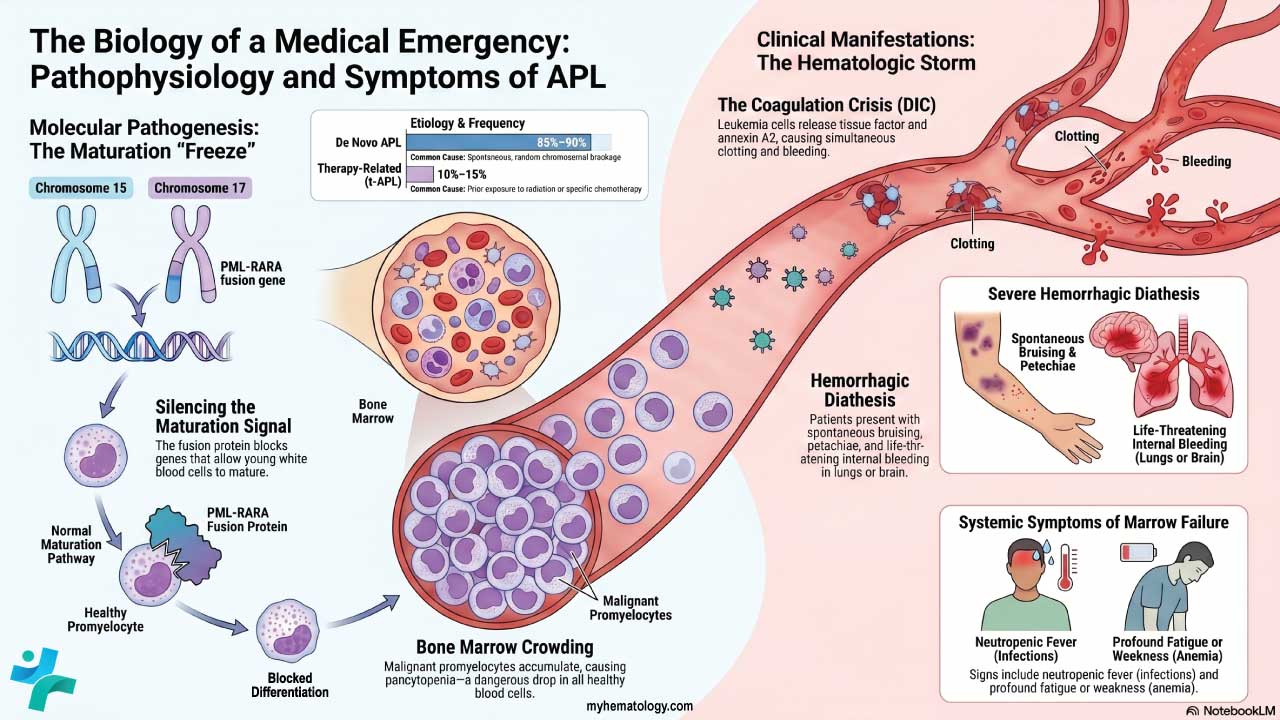
Mastocytosis is the abnormal, clonal expansion of mast cells in one or more organs. Most adult cases are driven by an activating mutation in the KIT gene, almost always KIT D816V [3].
- Cutaneous Mastocytosis (CM): The disease stays in the skin. Most common in children. The classic finding is urticaria pigmentosa — brown, itchy patches that swell up when rubbed (Darier's sign). In children, most cases resolve spontaneously around puberty, which is reassuring news for parents.
- Systemic Mastocytosis (SM): Mast cells spread beyond the skin, usually into the bone marrow. The 2022 WHO Classification divides SM by disease burden ("B-findings") and organ damage ("C-findings") [2]:
- Bone Marrow Mastocytosis (BMM): A subtype of indolent SM with marrow involvement but no skin lesions and low tryptase.
- Indolent SM (ISM): The most common adult form. Low burden, good prognosis.
- Smoldering SM (SSM): Higher burden (≥2 B-findings) without organ failure.
- Advanced SM: Includes Aggressive SM (ASM) and SM with an Associated Hematological Neoplasm (SM-AHN). These carry a worse prognosis due to organ damage.
- Mast Cell Leukemia (MCL): Rare and highly aggressive, defined by ≥20% mast cells in the bone marrow aspirate.
