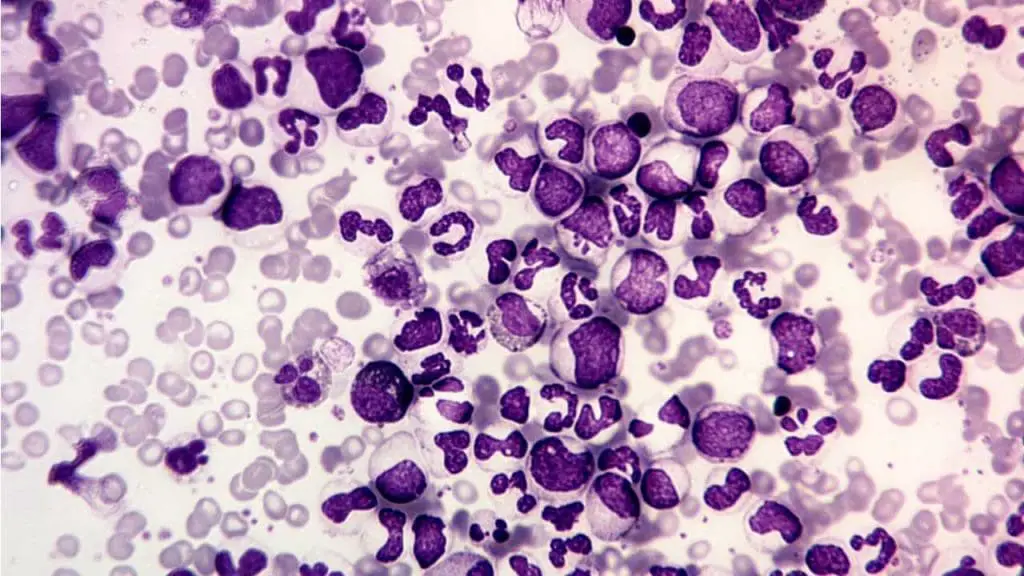
Procedure-at-a-Glance
| Step | Action | Duration |
| 1. Fixation | Fix fresh smears in Cold Formalin-Methanol (0-10°C). | 30 Seconds |
| 2. Washing | Gently rinse with running tap water and air dry. | — |
| 3. Incubation | Immerse in freshly prepared buffered substrate. | 15–30 Minutes |
| 4. Rinsing | Wash thoroughly in running water. | — |
| 5. Counterstain | Apply counterstain. | 3–10 Minutes |
| 6. Final Wash | Rinse and air dry before microscopic examination. | — |
Introduction
Leukocyte alkaline phosphatase (LAP) / Neutrophil alkaline phosphatase (NAP) stain stands as a valuable diagnostic tool, offering insights into the functionality of neutrophils, a critical component of the immune system. This enzymatic stain specifically targets alkaline phosphatase, an enzyme abundantly present in the cytoplasm of mature neutrophils. The intensity of the stain reflects the level of leukocyte alkaline phosphatase activity, providing a measure of neutrophil maturity and overall health.
Leukocyte alkaline phosphatase (LAP) staining serves as a diagnostic aid in various hematological conditions, particularly those involving neutrophil dysfunction. It plays a crucial role in differentiating between reactive neutrophilia, a normal response to infection or inflammation, or chronic myeloid leukemia which has a higher number of immature neutrophils or blast cells.
In the landscape of modern hematology, the traditional Leukocyte alkaline phosphatase (LAP) / Neutrophil alkaline phosphatase (NAP) stain has transitioned from a primary diagnostic tool to a supportive and educational one, largely superseded by definitive technologies like quantitative RT-PCR for BCR-ABL1 and multiparametric flow cytometry. While molecular testing provides the high sensitivity required to confirm a CML diagnosis and monitor treatment response at the genetic level, and flow cytometry offers an objective, automated alternative to subjective manual scoring, the Leukocyte alkaline phosphatase (LAP) stain remains globally relevant. Its enduring value lies in its affordability, rapid turnaround time, and accessibility in low-resource settings where expensive genetic testing may be unavailable, ensuring it remains both a vital clinical fallback and a foundational staple of hematology training.
