Key Takeaways
Chronic lymphocytic leukemia also known as CLL is a slow progressing cancer due to overgrowth and accumulation of small incompetent mature-looking B-lymphocytes in the blood, bone marrow and lymphoid tissues. Small lymphocytic leukemia is a different clinical manifestation of this disorder.
- Incidence ▾: Chronic lymphocytic leukemia is the most common adult leukemia in Western countries and almost always affects people over 60, with a male-to-female ratio of about 2:1.
- Symptoms ▾: Most patients with chronic lymphocytic leukemia have no symptoms at diagnosis and are picked up on a routine blood test showing persistent lymphocytosis [1].
- Laboratory Investigations ▾: flow cytometry confirms the CD5+, CD19+, CD20 weak, CD23+ signature. Two molecular tests, the IGHV mutation status and FISH/TP53, are essential before starting first-line therapy because they decide which drug class to use. Stage alone no longer drives treatment.
- Treatment and Management ▾: First-line treatment of chronic lymphocytic leukemia is now entirely targeted therapy: continuous BTK inhibitors (acalabrutinib or zanubrutinib) or fixed-duration regimens. The newest standard is the all-oral, 14-month combination of acalabrutinib + venetoclax (FDA-approved early 2026) [11]. Chemoimmunotherapy is virtually obsolete.
*Click ▾ for more information
What is Chronic Lymphocytic Leukemia?
Chronic lymphocytic leukemia (CLL) is a cancer that begins in the bone marrow. The marrow churns out B-lymphocytes but these B-cells are abnormal. They look mature under the microscope but cannot fight infection properly. They are also stubbornly long-lived. Over time they accumulate in blood, bone marrow, lymph nodes, and the spleen, slowly crowding out healthy blood cells.
You may also see the term small lymphocytic lymphoma (SLL). CLL and SLL are no longer considered separate diseases. They are the same disorder showing up in different places: CLL when the abnormal cells dominate the blood, SLL when they sit mainly in the lymph nodes.
Who Gets It?
CLL is largely a disease of older adults, with genetics playing a massive role in incidence rates. In 2026, the American Cancer Society projects roughly 22,000 new cases of CLL will be diagnosed in the United States [9].
- Age & Lifetime Risk: The median age at diagnosis is around 70, and cases under 40 are very unusual. The lifetime risk of developing CLL for the average person is approximately 0.6%, or about 1 in 175 people [9].
- Sex: Men are affected almost twice as often as women. The male-to-female ratio remains remarkably stable across global populations at roughly 1.9 to 1 [10].
- Race & Ethnicity: Geography and genetic background heavily influence risk. According to the latest SEER data, incidence rates per 100,000 people highlight stark differences among racial groups [10]:
- White/Caucasian: ~6.8 per 100,000 (Highest risk)
- Black/African American: ~4.5 per 100,000
- Hispanic/Latino: ~3.5 per 100,000
- Asian/Pacific Islander: ~1.5 per 100,000 (Lowest risk)
Where the Cells Come From
The CLL cell starts as a normal B-lymphocyte that gets stuck. It looks mature but behaves abnormally. On flow cytometry it shows a very specific pattern that lets pathologists tell CLL apart from related cancers:
- B-cell markers: CD19 (always), CD20 (weak), CD23 (strong)
- A T-cell marker that should not be there: CD5
- Surface immunoglobulin: weak, restricted to one light chain (kappa or lambda) — proving all cells came from a single ancestor
The combination of CD5 and CD23 is the CLL signature. Mantle cell lymphoma has CD5 but not CD23. Follicular lymphoma has neither.