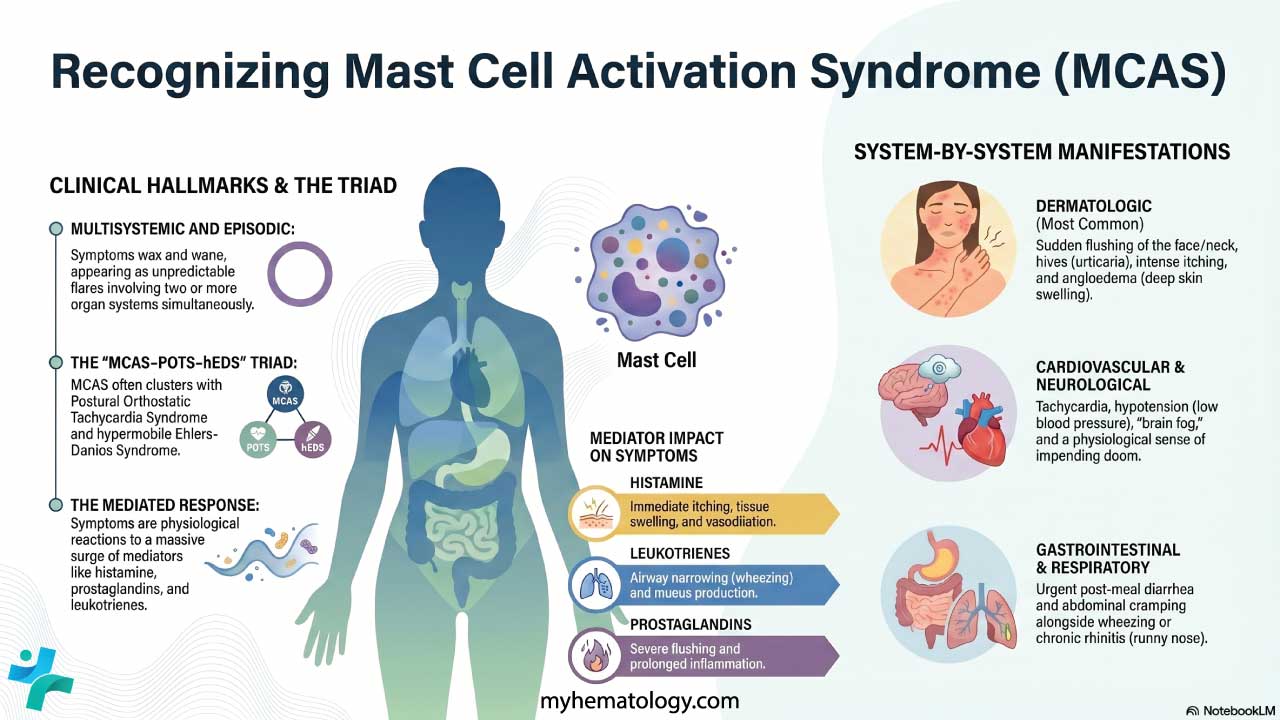
Mast Cell Activation Syndrome (MCAS)
In MCAS, mast cell numbers are usually normal, but the cells release their mediators inappropriately. Diagnosis requires three criteria [7]:
- Clinical: Recurrent symptoms involving at least two organ systems (for example, hives plus wheezing).
- Biochemical: A measurable rise in serum tryptase during an episode (see formula below).
- Treatment response: Symptoms improve with mast cell–blocking drugs like H1/H2 antihistamines or leukotriene antagonists.
MCAS subtypes
- Primary (clonal) MCAS: Clonal mast cells are present (often with the KIT mutation) but don't meet full SM criteria.
- Secondary MCAS: Normal mast cells reacting excessively to a known trigger like a true allergy.
- Idiopathic MCAS: No clonal markers, no clear trigger.
Hereditary Alpha-Tryptasemia (HAT)
HAT is a genetic trait — not a cancer — caused by extra copies of the TPSAB1 gene. People with HAT have permanently raised baseline tryptase (often above 8–10 ng/mL) and may have symptoms similar to MCAS. They are also at higher risk of severe anaphylaxis [6]. HAT is surprisingly common in the general population, and under updated WHO criteria, baseline tryptase should be corrected for TPSAB1 copy number to avoid overestimating mast cell burden.
Laboratory Investigations
The diagnostic workup moves from non-invasive screening to definitive tissue analysis. The key distinction is between a screening test (suggests disease) and a diagnostic test (confirms it).
Serum Tryptase: The Primary Biomarker
Tryptase is the most specific blood marker of mast cell activity [8].
- Baseline tryptase: Measured when the patient feels well. A level above 20 ng/mL is a minor diagnostic criterion for systemic mastocytosis.
- Acute (event) tryptase: Measured within 1 to 4 hours of a suspected episode.