Differentiation of CML from Leukemoid Reaction
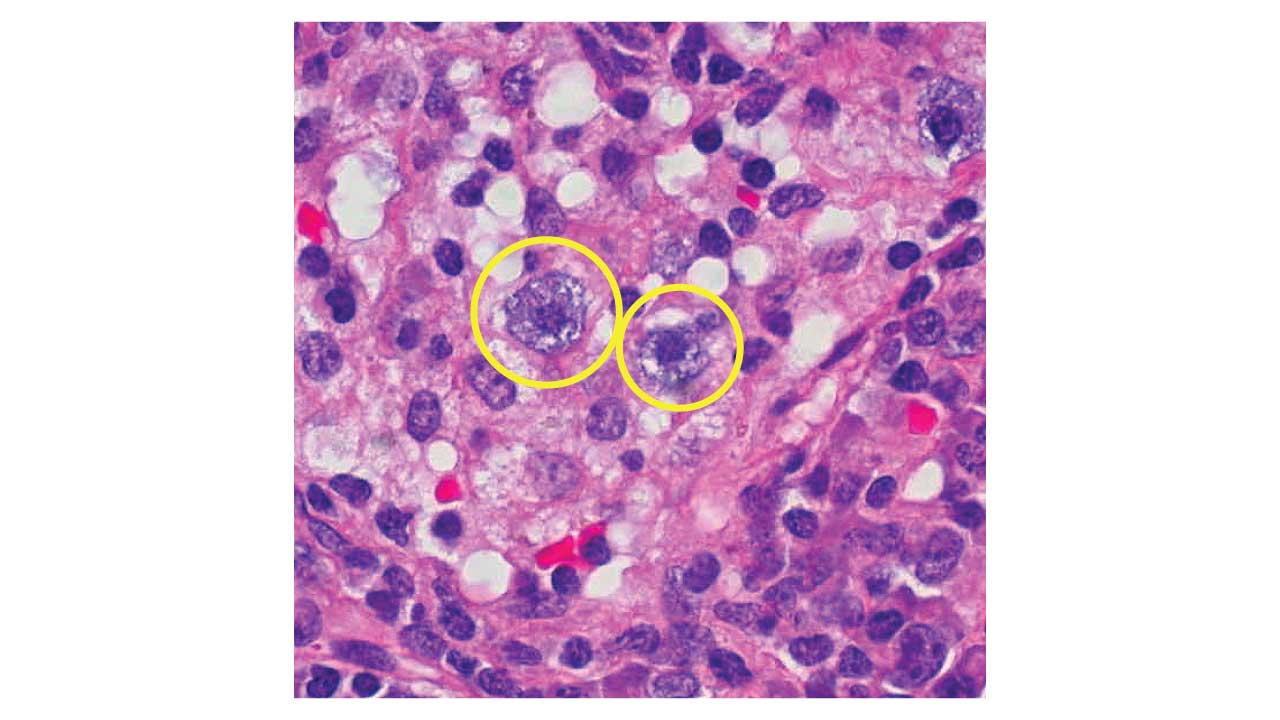
Differentiating Chronic Myeloid Leukemia (CML) from a leukemoid reaction is a critical task in hematology, as one is a myeloproliferative neoplasm and the other is a reactive, benign condition. Both can present with a significantly elevated white blood cell (WBC) count, primarily due to an increase in neutrophils, but their underlying causes, specific lab findings, and clinical courses differ.
In modern practice, the presence of the Philadelphia chromosome or the BCR-ABL1 fusion gene by cytogenetic or molecular testing is the definitive diagnostic criterion for CML, making these tests crucial for confirming the diagnosis and distinguishing it from a benign leukemoid reaction.
| Feature | Leukemoid Reaction | Chronic Myeloid Leukemia (CML) |
|---|---|---|
| Cause | Reactive (infection, inflammation, etc.) | Malignant (clonal myeloproliferative neoplasm) |
| Onset | Acute | Insidious, chronic |
| Symptoms | Related to underlying cause | Non-specific (fatigue, weight loss, night sweats), splenomegaly |
| WBC Count | Often <50,000/µL, but can be higher | Often >100,000/µL |
| Neutrophil Maturation | Predominantly mature neutrophils, clear left shift | Full spectrum of granulocytes (myelocyte bulge) |
| Blasts | Absent or <5% | <5% in chronic phase (crucial for staging) |
| Toxic Changes | Often present (granulation, Döhle bodies, vacuoles) | Usually absent |
| Basophilia/Eosinophilia | Generally absent or mild | Almost universally present and absolute |
| LAP Score (NAP Score) | Normal or Elevated | Low or Absent |
| Philadelphia Chromosome | Absent | Present in 90-95% of cases |
| BCR-ABL1 Fusion Gene | Absent | Present in virtually all cases |
| Splenomegaly | Absent or mild | Common and often significant |
Principle of Leukocyte Alkaline Phosphatase (LAP / NAP) Stain & LAP Score
Leukocyte alkaline phosphatase (LAP) / Neutrophil alkaline phosphatase (NAP) stain relies on the principle of diazotization and coupling reactions to visualize the activity of alkaline phosphatase (ALP), an enzyme abundant in mature neutrophils. The intensity of the stain reflects the level of alkaline phosphatase (ALP) activity, providing a measure of neutrophil maturity and overall health.