How CLL Behaves: The Pathophysiology
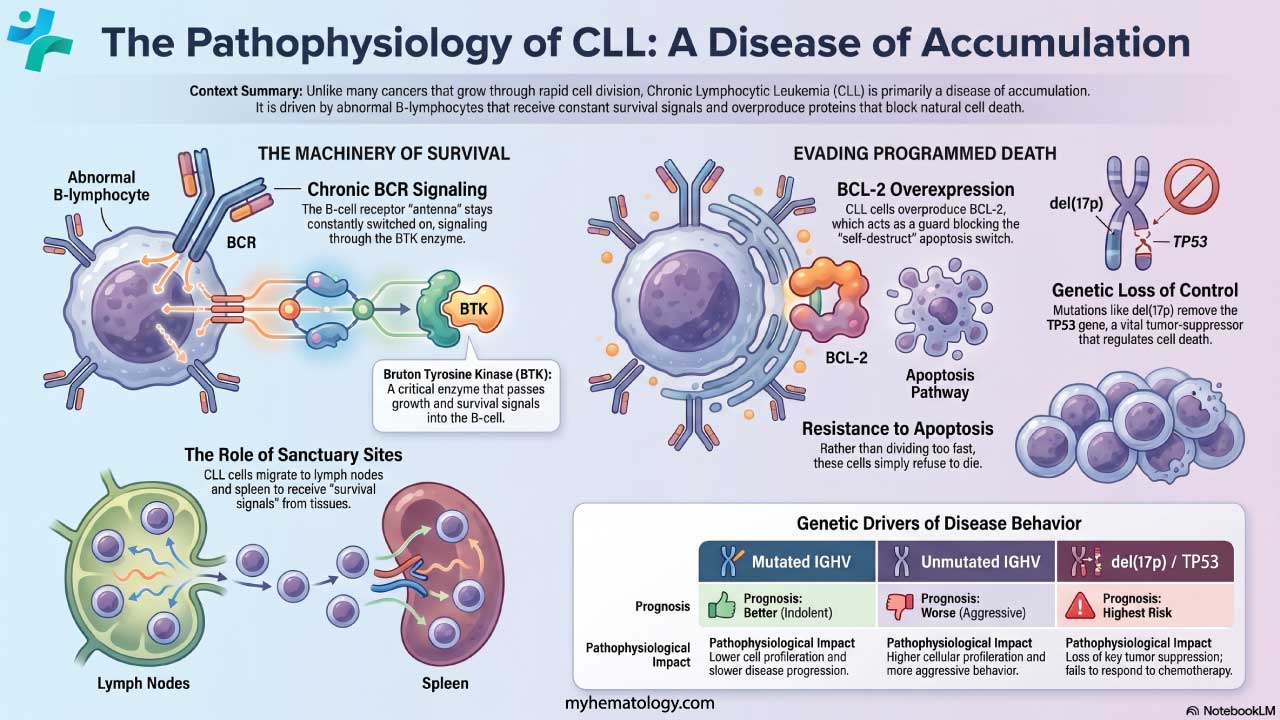
Most cancers grow because cells divide too fast. Chronic lymphocytic leukemia is different: the cells refuse to die. This makes CLL primarily a disease of accumulation [1,4].
The B-Cell Receptor and BTK
The B-cell receptor (BCR) is the antenna on the surface of every B-cell. In CLL, this antenna is constantly switched on — often reacting to the body's own proteins. The signal travels inside the cell through an enzyme called Bruton tyrosine kinase (BTK), which keeps the cell alive and tells it to move into protective tissues. This dependence on BTK is exactly why BTK inhibitor drugs work so well.
Resistance to Cell Death
CLL cells stay alive because they overproduce BCL-2, a protein that blocks apoptosis (programmed cell death). Think of BCL-2 as a guard at the cell's self-destruct switch. By keeping the guard on duty around the clock, CLL cells avoid the death they should naturally undergo. The drug venetoclax removes that guard.
Sanctuary Sites
CLL cells cannot survive alone in blood for long. They migrate to proliferative centers in lymph nodes, spleen, and bone marrow, where supporting cells (nurse-like cells, T-helper cells, and stromal cells) feed them survival signals. These signals all converge on the BTK pathway, which is why BTK inhibitors work even when most cells are hiding in tissues.
What Causes CLL?
The exact cause is unknown. CLL almost certainly arises from a mix of inherited susceptibility, aging, and possibly some environmental exposures.
Genetic Susceptibility
Genetics play a stronger role in CLL than in most adult leukemias [4].
- Family history. Having a first-degree relative with CLL roughly doubles the risk, with some studies reporting higher figures in tightly clustered families.
- MBL (monoclonal B-cell lymphocytosis). This is a precursor state. A small clone of CLL-like cells is present in the blood but at less than 5 × 10⁹/L (the cutoff for diagnosing CLL). MBL is surprisingly common in older adults. Most people with MBL never progress, but a small number convert to true CLL each year [1].
Environmental Factors
The evidence here is much weaker. Some studies link long-term, high-level exposure to certain pesticides and herbicides to CLL risk. Unlike acute leukemias, ionizing radiation has not been shown to cause CLL. Common viruses and bacteria have not been linked to CLL either.