- The rise formula to confirm activation:
Tpeak ≥ (1.2 x Tbaseline) + 2 ng/mL
Worked example: A patient with a baseline tryptase of 5 ng/mL has an episode of hives, wheezing, and low blood pressure. The threshold to confirm activation is (1.2 × 5) + 2 = 8 ng/mL. If their peak tryptase during the episode is 10 ng/mL, the criterion is met.
Bone Marrow Examination
If systemic mastocytosis is suspected — for example, due to elevated baseline tryptase or unexplained anaphylaxis — a bone marrow aspirate and trephine biopsy are required.
- Aspirate morphology: Looks for atypical mast cells. Normal mast cells are round; neoplastic ones are often spindle-shaped (fusiform), have fewer granules, or have off-center nuclei.
- Trephine histology: The gold standard. Pathologists look for dense mast cell infiltrates of 15 or more cells clustered together.
- Immunohistochemistry (IHC): Routine stains can miss mast cells. Specific stains help:
- Tryptase and CD117 mark all mast cells.
- CD25 and CD2 are aberrant markers — normal mast cells don't express them, so their presence suggests clonality.
Flow Cytometry (Immunophenotyping)
Flow cytometry analyzes surface markers in a liquid marrow sample.
- Normal mast cells: CD117+, CD34−, CD25−, CD2−.
- Neoplastic mast cells: CD117+, CD25+ (most common) and/or CD2+.
CD25 expression on mast cells is a major clue for systemic mastocytosis.
Molecular Genetic Testing
Identifying the KIT D816V mutation has transformed mast cell disease diagnosis [3]. The mutation is found in over 90% of adults with systemic mastocytosis.
Detection uses highly sensitive PCR — most commonly digital droplet PCR — on bone marrow or peripheral blood. It is one of the WHO minor criteria, and it shapes treatment because it makes the disease resistant to first-generation tyrosine kinase inhibitors like imatinib [1].
Clinical Tools: The REMA Score
The REMA score, developed by the Spanish Network on Mastocytosis, helps clinicians predict clonal mast cell disease in patients with anaphylaxis and elevated tryptase. It uses sex, symptom pattern, and tryptase level to flag who needs a bone marrow biopsy. It is a useful example of how clinical reasoning translates into diagnostic decisions.
WHO Diagnostic Criteria for Systemic Mastocytosis
Major criterion:
- Multifocal, dense infiltrates of mast cells (≥15 cells in aggregates) in the bone marrow or another extracutaneous organ.
Minor criteria:
- More than 25% of mast cells in the biopsy or aspirate are atypical or spindle-shaped.
- Detection of the KIT D816V mutation.
- Mast cells express CD25 (with or without CD2 or CD30).
- Baseline serum tryptase greater than 20 ng/mL (not applicable if there is an associated myeloid neoplasm; correct for HAT where relevant).
Diagnosis requires the major criterion plus one minor, or three minor criteria [2].
Treatment and Management
Treatment depends on the type of mast cell disorder, but a few principles apply to almost all patients.
General Principles
- Trigger avoidance: Identifying and avoiding personal triggers reduces episodes.
- Patient education: Patients and caregivers learn to recognize early symptoms and respond quickly.
- Emergency action plan: Anyone at risk of anaphylaxis carries an epinephrine auto-injector and a written plan.
Symptom-Directed Therapy
- H1 antihistamines (cetirizine, loratadine, fexofenadine) — for itching, hives, and flushing.
- H2 antihistamines (famotidine) — for stomach symptoms and added skin control.
- Leukotriene receptor antagonists (montelukast) — for respiratory and skin symptoms.
- Mast cell stabilizers (cromolyn sodium) — particularly useful for gut symptoms.
- Epinephrine auto-injectors — first-line for anaphylaxis.
Targeted Therapy for Advanced Disease
Recent years have transformed treatment of systemic mastocytosis [1]:
- Avapritinib is a selective inhibitor of the KIT D816V mutation. It was approved by the FDA in 2021 for advanced systemic mastocytosis and in 2023 for indolent systemic mastocytosis. Trials show major improvements in symptoms, tryptase levels, and bone marrow burden [4].
- Midostaurin is a multikinase inhibitor approved in 2017 for advanced SM. It was the first targeted therapy to show survival benefit [5].
- Bezuclastinib is an investigational selective KIT inhibitor in clinical trials for both advanced and non-advanced SM.
For aggressive disease, options also include cytoreductive chemotherapy and, in selected cases, allogeneic stem cell transplant.