The staining procedure begins with the application of a diazonium salt solution to blood smears. Diazonium salts are highly reactive compounds that readily form azo dyes when coupled with aromatic compounds. In this case, the diazonium salt reacts with naphthol AS-BI phosphoric acid, a substrate for alkaline phosphatase (ALP).
Alkaline phosphatase (ALP), when present in the cytoplasm of leukocyte alkaline phosphatase-positive neutrophils, catalyzes the hydrolysis of naphthol AS-BI phosphoric acid, releasing naphthol. This naphthol then reacts with the diazonium salt, forming an insoluble blue-violet or reddish brown precipitate (depending on the dye) used within the neutrophil cytoplasm.
The intensity of the stain depends on the amount of alkaline phosphatase (ALP) present in the neutrophil. Mature neutrophils, with their high alkaline phosphatase (ALP) content, exhibit strong staining, while immature neutrophils, with lower alkaline phosphatase (ALP) activity, show weaker staining. This differential staining pattern allows for the assessment of neutrophil maturity and the identification of potential abnormalities in neutrophil function.
Method differs slightly according to the manufacturer's protocol.

Materials
- Fresh unstained peripheral blood smear slide, a positive control slide from a hamster and a negative control slide from mice
- Freshly prepared fixative: 1:9 ration of 40% formaldehyde: absolute methanol
- Naphthol AS phosphate
- 0.2 mol/L Tris buffer pH 9.0
- Fast Blue BB salt (coupling azo dye)
- Neutral red, 0.02% aqueous solution (counterstain)
- Filter paper
- Funnel
Protocol
- Prepare stock substrate solution: Dissolve 30 mg naphthol AS phosphate in 0.5 mL N,N-dimethylformamide and add 100 mL 0.2 mol/L Tris buffer.
- Prepare working buffered substrate solution: Warm 40 mL of stock substrate solution to room temperature and add 24 mg of Fast Blue BB salt. Mix well until the salt is fully dissolved. Filter the mixture using a filter paper and funnel before use.
- Fix all the blood smear slides in the freshly prepared fixative.
- Rinse the slide with slow running tap water for 2 - 3 minutes.
- Incubate the slides in working buffered substrate solution for 15 minutes.
- Rinse the slide with slow running tap water for 1 - 2 minutes.
- Air dry the slides.
- Incubate the slides in counterstain for 3 minutes.
- Rinse the slides briefly and allow to air dry.
- Mount the slide with Depex and cover the zone of morphology with a cover slip.
- This slide is now ready for viewing.
Interpretation
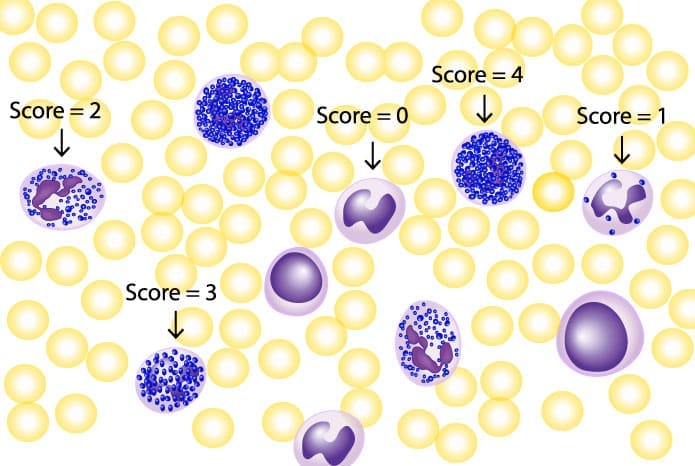
Leukocyte alkaline phosphatase (LAP) score (NAP score) indicates the maturity and activity level of neutrophils. The intensity of the stain corresponds to the enzyme activity within the neutrophils.