Chronic Lymphocytic Leukemia (CLL) Symptoms
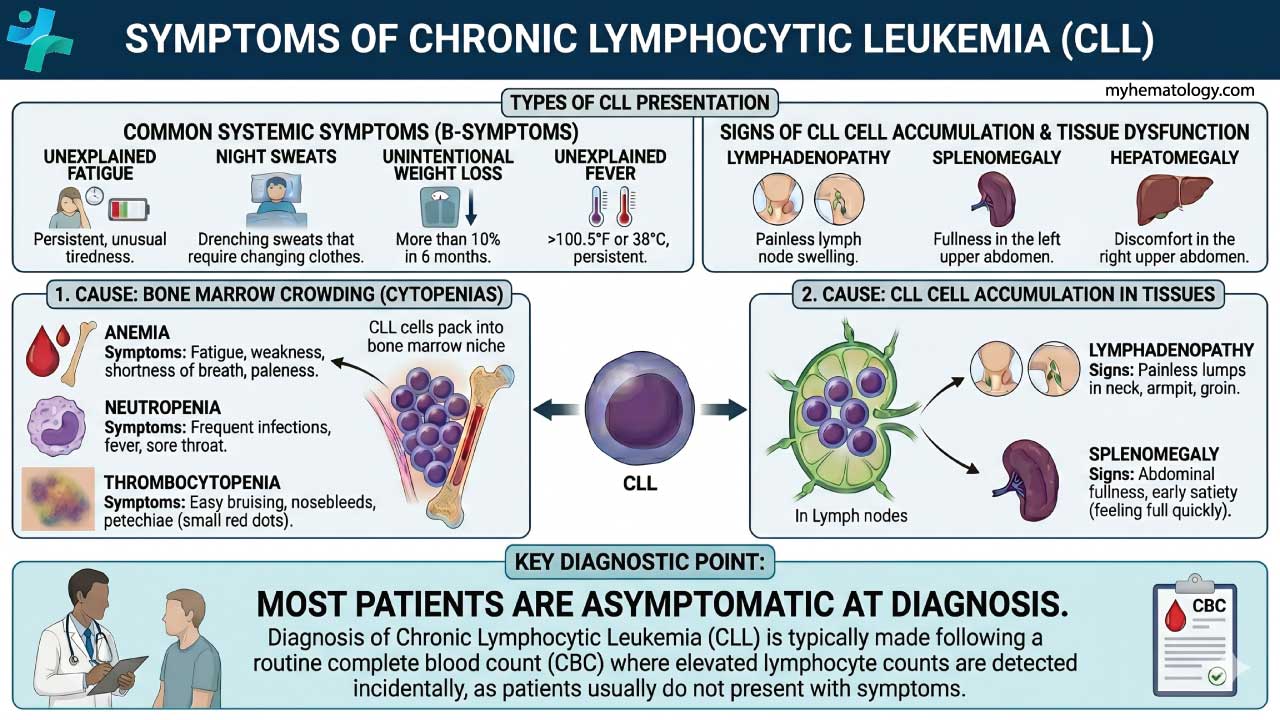
CLL is sometimes called the "silent" leukemia because most patients have no symptoms at diagnosis [1,4].
The Most Common Presentation: No Symptoms at All
Around two-thirds of patients are diagnosed by accident. A routine blood test — perhaps before surgery or at an annual physical — shows an unexpectedly high lymphocyte count. Flow cytometry then confirms CLL.
Constitutional ("B") Symptoms
These are nonspecific signs of active disease and often signal that treatment is needed.
Physical Signs
As CLL cells build up in lymphoid tissues, the doctor may find:
- Enlarged lymph nodes that are firm, rubbery, painless, and often symmetric — typically in the neck, armpits, or groin.
- Splenomegaly (enlarged spleen), sometimes felt as fullness in the upper-left abdomen.
- Hepatomegaly (enlarged liver), less common.
Signs of Bone Marrow Failure
When CLL cells fill the bone marrow, normal blood production suffers. This causes cytopenias (low blood cell counts):
- Anemia (low red cells): fatigue, breathlessness, pallor.
- Thrombocytopenia (low platelets): easy bruising, pinpoint skin spots called petechiae, nosebleeds.
- Neutropenia (low neutrophils): repeated or severe infections.
How Is Chronic Lymphocytic Leukemia Diagnosed?
A workup for chronic lymphocytic leukemia combines blood tests, microscopy, and flow cytometry. Genetic testing is essential before any treatment begins [1,4].