Pediatric Considerations
Most childhood cutaneous mastocytosis improves on its own by adolescence. Children rarely need systemic treatment, though severe cases benefit from antihistamines and trigger education for caregivers.
Frequently Asked Questions (FAQs)
What is a mast cell and what does it do?
A mast cell is a tissue-based immune cell that protects the body from parasites, toxins, and pathogens. It sits in the skin, gut, and airways and stores chemicals like histamine in tiny granules. When triggered, it releases these chemicals quickly to start an inflammatory response. Mast cells are also the main drivers of allergic reactions, including hay fever, hives, and anaphylaxis.
How are mast cells different from basophils?
Mast cells live in tissues; basophils circulate in blood. Mast cells are larger (10–20 µm) and have a single round nucleus, while basophils are smaller (5–10 µm) with a segmented nucleus. Mast cells survive for weeks to years, but basophils last only a few days. Both contain histamine and respond to IgE, but their roles and locations differ.
What is mastocytosis?
Mastocytosis is a rare disorder where abnormal mast cells build up in one or more organs. In children it usually affects only the skin and often resolves on its own. In adults it more often involves the bone marrow (systemic mastocytosis) and is driven by a mutation called KIT D816V. Symptoms include flushing, itching, abdominal pain, and anaphylaxis.
How do doctors test for mast cell disease?
The first test is usually a blood serum tryptase level. A baseline above 20 ng/mL suggests excess mast cells. If systemic mastocytosis is suspected, a bone marrow biopsy is performed to look for clusters of abnormal mast cells, KIT D816V mutation testing, and surface marker analysis using flow cytometry. The diagnosis follows the World Health Organization (WHO) 2022 criteria.
Can mast cell disorders be treated?
Yes. Treatment depends on the type and severity. Most patients use trigger avoidance combined with H1 and H2 antihistamines, leukotriene blockers, and mast cell stabilizers like cromolyn sodium. Anyone at risk of anaphylaxis carries an epinephrine auto-injector. For advanced systemic mastocytosis, targeted drugs such as avapritinib and midostaurin block the KIT mutation directly and can dramatically improve outcomes.
What triggers a mast cell reaction?
Common triggers include allergens (foods, insect stings, medications), heat, cold, exercise, alcohol, stress, infections, and certain drugs such as NSAIDs, opioids, contrast dye, and some anesthetics. Triggers vary between people, so identifying personal triggers through a symptom diary is an important part of management.
Glossary of Related Medical Terms
- Mast cell — a tissue-based immune cell packed with granules of histamine and other chemicals; releases them rapidly to fight infection or trigger allergic reactions.
- Granules — small storage packets inside a cell that hold chemical messengers ready for instant release.
- Histamine — a chemical released by mast cells that causes itching, redness, swelling, and widening of blood vessels.
- Tryptase — an enzyme stored almost exclusively in mast cells; measured in blood as a marker of mast cell activity.
- Heparin — a natural blood thinner stored in mast cell granules.
- IgE (Immunoglobulin E) — an antibody made by the immune system that binds to mast cells and triggers allergic reactions when it meets its matching allergen.
- FcεRI — the high-affinity receptor on mast cells that holds IgE antibodies in place.
- CD117 (c-KIT) — a surface receptor on mast cells; binds stem cell factor and is essential for mast cell survival.
- Stem Cell Factor (SCF) — a signaling protein that tells mast cells to grow, survive, and stay where they are.
- Degranulation — the process by which a mast cell empties its granules, releasing chemicals into the surrounding tissue.
- Anaphylaxis — a severe, rapid, whole-body allergic reaction that can cause breathing difficulty, low blood pressure, and shock.
- Mastocytosis — a group of disorders where too many mast cells build up in the skin, bone marrow, or other organs.
- Mast Cell Activation Syndrome (MCAS) — a condition where mast cells release their chemicals inappropriately even though their numbers are normal.
- KIT D816V — a specific change (mutation) in the KIT gene found in over 90% of adults with systemic mastocytosis; it makes mast cells grow without proper signals.
- Hereditary Alpha-Tryptasemia (HAT) — a genetic trait where extra copies of the TPSAB1 gene cause permanently raised tryptase levels.
- Urticaria Pigmentosa — itchy brown spots on the skin caused by mast cell buildup, most common in children.
- Darier's Sign — when rubbing a mastocytosis skin lesion makes it swell or hive; a classic clinical clue.
- Helminths — parasitic worms; mast cells help expel them from the gut.
- Metachromasia — when a dye changes color after binding to a tissue; mast cell granules turn purple-red with blue dyes.