- Higher LAP score (NAP score) (darker staining): Indicates more mature neutrophils with higher enzyme activity, usually signifying normal function.
- Lower LAP score (NAP score) score (lighter staining): May suggest less mature neutrophils with lower enzyme activity, potentially linked to impaired function.
Leukocyte alkaline phosphatase (LAP) scores (NAP scores) are used to grade the intensity of the leukocyte alkaline phosphatase (LAP / NAP) stain on a scale of 0 to 4+. A LAP score (NAP score) of 0 indicates no staining, while a LAP score (NAP score) of 4+ indicates the strongest staining. LAP scores (NAP scores) are interpreted in the context of the patient's clinical presentation and other laboratory findings. The LAP score (NAP score) has a possible scoring of 0 - 400.
In general, a Leukocyte alkaline phosphatase (LAP) score (NAP score) of 3+ or 4+ is considered normal, indicating that the neutrophils are mature and functioning normally. A Leukocyte alkaline phosphatase (LAP) score (NAP score) of 2+ or less may suggest impaired neutrophil maturation or function. This could be due to a variety of factors, including chronic myeloid leukemia (CML).
However, interpreting Leukocyte alkaline phosphatase (LAP) score (NAP score) is always done in the context of a patient's overall clinical picture and other laboratory findings. The presence of immature neutrophils, as indicated by a low Leukocyte alkaline phosphatase (LAP) score (NAP score), must be considered in conjunction with other laboratory findings, such as a complete blood count (CBC) and a bone marrow biopsy, to make an accurate diagnosis. It's not a standalone diagnostic tool but adds valuable information when used in conjunction with other tests.
Presence of alkaline phosphatase is based on the precipitates of blue or red granules in the neutrophils. A positive control (hamster) and a negative control (mice) is used as comparison for the measurements of the Leukocyte alkaline phosphatase (LAP) score (NAP score).
Assessment of Leukocyte Alkaline Phosphatase (LAP / NAP) Scoring
- Count 100 neutrophils (including band forms)
- Rate the Leukocyte alkaline phosphatase (LAP) score (NAP score) for each neutrophil as below and add the score for 100 neutrophils.
| NAP (LAP) Score | Neutrophil staining | Interpretation |
| 0 | Absence of stained granules | Negative |
| 1 | Occasional stained granules in the cytoplasm | Positive |
| 2 | Moderate number of stained granules | Positive |
| 3 | Numerous stained granules in the cytoplasm | Strong positive |
| 4 | Numerous stained granules crowding and overlying the nucleus in the cytoplasm | Heavy positivity |
Normal range: 20 - 100.
A high LAP score (NAP score) (>100) may indicate a leukemoid reaction for e.g. severe infections such as sepsis and pneumonia and tissue necrosis such as from burns or pancreatitis.
A low LAP score (NAP score) (< 20) may indicate certain disorders for e.g. chronic myeloid leukemia, myeloproliferative neoplasm or aplastic anemia.
Clinical Significance of a High LAP score (NAP score)
- Leukemoid Reaction: This is the most common and classic reason for a high Leukocyte alkaline phosphatase (LAP) score (NAP score). As discussed previously, a leukemoid reaction is a benign, reactive increase in white blood cells (especially neutrophils and immature granulocytes) in response to:
- Severe infections: Such as sepsis, pneumonia, abscesses, or other widespread bacterial infections.
- Inflammation: Conditions like rheumatoid arthritis, vasculitis, or inflammatory bowel disease.
- Tissue necrosis/injury: Seen in conditions like burns, myocardial infarction, or pancreatitis.
- Malignancies (non-hematologic): Solid tumors can sometimes cause a paraneoplastic leukemoid reaction.
- Certain drugs: Corticosteroids can induce neutrophilia with a high Leukocyte alkaline phosphatase (LAP) score (NAP score).
- Myeloproliferative Neoplasms (MPNs) other than CML: While CML is characterized by a low Leukocyte alkaline phosphatase score (NAP score), other MPNs typically show normal or high Leukocyte alkaline phosphatase scores (NAP scores). These include:
- Polycythemia Vera (PV): A high Leukocyte alkaline phosphatase (LAP) score (NAP score) is a characteristic feature of PV, where there is an overproduction of red blood cells, white blood cells, and platelets. The neutrophils in PV are mature and have increased LAP activity. The JAK2 V617F mutation, commonly found in PV, has been shown to induce elevated Leukocyte alkaline phosphatase (LAP) scores (NAP scores).