Core Diagnostic Tests
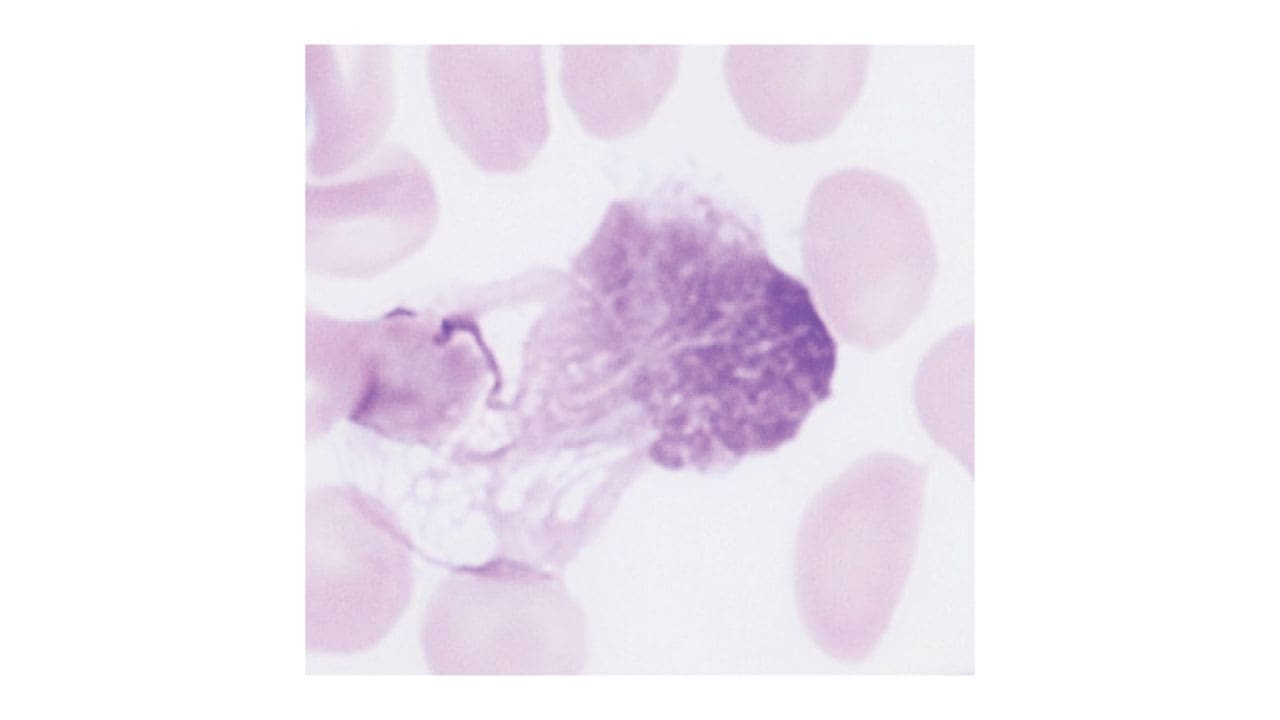
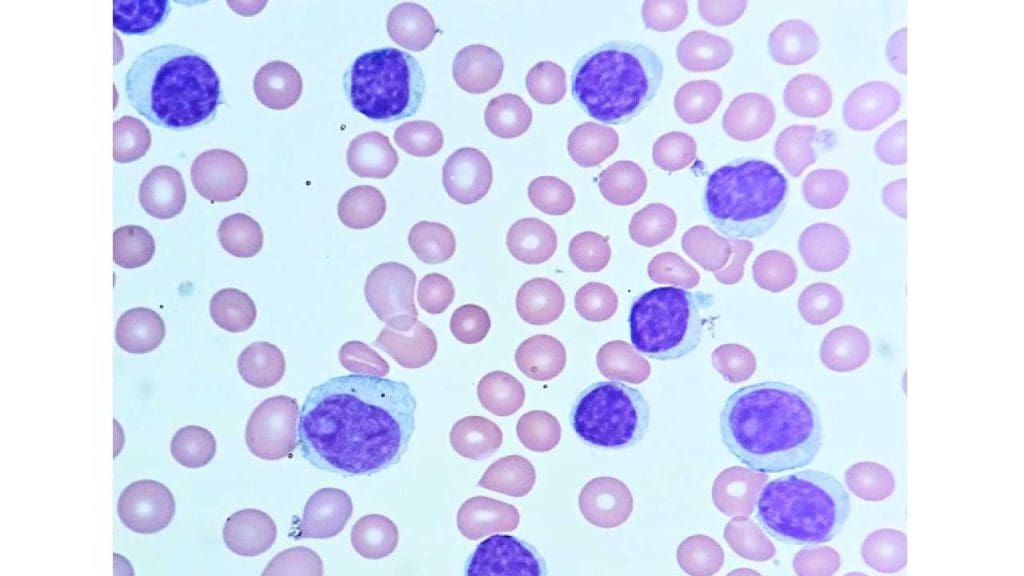
- Complete blood count (CBC): confirms persistent lymphocytosis. Anemia or thrombocytopenia in late disease.
- Peripheral blood smear: shows small mature-looking lymphocytes and smudge cells (fragile CLL cells that smear when spread on a slide). Smudge cells are a classic CLL fingerprint.
- Flow cytometry / immunophenotyping: the diagnostic test. Confirms the CLL signature (CD5+, CD19+, CD20 weak, CD23+, single light chain). At least 5 × 10⁹/L clonal B-cells must be present to diagnose CLL [1].
- Bone marrow biopsy: not needed for diagnosis in most cases but useful for evaluating cytopenias.
- Immunoglobulin levels: often low, reflecting hypogammaglobulinemia and partly explaining why infections are common.
Tests That Predict Behavior
These two tests are required before starting treatment because they shape the choice of drug [2,4]:
- IGHV mutation status: unmutated = more aggressive disease, mutated = more indolent.
- FISH and TP53 sequencing: detects key chromosome changes, especially del(17p) and TP53 mutations.
Other useful markers include serum beta-2 microglobulin and LDH, both of which rise with disease burden.