Disclaimer: This article is intended for informational purposes only and is specifically targeted towards medical students. It is not intended to be a substitute for informed professional medical advice, diagnosis, or treatment. While the information presented here is derived from credible medical sources and is believed to be accurate and up-to-date, it is not guaranteed to be complete or error-free. See additional information.
References
- Valent, P., Akin, C., Hartmann, K., Nilsson, G., Reiter, A., Hermine, O., Sotlar, K., Sperr, W. R., Escribano, L., George, T. I., Kluin-Nelemans, H. C., Ustun, C., Triggiani, M., Brockow, K., Gotlib, J., Orfao, A., Schwartz, L. B., Broesby-Olsen, S., Bindslev-Jensen, C., Kovanen, P. T., … Metcalfe, D. D. (2017). Advances in the Classification and Treatment of Mastocytosis: Current Status and Outlook toward the Future. Cancer research, 77(6), 1261–1270. https://doi.org/10.1158/0008-5472.CAN-16-2234
- Khoury, J. D., Solary, E., Abla, O., Akkari, Y., Alaggio, R., Apperley, J. F., Bejar, R., Berti, E., Busque, L., Chan, J. K. C., Chen, W., Chen, X., Chng, W. J., Choi, J. K., Colmenero, I., Coupland, S. E., Cross, N. C. P., De Jong, D., Elghetany, M. T., Takahashi, E., … Hochhaus, A. (2022). The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia, 36(7), 1703–1719. https://doi.org/10.1038/s41375-022-01613-1
- Valent, P., Akin, C., & Metcalfe, D. D. (2017). Mastocytosis: 2016 updated WHO classification and novel emerging treatment concepts. Blood, 129(11), 1420–1427. https://doi.org/10.1182/blood-2016-09-731893
- Gotlib, J., Reiter, A., Radia, D. H., Deininger, M. W., George, T. I., Panse, J., Vannucchi, A. M., Platzbecker, U., Alvarez-Twose, I., Mital, A., Hermine, O., Dybedal, I., Hexner, E. O., Hicks, L. K., Span, L., Mesa, R., Bose, P., Pettit, K. M., Heaney, M. L., Oh, S. T., … DeAngelo, D. J. (2021). Efficacy and safety of avapritinib in advanced systemic mastocytosis: interim analysis of the phase 2 PATHFINDER trial. Nature medicine, 27(12), 2192–2199. https://doi.org/10.1038/s41591-021-01539-8
- Gotlib, J., Kluin-Nelemans, H. C., George, T. I., Akin, C., Sotlar, K., Hermine, O., Awan, F. T., Hexner, E., Mauro, M. J., Sternberg, D. W., Villeneuve, M., Huntsman Labed, A., Stanek, E. J., Hartmann, K., Horny, H. P., Valent, P., & Reiter, A. (2016). Efficacy and Safety of Midostaurin in Advanced Systemic Mastocytosis. The New England journal of medicine, 374(26), 2530–2541. https://doi.org/10.1056/NEJMoa1513098
- Lyons, J. J., Yu, X., Hughes, J. D., Le, Q. T., Jamil, A., Bai, Y., Ho, N., Zhao, M., Liu, Y., O'Connell, M. P., Trivedi, N. N., Nelson, C., DiMaggio, T., Jones, N., Matthews, H., Lewis, K. L., Oler, A. J., Carlson, R. J., Arkwright, P. D., Hong, C., … Milner, J. D. (2016). Elevated basal serum tryptase identifies a multisystem disorder associated with increased TPSAB1 copy number. Nature genetics, 48(12), 1564–1569. https://doi.org/10.1038/ng.3696
- Valent, P., Akin, C., Bonadonna, P., Hartmann, K., Brockow, K., Niedoszytko, M., Nedoszytko, B., Siebenhaar, F., Sperr, W. R., Oude Elberink, J. N. G., Butterfield, J. H., Alvarez-Twose, I., Sotlar, K., Reiter, A., Kluin-Nelemans, H. C., Hermine, O., Gotlib, J., Broesby-Olsen, S., Orfao, A., Horny, H. P., … Metcalfe, D. D. (2019). Proposed Diagnostic Algorithm for Patients with Suspected Mast Cell Activation Syndrome. The journal of allergy and clinical immunology. In practice, 7(4), 1125–1133.e1. https://doi.org/10.1016/j.jaip.2019.01.006
- Schwartz L. B. (2006). Diagnostic value of tryptase in anaphylaxis and mastocytosis. Immunology and allergy clinics of North America, 26(3), 451–463. https://doi.org/10.1016/j.iac.2006.05.010