- Essential Thrombocythemia (ET): While not as consistently high as in PV, LAP scores (NAP scores) can be normal to elevated.
- Primary Myelofibrosis (PMF): Often presents with a high LAP score (NAP score).
- Other Conditions: A high Leukocyte alkaline phosphatase score (NAP score) can also be observed in a variety of other conditions, though it's usually not the primary diagnostic marker:
- Hodgkin Lymphoma: LAP scores (NAP scores) can be elevated in some cases.
- Hairy Cell Leukemia: Paradoxically, despite being a lymphoid malignancy, high Leukocyte alkaline phosphatase scores (NAP scores) have been noted in some patients with hairy cell leukemia.
- Down Syndrome: Individuals with Down syndrome often have naturally higher Leukocyte alkaline phosphatase (LAP) scores (NAP scores).
- Pregnancy and Lactation: Leukocyte alkaline phosphatase activity naturally increases during pregnancy and lactation due to hormonal influences.
- Post-splenectomy state: After spleen removal, LAP scores (NAP scores) can be elevated.
- Stress and Trauma: Severe physical or emotional stress, or significant trauma, can trigger an increase in LAP.
- Certain Solid Tumors: Some cancers, even without a frank leukemoid reaction, can be associated with elevated LAP. There's some research suggesting it might even serve as a non-specific marker for metastasis in some cases (e.g., breast and colon cancer), though this is not a routine diagnostic application.
MPN Differentiation Summary Table
| Condition | Typical LAP/NAP Score | Genetic Hallmark |
| CML | Very Low to Absent (0–15) | BCR-ABL1 (Ph+) |
| Polycythemia Vera (PV) | High (>100) | JAK2V617F |
| Essential Thrombocythemia (ET) | Normal to Slightly High | JAK2, CALR, or MPL |
| Primary Myelofibrosis (PMF) | Variable (Normal to Low) | JAK2, CALR, or MPL |
| Leukemoid Reaction | High | None (Reactive) |
Clinical Significance of a Low LAP score (NAP score)
A very low Leukocyte alkaline phosphatase score (NAP score) is a key indicator for certain hematological conditions, most notably:
- Chronic Myeloid Leukemia (CML): This is the classic condition associated with a very low or absent LAP score (NAP score). The immature and functionally abnormal neutrophils in CML exhibit markedly reduced alkaline phosphatase activity due to the underlying genetic defect (Philadelphia chromosome/BCR-ABL1 fusion gene).
- Paroxysmal Nocturnal Hemoglobinuria (PNH): In PNH, a low LAP score (NAP score) reflects a deficiency in glycosylphosphatidylinositol (GPI)-anchored proteins, including NAP, on the surface of blood cells.
- Aplastic Anemia: This condition involves the failure of the bone marrow to produce enough blood cells, including neutrophils, and can be associated with low LAP scores (NAP scores).
- Pernicious Anemia (severe vitamin B12 deficiency): While less common as a primary indicator, severe vitamin B12 deficiency can also lead to low LAP scores (NAP scores) due to its impact on neutrophil maturation.
- Myelodysplastic Syndromes (MDS): In some cases of MDS, particularly those with higher blast percentages, lower LAP scores (NAP scores) can be observed, indicating more severe disease or impaired maturation.
Troubleshooting the LAP/NAP Stain
| Observation | Potential Cause | Corrective Action |
| All cells appear negative (Score 0) | 1. Use of EDTA anticoagulant. 2. Inactive/Outdated reagents. 3. Fixative was at room temperature. | 1. Use heparinized or fresh capillary blood. 2. Prepare fresh substrate/diazo dye. 3. Ensure fixative is chilled (0–10°C). |
| Weak or "Faded" Staining | 1. Short incubation time. 2. Reagents were too cold during incubation. 3. Smears are older than 24–48 hours. | 1. Increase incubation to full 15–30 minutes. 2. Allow reagents to reach room temp before use. 3. Always use fresh smears for best results. |
| "Muddy" Background or Precipitate | 1. Inadequate rinsing. 2. Dye was not filtered. 3. Slides dried out during incubation. | 1. Increase rinse time with gentle running water. 2. Filter the staining solution before use. 3. Ensure slides are fully immersed in the stain. |
| Neutrophil Degranulation / Poor Morphology | 1. Fixative was too warm. 2. Prolonged fixation time. 3. Forceful rinsing. | 1. Use ice-cold fixative. 2. Keep fixation strictly to 30–60 seconds. 3. Use a gentle stream of water, not direct pressure. |
| Non-specific Staining (Granules in Lymphocytes) | 1. Over-staining or high pH. 2. Contaminated counterstain. | 1. Check pH of the buffer (should be ~9.4). 2. Replace counterstain (Hematoxylin/Safranin). |
| High Background in RBCs | 1. Improper washing after fixation. 2. Poor quality of the diazo-salt. | 1. Ensure slides are washed and dried well after fixation. 2. Use a high-purity Fast Garnet GBC or equivalent. |
Frequently Asked Questions (FAQs)
What does it mean if alkaline phosphatase is high?