Chronic Lymphocytic Leukemia (CLL) Staging
Two staging systems are used worldwide. Both rely on physical exam and a CBC; neither needs imaging or bone marrow [1].
The Binet Staging System
The Binet staging system is based on two factors: the number of involved lymph node areas as defined by the presence of enlarged lymph nodes of greater than 1 cm in diameter or organomegaly and the presence of anemia or thrombocytopenia.
The Binet staging system has three stages:
- Stage A: Fewer than 3 lymph node areas are involved and there is no anemia or thrombocytopenia.
- Stage B: 3 or more lymph node areas are involved and there is no anemia or thrombocytopenia.
- Stage C: Anemia (Hb < 10 g/dL) or thrombocytopenia (< 100 × 10⁹/L), regardless of node involvement
Lymphoid areas counted: head/neck, axillae, groins, spleen, liver. Each area counts once even if both sides are involved.
| Risk Category | Old Rai Stage | Findings |
|---|---|---|
| LOW | 0 | Lymphocytosis only |
| INTERMEDIATE | I or II | Lymphocytosis + enlarged nodes and/or enlarged spleen/liver |
| HIGH | III or IV | Anemia (Hb < 11 g/dL) or thrombocytopenia (platelets < 100 × 10⁹/L) |
The Big Shift
Stage alone no longer drives treatment. Today, molecular markers (IGHV, TP53/del(17p)) are at least as important as stage in choosing therapy [2,4].
Molecular Risk Markers: The Genetics That Matter
These two tests are essential before any first-line treatment for chronic lymphocytic leukemia [2,4].
Del(17p): Direct to Targeted Therapy
A patient with active disease and del(17p) goes straight to a BTK or BCL-2 inhibitor. Old chemotherapy regimens like FCR are no longer acceptable for these patients.
Differential Diagnosis of Chronic Lymphocytic Leukemia (CLL)
A persistent high lymphocyte count has many causes. The combination of cell appearance, immunophenotype, and clinical course usually settles the diagnosis.
Reactive (Non-Malignant) Lymphocytosis
Many benign conditions raise lymphocyte counts temporarily:
- Viral infections: infectious mononucleosis (EBV) — driven by T-cells, not B-cells; CMV, HIV
- Pertussis: marked but transient lymphocytosis
- Chronic inflammation: can produce lasting polyclonal lymphocytosis
The decisive feature
Reactive lymphocytosis is polyclonal. CLL is monoclonal which means every cell carries the same light chain.
Atypical CLL Presentation
Prolymphocytic transformation: larger, more aggressive cells in the blood; poor prognosis.
Richter transformation: CLL transforms into an aggressive lymphoma (most often diffuse large B-cell lymphoma). Suspect when a single lymph node grows rapidly and becomes painful, or B symptoms erupt suddenly. Prompt biopsy is essential.
Chronic Lymphocytic Leukemia (CLL) Treatment
CLL treatment has been transformed in the last decade. Chemotherapy has largely been replaced by oral, targeted drugs that patients tolerate well [2,4].
Watchful Waiting (Active Surveillance)
For most newly diagnosed patients, the right answer is to do nothing. Trials show that treating early-stage, asymptomatic CLL does not extend life [1,4]. Patients on active surveillance are seen every 3–6 months for an exam and CBC.
When to Start Treatment
Treatment begins when there is clear evidence of active or progressive disease, including [1]:
- B symptoms (fever, drenching night sweats, weight loss > 10% in 6 months)
- Progressive cytopenias from marrow failure
- Bulky or rapidly growing lymph nodes (often ≥ 10 cm)
- Massive or progressive splenomegaly
- Lymphocyte doubling time under 6 months
- Severe autoimmune complications not responding to standard treatment
Modern First-Line Treatment: Choosing a Targeted Therapy
The choice rests on the patient's molecular profile and what kind of treatment course they prefer (continuous versus time-limited) [2,4].
1. Continuous BTK inhibitors (taken daily, indefinitely): Acalabrutinib and zanubrutinib are the preferred continuous BTK inhibitors today. Both proved more tolerable than ibrutinib in head-to-head trials, especially regarding cardiovascular side effects [5,6]. In late 2025, a 160-mg tablet formulation of zanubrutinib was approved, cutting the daily pill burden in half (from four capsules to two tablets), which significantly improves adherence and quality of life for older adults [12]. BTK inhibitors are particularly important for patients with del(17p) or TP53 mutations.
2. Fixed-duration Acalabrutinib + Venetoclax (all-oral, given for 14 months): Approved by the FDA in February 2026 based on the AMPLIFY trial, this is the new standard for fixed-duration therapy. It is the first highly effective, entirely all-oral regimen, eliminating the need for clinic-based IV infusions [11]. Patients must still undergo strict blood monitoring during the venetoclax "ramp-up" phase to prevent tumor lysis syndrome (TLS).