Having a high alkaline phosphatase (ALP) level can be concerning, but it doesn't automatically diagnose any specific condition. High alkaline phosphatase (ALP) levels can point to various possibilities, some more serious than others.
Possible Causes
- Liver problems: This is the most common cause, including conditions like:
- Blocked bile ducts: Gallstones, tumors, or inflammation can hinder bile flow, leading to high alkaline phosphatase (ALP).
- Hepatitis: Viral infections like hepatitis B or C can damage liver cells, increasing alkaline phosphatase (ALP) release.
- Cirrhosis: Scarring of liver tissue can also elevate alkaline phosphatase (ALP) levels.
- Bone disorders: Alkaline phosphatase (ALP) is also produced in bones, so conditions like:
- Paget's disease: This causes abnormal bone growth and remodeling, leading to high alkaline phosphatase (ALP).
- Fractures or bone tumors: These can temporarily increase alkaline phosphatase (ALP) as bones heal or rebuild.
- Rapid bone growth in children and adolescents: This is natural and doesn't usually require concern.
- Other possible causes: Less common contributors include:
- Certain medications: Some drugs can interfere with alkaline phosphatase (ALP) levels.
- Pregnancy: Cholestasis of pregnancy can cause a temporary rise in alkaline phosphatase (ALP).
- Heart failure: In rare cases, heart problems can affect alkaline phosphatase (ALP) levels.
Why is leukocyte alkaline phosphatase score low in Paroxysmal Nocturnal Hemoglobinuria (PNH)?
In Paroxysmal Nocturnal Hemoglobinuria (PNH), a low leukocyte Alkaline Phosphatase score reflects a fundamental abnormality in the affected cells.
PNH and GPI-anchored proteins: PNH is caused by a genetic mutation that affects a specific type of molecule called glycosylphosphatidylinositol (GPI). GPI anchors various proteins to the cell membrane, including:
- Decay-accelerating factor (DAF): Crucial for protecting red blood cells from complement-mediated destruction.
- Neutrophil alkaline phosphatase (NAP): The enzyme measured in the leukocyte alkaline phosphatase score.
Loss of GPI-anchored proteins: In PNH, the mutation disrupts the GPI anchor, causing these vital proteins to be deficient on affected blood cells, including red blood cells and neutrophils.
Consequences on leukocyte alkaline phosphatase: Since leukocyte alkaline phosphatase is a GPI-anchored protein, PNH patients have defective anchoring and reduced activity of the enzyme. This translates to a low leukocyte alkaline phosphatase score, indicating less mature and potentially less functional neutrophils.
Limitations of leukocyte alkaline phosphatase score: While a low leukocyte alkaline phosphatase score is often seen in PNH, it is not specific to the disease. Other conditions like Chronic Myelogenous Leukemia (CML) can also have low scores. Additionally, leukocyte alkaline phosphatase score is not routinely used for PNH diagnosis due to the availability of more definitive tests like flow cytometry.
Can infections affect the LAP score (NAP score)?
Yes, infections can definitely affect the Leukocyte alkaline phosphatase score (NAP score), and it is important to consider this during interpretation.
Impact of Infections on LAP score (NAP score)
- Increased score
- Infections can trigger an increased production of white blood cells, including neutrophils. This increased production, often called a leukemoid reaction, can lead to higher NAP activity and consequently, a higher score.
- Some specific infections, like viral hepatitis or Epstein-Barr virus, can also directly stimulate NAP activity in neutrophils, leading to an elevated score.
- Decreased score
- In some cases, particularly severe infections, the body's immune system may be overwhelmed, leading to immature neutrophils with lower NAP activity. This can result in a decreased score.
- Additionally, certain medications used to treat infections, like some antibiotics, can affect NAP activity and potentially lower the leukocyte alkaline phosphatase score.
Importance of Considering Infections
When interpreting a leukocyte alkaline phosphatase score, it is important to factor in the presence of any recent or ongoing infections. Knowing the type of infection and its severity can help explain potential changes in the score. Additional tests may be ordered to confirm the diagnosis and identify the specific cause.
Limitations of Leukocyte Alkaline Phosphatase Score in Infections
Leukocyte alkaline phosphatase score is not a specific test for infections. Many other factors, besides infections, can influence the score. The type and severity of the infection can also impact how it affects the leukocyte alkaline phosphatase score, making interpretation complex.
What factors can affect the LAP score (NAP score)?
Underlying Medical Conditions
- Myeloproliferative disorders: Conditions like chronic myeloid leukemia (CML) historically showed lower scores due to abnormal neutrophil production. Although more specific tests like cytogenetic analysis are preferred for CML diagnosis nowadays, LAP score (NAP score) may still be considered in specific contexts.
- Bone marrow disorders: Conditions affecting bone marrow function, like Paget's disease or bone fractures, can affect LAP score (NAP score) due to their impact on neutrophil production and maturation.
- Vitamin deficiencies: Deficiencies in vitamin B12 or folate can lead to lower scores due to their importance in neutrophil development.
- Infections: Both acute and chronic infections can influence Leukocyte alkaline phosphatase (LAP) score (NAP score) depending on the severity and type of infection. Some infections trigger higher production of immature neutrophils with lower NAP activity, while others stimulate NAP activity, leading to higher scores.
- Immune system disorders: Certain autoimmune diseases and immune deficiencies can affect neutrophil function and LAP score (NAP score).
Medications
- Corticosteroids: These drugs can stimulate neutrophil production and maturation, leading to higher scores.
- Chemotherapy drugs: These drugs can damage bone marrow and suppress neutrophil production, resulting in lower scores.
- Antibiotics: Some antibiotics can stimulate NAP activity, potentially raising the score, while others might affect neutrophil maturation and lower it.
- Methotrexate: This immunosuppressant drug can decrease NAP activity and lower the LAP score (NAP score).
- Antithyroid medications: These medications can suppress neutrophil production and lower the score.
Other Factors
- Age: The score tends to be higher in children and adolescents due to increased bone growth and neutrophil turnover.
- Pregnancy: LAP score (NAP score) naturally increases during pregnancy due to hormonal changes and placental influences.
- Diet: Certain vitamin and mineral deficiencies can affect LAP score (NAP score) indirectly through their impact on neutrophil function.
What is the difference between serum alkaline phosphatase and LAP score?
Serum alkaline phosphatase (ALP) and LAP score (NAP score) are both related to the enzyme alkaline phosphatase (ALP), but they differ in several key aspects:
What they measure
- Serum ALP: Measures the total activity of alkaline phosphatase in the blood, regardless of the cell type it originates from. This includes ALP from the liver, bones, intestines, kidneys, and placenta.
- LAP score (NAP score): Specifically measures the activity of neutrophil alkaline phosphatase (NAP), an isoenzyme of ALP found mainly in neutrophils.
Purpose of the test
- Serum ALP: Used to assess various potential issues, including liver problems (most common), bone disorders, bile duct blockages, and some cancers.
- LAP score (NAP score): Primarily used to:
- Investigate abnormal white blood cell counts (neutropenia or leukocytosis).
- Aid in differentiating certain blood disorders, especially historically for chronic myeloid leukemia (CML) - however, more specific tests like cytogenetic analysis are now preferred for CML diagnosis.
- Monitor treatment response in some cases involving neutrophil function.
Interpretation
- Serum ALP: Interpretation depends on the specific situation and other contributing factors. Increased levels can indicate various issues, while low levels are less common.
- LAP score (NAP score): Higher scores generally suggest more mature neutrophils, while lower scores may indicate less mature or impaired neutrophils. However, interpretation needs to consider other factors like age, medications, and infections.
Limitations
- Serum ALP: Not specific to a single organ or condition, requiring further investigations for definitive diagnosis.
- LAP score (NAP score): Cannot diagnose specific conditions definitively and should be interpreted alongside other clinical information and tests.