3. Fixed-duration Venetoclax + Obinutuzumab (given for 12 months): This remains an effective fixed-duration option combining a BCL-2 inhibitor pill with an IV antibody [3]. It also requires a careful weekly ramp-up to avoid TLS.
4. Chemoimmunotherapy (FCR: fludarabine + cyclophosphamide + rituximab): Chemoimmunotherapy has been virtually eliminated from modern CLL care due to the superiority and lower toxicity of targeted agents [4]. It is now considered an almost entirely historical approach.
Treatment for Relapsed or Refractory Chronic Lymphocytic Leukemia
When CLL returns after first-line therapy, several modern options are available:
- Pirtobrutinib (Jaypirca): This non-covalent (reversible) BTK inhibitor received accelerated approval in late 2023 and was converted to full, traditional FDA approval in December 2025 [8]. Because it binds to the BTK enzyme differently, it works against CLL that has developed resistance mutations to first-generation covalent drugs like ibrutinib or acalabrutinib, allowing patients to stay within this highly effective drug class.
- Lisocabtagene maraleucel (liso-cel) became the first CAR-T cell therapy approved for CLL in 2024, used for patients who have failed both BTK and BCL-2 inhibitors.
- Switching BTK inhibitors (for example, from ibrutinib to acalabrutinib) is often feasible if a patient stops the first drug for side effects rather than disease progression.
- Allogeneic stem cell transplant remains an option for selected patients, especially those who have exhausted targeted drugs.
Management of Common Complications
- Infections are the leading cause of death in chronic lymphocytic leukemia. Patients should stay current with inactivated vaccines (influenza, pneumococcal, COVID-19, recombinant zoster) and avoid live vaccines while on treatment. Severe, recurrent infections may warrant intravenous immunoglobulin (IVIG).
- Autoimmune complications: autoimmune hemolytic anemia (AIHA) and immune thrombocytopenia (ITP) are usually treated with corticosteroids first.
- Richter transformation: rapid lymph node growth or sudden B symptoms demand urgent biopsy. Treatment requires aggressive lymphoma chemotherapy and, when possible, CAR-T or transplant.
- Second cancers, especially skin cancers, are more common in CLL. Annual skin checks are recommended.
Clinical Trials
For patients with relapsed disease or aggressive features, clinical trials offer access to next-generation BTK degraders, second-generation BCL-2 inhibitors, and bispecific antibodies that engage T-cells against CLL.
Living With Chronic Lymphocytic Leukemia: A Note for Patients and Caregivers
Many people with CLL live for decades, often with little disruption to daily life. Practical priorities:
- Don't skip routine monitoring, even when feeling well.
- Treat fevers seriously — call the care team for any persistent fever, severe cough, or shaking chills.
- Stay vaccinated with inactivated vaccines.
- Protect your skin: use sun protection, do regular self-checks, and see a dermatologist yearly.
- Watch for "different" lymph node growth — a single rapidly enlarging painful node deserves urgent assessment.
Frequently Asked Questions (FAQs)
What is chronic lymphocytic leukemia?
Chronic lymphocytic leukemia (CLL) is a slow-growing blood cancer in which the bone marrow makes too many abnormal B-lymphocytes, a type of white blood cell. These cells look mature but cannot fight infection properly, and they pile up in the blood, bone marrow, lymph nodes, and spleen. CLL is the most common adult leukemia in Western countries and most often affects people over 65.
Do all patients with chronic lymphocytic leukemia need treatment right away?
No. Most people with CLL do not start treatment at diagnosis. About two-thirds are diagnosed by chance on a routine blood test and feel completely well. Trials have shown that treating early-stage, symptom-free CLL does not extend life and only adds drug side effects, so doctors use "active surveillance" (also called watchful waiting) with regular check-ups. Treatment begins only when there are clear signs of progression, such as worsening anemia, low platelets, drenching night sweats, large lymph nodes, or rapidly rising lymphocyte counts.
How is chronic lymphocytic leukemia diagnosed?
Diagnosis usually starts with an unexpected high lymphocyte count on a complete blood count. Doctors confirm CLL with flow cytometry, a test that identifies the typical CLL fingerprint on the cell surface: CD5, CD19, CD20 (weak), CD23, and a single light chain (kappa or lambda). At least 5 × 10⁹/L clonal B-cells in the blood are required for a CLL diagnosis. Two further tests — IGHV mutation status and FISH for del(17p)/TP53 — are essential before starting treatment because they decide which drugs will work best.
What are the modern treatments for chronic lymphocytic leukemia?
First-line treatment is now built around targeted oral drugs, not chemotherapy. The two main options are continuous BTK inhibitors (acalabrutinib or zanubrutinib are preferred over older ibrutinib due to fewer heart side effects) and fixed-duration venetoclax combined with obinutuzumab for one year. For relapsed CLL after these drugs, newer options include the non-covalent BTK inhibitor pirtobrutinib (FDA-approved December 2025) and CAR-T cell therapy with lisocabtagene maraleucel (FDA-approved 2024). Chemoimmunotherapy like FCR is now used only in rare cases.
What is the prognosis for someone with chronic lymphocytic leukemia?
Prognosis varies widely. Many patients live for decades with CLL, particularly those with mutated IGHV and favorable genetics like del(13q). Patients with del(17p)/TP53 mutations or unmutated IGHV face more aggressive disease and shorter remissions, although modern targeted therapies have dramatically improved their outlook. Some studies of first-line BTK inhibitor therapy suggest survival approaching that of an age-matched general population. CLL is still generally considered incurable, but it is increasingly manageable as a long-term condition.
How can patients and caregivers reduce the risk of complications?
The biggest risk in CLL is infection, because the abnormal B-cells crowd out healthy immune cells and produce few useful antibodies. Patients should stay current with inactivated vaccines (flu, pneumococcal, COVID-19, shingles) and avoid live vaccines while on treatment. Skin checks every 6–12 months are important because second cancers, especially skin cancers, are more common in CLL. Any new fever, persistent cough, rapidly enlarging lymph node, or unexplained bleeding deserves a prompt call to the care team.
Glossary of Medical Terms
- Apoptosis — programmed cell death; the body's controlled way of removing damaged or unwanted cells.
- B-lymphocyte (B-cell) — a white blood cell that normally makes antibodies to fight infection.
- BCL-2 — a protein that blocks apoptosis; CLL cells make too much of it, which is why they refuse to die.
- BTK (Bruton tyrosine kinase) — an enzyme inside B-cells that passes "stay alive and grow" signals; the target of BTK inhibitor drugs.
- Chronic lymphocytic leukemia (CLL) — a slow-growing blood cancer in which abnormal mature-looking B-cells build up in the blood, bone marrow, and lymph nodes.
- Clonal — all coming from a single ancestor cell, so every cancer cell is genetically identical.
- Cytopenia — a low count of any blood cell type (red cells, platelets, or white cells).
- del(17p) / TP53 mutation — loss of part of chromosome 17 (or a fault in the TP53 gene). It removes a key tumor-suppressor and predicts the worst outcomes.
- FISH (fluorescence in situ hybridization) — a lab test that uses fluorescent probes to spot missing or extra pieces of chromosomes.
- Flow cytometry / immunophenotyping — a laser-based test that identifies cells by the proteins on their surface.
- Hypogammaglobulinemia — low levels of antibodies in the blood, leading to frequent infections.
- IGHV mutation status — whether the immunoglobulin gene of the CLL cell has accumulated mutations (mutated = better prognosis) or not (unmutated = more aggressive disease).
- Lymphadenopathy — enlarged lymph nodes.
- Lymphocytosis — too many lymphocytes in the blood.
- MBL (monoclonal B-cell lymphocytosis) — a small population of CLL-like cells in the blood, below the level needed to diagnose CLL. Considered a precursor state.
- Richter transformation — a rare but serious shift of CLL into an aggressive lymphoma, usually diffuse large B-cell lymphoma.
- Smudge cell — a fragile CLL cell that breaks apart when smeared on a slide; a classic CLL fingerprint.
- Splenomegaly — an enlarged spleen.
- Thrombocytopenia — low platelet count, leading to easy bruising or bleeding.
- Tumor lysis syndrome (TLS) — a sudden release of contents from dying cancer cells that can damage the kidneys; a known risk when starting venetoclax.
Disclaimer: This article is intended for educational and informational purposes only. It is not intended to be a substitute for informed professional medical advice, diagnosis, or treatment. While the information presented here is derived from credible medical sources and is believed to be accurate and up-to-date, it is not guaranteed to be complete or error-free. See additional information.