Can I use blood collected in an EDTA tube for LAP staining?
No. EDTA acts as a chelating agent that inhibits alkaline phosphatase activity. For accurate results, use heparinized blood or fresh finger-prick (capillary) blood smears.
Why is the LAP score high in pregnancy?
Leukocyte alkaline phosphatase (LAP) activity naturally increases due to hormonal changes, specifically increased estrogen and progesterone levels during the second and third trimesters.
What is a "Normal" LAP score?
While ranges vary by lab, a typical normal range is between 20 and 100. Scores below 13 are highly suggestive of CML.
How many cells must be counted for a valid LAP score?
You must evaluate 100 consecutive mature neutrophils and bands. Eosinophils and basophils should be ignored as they do not contain the enzyme.
Is the LAP score useful for diagnosing AML?
Generally, no. The Leukocyte alkaline phosphatase (LAP) stain is specific to the granulocytic series (neutrophils). For Acute Myeloid Leukemia (AML), stains like Myeloperoxidase (MPO) or Sudan Black B are preferred.
Glossary of Related Medical Terms
- Azo-dye: A class of synthetic dyes used in this stain that reacts with the phosphate released by the enzyme to form a colored precipitate.
- Constitutively Active: Refers to an enzyme (like the one produced by the BCR-ABL gene in CML) that is always "on," leading to uncontrolled cell growth.
- GPI-Anchor: (Glycosylphosphatidylinositol) A "tether" that holds the LAP enzyme to the cell membrane. Deficiencies in this anchor (as seen in PNH) lead to low LAP scores.
- Leukemoid Reaction: A benign, temporary increase in white blood cell count (often >50,000/µL) mimicking leukemia, usually caused by severe infection.
- Left Shift: The presence of increased proportions of immature neutrophils (bands, myelocytes) in the peripheral blood.
- Philadelphia Chromosome (Ph+): The genetic hallmark of CML [t(9;22)] which results in the reduction of the LAP enzyme in neutrophils.
Disclaimer: This protocol is intended for informational purposes only and may need to be modified depending on the specific laboratory procedures and patient circumstances. Always consult with a qualified healthcare professional for guidance. See additional information.
References
- Bain BJ, Bates I, Laffan MA. Dacie and Lewis Practical Haematology: Expert Consult: Online and Print (Elsevier) 12th Edition. 2016.
- Sugano, K., Hashi, K., Goto, M., Nishi, K., Maeda, R., Kono, K., Yamamoto, M., Okada, K., Kaga, S., Miwa, K., Mikami, T., & Masauzi, N. (2017). Semi-Automatic Rating Method for Neutrophil Alkaline Phosphatase Activity. Journal of clinical laboratory analysis, 31(1), e22022. https://doi.org/10.1002/jcla.22022
- Kondo, T., Tasaka, T., Tomioka, N., Sano, F., Tokunaga, H., Suemori, S., Tsujioka, T., Matsuhashi, Y., Nakanishi, H., Wada, H., Tohyama, K., & Sugihara, T. (2016). Low neutrophil alkaline phosphatase score is a new aspect of calreticulin-mutated myeloproliferative neoplasms. SpringerPlus, 5(1), 1146. https://doi.org/10.1186/s40064-016-2829-6
- Majumdar, G., Hunt, M., & Singh, A. K. (1991). Use of leucocyte alkaline phosphatase (LAP) score in differentiating malignant from benign paraproteinaemias. Journal of clinical pathology, 44(7), 606–607. https://doi.org/10.1136/jcp.44.7.606
- Majumdar, G., Hunt, M., & Singh, A. K. (1991). Use of leucocyte alkaline phosphatase (LAP) score in differentiating malignant from benign paraproteinaemias. Journal of clinical pathology, 44(7), 606–607. https://doi.org/10.1136/jcp.44.7.606
- Lipshitz, J., Limaye, S., & Patel, D. (2010). Leukocyte alkaline phosphatase score correlation with bone marrow blast percentage in myelodysplastic syndrome. Acta haematologica, 124(3), 179–181. https://doi.org/10.1159/000320272
- Basquiera, A. L., Fassetta, F., Soria, N., Barral, J. M., Ricchi, B., & García, J. J. (2007). Accuracy of leukocyte alkaline phosphatase score to predict JAK2 V617F mutation. Haematologica, 92(5), 704–705. https://doi.org/10.3324/haematol.10991