References
- Hallek, M., Cheson, B. D., Catovsky, D., Caligaris-Cappio, F., Dighiero, G., Döhner, H., Hillmen, P., Keating, M., Montserrat, E., Chiorazzi, N., Stilgenbauer, S., Rai, K. R., Byrd, J. C., Eichhorst, B., O'Brien, S., Robak, T., Seymour, J. F., & Kipps, T. J. (2018). iwCLL guidelines for diagnosis, indications for treatment, response assessment, and supportive management of CLL. Blood, 131(25), 2745–2760. https://doi.org/10.1182/blood-2017-09-806398
- Eichhorst, B., Robak, T., Montserrat, E., Ghia, P., Niemann, C. U., Kater, A. P., Gregor, M., Cymbalista, F., Buske, C., Hillmen, P., Hallek, M., Mey, U., & ESMO Guidelines Committee. Electronic address: [email protected] (2021). Chronic lymphocytic leukaemia: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of oncology : official journal of the European Society for Medical Oncology, 32(1), 23–33. https://doi.org/10.1016/j.annonc.2020.09.019
- Eichhorst, B., Niemann, C. U., Kater, A. P., Fürstenau, M., von Tresckow, J., Zhang, C., Robrecht, S., Gregor, M., Juliusson, G., Thornton, P., Staber, P. B., Tadmor, T., Lindström, V., da Cunha-Bang, C., Schneider, C., Poulsen, C. B., Illmer, T., Schöttker, B., Nösslinger, T., Janssens, A., … GCLLSG, the HOVON and Nordic CLL Study Groups, the SAKK, the Israeli CLL Association, and Cancer Trials Ireland (2023). First-Line Venetoclax Combinations in Chronic Lymphocytic Leukemia. The New England journal of medicine, 388(19), 1739–1754. https://doi.org/10.1056/NEJMoa2213093
- Shadman M. (2023). Diagnosis and Treatment of Chronic Lymphocytic Leukemia: A Review. JAMA, 329(11), 918–932. https://doi.org/10.1001/jama.2023.1946
- Brown, J. R., Eichhorst, B., Hillmen, P., Jurczak, W., Kaźmierczak, M., Lamanna, N., O'Brien, S. M., Tam, C. S., Qiu, L., Zhou, K., Simkovic, M., Mayer, J., Gillespie-Twardy, A., Ferrajoli, A., Ganly, P. S., Weinkove, R., Grosicki, S., Mital, A., Robak, T., Osterborg, A., … Shadman, M. (2023). Zanubrutinib or Ibrutinib in Relapsed or Refractory Chronic Lymphocytic Leukemia. The New England journal of medicine, 388(4), 319–332. https://doi.org/10.1056/NEJMoa2211582
- Byrd, J. C., Hillmen, P., Ghia, P., Kater, A. P., Chanan-Khan, A., Furman, R. R., O'Brien, S., Yenerel, M. N., Illés, A., Kay, N., Garcia-Marco, J. A., Mato, A., Pinilla-Ibarz, J., Seymour, J. F., Lepretre, S., Stilgenbauer, S., Robak, T., Rothbaum, W., Izumi, R., Hamdy, A., … Jurczak, W. (2021). Acalabrutinib Versus Ibrutinib in Previously Treated Chronic Lymphocytic Leukemia: Results of the First Randomized Phase III Trial. Journal of clinical oncology : official journal of the American Society of Clinical Oncology, 39(31), 3441–3452. https://doi.org/10.1200/JCO.21.01210
- Sharman, J. P., Egyed, M., Jurczak, W., Skarbnik, A., Pagel, J. M., Flinn, I. W., Kamdar, M., Munir, T., Walewska, R., Corbett, G., Fogliatto, L. M., Herishanu, Y., Banerji, V., Coutre, S., Follows, G., Walker, P., Karlsson, K., Ghia, P., Janssens, A., Cymbalista, F., … Byrd, J. C. (2020). Acalabrutinib with or without obinutuzumab versus chlorambucil and obinutuzmab for treatment-naive chronic lymphocytic leukaemia (ELEVATE TN): a randomised, controlled, phase 3 trial. Lancet (London, England), 395(10232), 1278–1291. https://doi.org/10.1016/S0140-6736(20)30262-2
- Mato, A. R., Woyach, J. A., Brown, J. R., Ghia, P., Patel, K., Eyre, T. A., Munir, T., Lech-Maranda, E., Lamanna, N., Tam, C. S., Shah, N. N., Coombs, C. C., Ujjani, C. S., Fakhri, B., Cheah, C. Y., Patel, M. R., Alencar, A. J., Cohen, J. B., Gerson, J. N., Flinn, I. W., … Jurczak, W. (2023). Pirtobrutinib after a Covalent BTK Inhibitor in Chronic Lymphocytic Leukemia. The New England journal of medicine, 389(1), 33–44. https://doi.org/10.1056/NEJMoa2300696
- American Cancer Society. (2026). Cancer Facts & Figures 2026. Atlanta: American Cancer Society.
- National Cancer Institute. (2026). SEER Cancer Stat Facts: Leukemia - Chronic Lymphocytic Leukemia (CLL). Bethesda, MD: National Institutes of Health.
- Brown, J. R., Seymour, J. F., Jurczak, W., Aw, A., Wach, M., Illes, A., Tedeschi, A., Owen, C., Skarbnik, A., Lysak, D., Eom, K. S., Šimkovič, M., Pavlovsky, M. A., Kater, A. P., Eichhorst, B., Miller, K., Munugalavadla, V., Yu, T., de Borja, M., Ghia, P., … AMPLIFY Investigators (2025). Fixed-Duration Acalabrutinib Combinations in Untreated Chronic Lymphocytic Leukemia. The New England journal of medicine, 392(8), 748–762. https://doi.org/10.1056/NEJMoa2409804
- https://www.targetedonc.com/view/fda-approves-zanubrutinib-tablet-for-all-indications