Key Takeaways
Leukemia is a cancer of the blood and bone marrow. It affects the cells responsible for fighting infection and carrying oxygen throughout the body.
- How does it occur? ▾: Normally, blood cells develop from stem cells in the bone marrow. In leukemia, the development goes wrong, leading to the production of abnormal cells called blasts. These blasts crowd out healthy blood cells leading to:
- Anemia (decreased RBCs) → Fatigue, pallor.
- Neutropenia (decreased functional WBCs) → Recurrent infections.
- Thrombocytopenia (decreased platelets) → Easy bruising and mucosal bleeding.
- Signs and Symptoms ▾:
- Feeling tired and weak
- Frequent infections, fever
- Easy bruising and bleeding
- Swollen lymph nodes
- Types of leukemia ▾:
- How is it diagnosed? ▾:
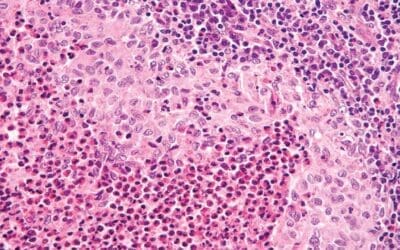
- Morphology & Cytochemistry: Identifying blasts and specific features like Auer rods (AML) on a bone marrow aspirate.
- Immunophenotyping (Flow Cytometry): Using CD markers (e.g., CD33 for myeloid, CD19/20 for B-lymphoid, cCD3 for T-lymphoid) to define lineage.
- Cytogenetics & Molecular Genetics: Detecting driver mutations and translocations (e.g., Philadelphia chromosome $t(9;22)$ in CML; NPM1 in AML).
- How is it treated? ▾:
- Phased Treatment: Acute leukemia therapy is split into Induction (to reach remission), Consolidation (to clear residual cells), and Maintenance (to prevent relapse).
- Targeted Therapy: Shifting away from broad chemo toward precision agents like TKIs (Imatinib), FLT3 inhibitors, and CAR-T cell therapy.
- MRD (Measurable Residual Disease): The use of flow cytometry or NGS to detect 1 in $10^5$ or $10^6$ leukemic cells. MRD status is the single most important predictor of clinical relapse.
*Click ▾ for more information
Introduction
Leukemia is a group of cancers that affect the blood and bone marrow. It arises when immature blood cells, called blasts, multiply uncontrollably in the bone marrow, crowding out healthy blood cell production. These abnormal cells often don’t function properly, leading to various health problems.
A Brief History of Leukemia Research and Understanding
While traces of leukemia were potentially observed in ancient times, our scientific understanding emerged much later. Here’s a glimpse into the key milestones:
Early Observations (18th-19th Centuries)
- 1700s: First descriptions of unusual blood findings resembling leukemia emerge.
- 1827: French Physician Jean Cruveilhier coins the term “leukemia” based on the white blood cell count observed in patients.
- 1845: Scottish physician John Hughes Bennett first uses the term “leucocythemia” to describe the disease.
- Mid-19th century: Rudolf Virchow distinguishes between lymphatic and myelogenous forms of leukemia based on the affected cell type.
Understanding the Disease (20th Century)
- 1900s: Advances in microscopy and blood analysis facilitate diagnosis and differentiation of leukemia subtypes.
- 1920s and 1930s: X-ray therapy emerges as an initial treatment option.
- 1940s: Nitrogen mustards, derived from chemical warfare agents, become the first chemotherapy drugs used for leukemia.
- 1950s: Bone marrow transplantation shows promise as a potential cure.
- 1960s and 1970s: Development of more effective chemotherapy regimens improves survival rates.
- 1980s and 1990s: Identification of chromosomal abnormalities and genetic mutations associated with specific leukemia types.
- Late 20th century: Targeted therapies based on specific genetic mutations begin to emerge.
Modern Era (21st Century)
- Focus on personalized medicine: Tailoring treatment based on individual patients’ genetic make-up and leukemia characteristics.
- Advances in immunotherapy: Harnessing the immune system to fight cancer cells.
- Gene therapy and stem cell-based therapies: Exploring approaches to repair or replace defective genes or eliminate malignant cells.
Continued progress
The field of leukemia research continues to rapidly evolve, with ongoing exploration of new therapies, early detection methods, and improved supportive care. Today, the outlook for many leukemia patients has significantly improved, thanks to the dedication of researchers and advancements in our understanding of this complex disease.
Global burden of leukemia
Leukemia poses a significant global health challenge, impacting individuals, families, and healthcare systems worldwide.
- Incidence: Globally, in 2020, 474,519 new cases of leukemia were estimated, constituting roughly 2.5% of all new cancer diagnoses. The exact number varies depending on various factors like age, sex, and geographical location. Although incidence might not be increasing dramatically, regional disparities exist. Countries with lower socioeconomic development tend to have higher rates, likely due to limited access to early diagnosis and preventative measures.
- Prevalence: Due to improved treatment options, the number of people living with leukemia is rising. Estimates suggest over 4 million individuals currently have the disease globally. This growing prevalence, especially in aging populations, adds to the healthcare burden and resource utilization.
- Mortality: In 2020, leukemia remained a significant cause of cancer-related deaths, claiming 311,594 lives, accounting for 3.1% of all cancer deaths. While mortality rates have been decreasing slowly in some regions due to advancements in diagnosis and treatment, disparities exist between developed and developing nations.
- Childhood leukemia: Remains the most common cancer in children under five years old, leading to high mortality rates in regions with limited access to pediatric cancer care.
- Type variations: Acute lymphoblastic leukemia (ALL) is the most prevalent type in children, while acute myeloid leukemia (AML) is more common in adults. Chronic lymphocytic leukemia (CLL) is the most common adult type overall.
Classification of Leukemia
Leukemia classification helps healthcare professionals understand the specific type and characteristics of a patient’s disease, guiding treatment decisions and predicting potential outcomes.
By Cell Type
- Lymphoid leukemias (lymphoblastic): These arise from abnormal lymphoid progenitor cells, which normally develop into infection-fighting B or T lymphocytes. They are more common in children and young adults.
- Myeloid leukemias (myelocytic, myelogenous): These originate from abnormal myeloid progenitor cells, which typically mature into red blood cells, white blood cells (other than lymphocytes), and platelets. They are more prevalent in adults and older adults.
By Course
- Acute leukemias: These progress rapidly, with blasts multiplying quickly and disrupting healthy blood cell production within weeks or months.
- Chronic leukemias: These develop slowly over months or years, with an accumulation of abnormal cells but often maintaining some ability to produce mature blood cells.
Subtypes
Acute Lymphoblastic Leukemia (ALL)
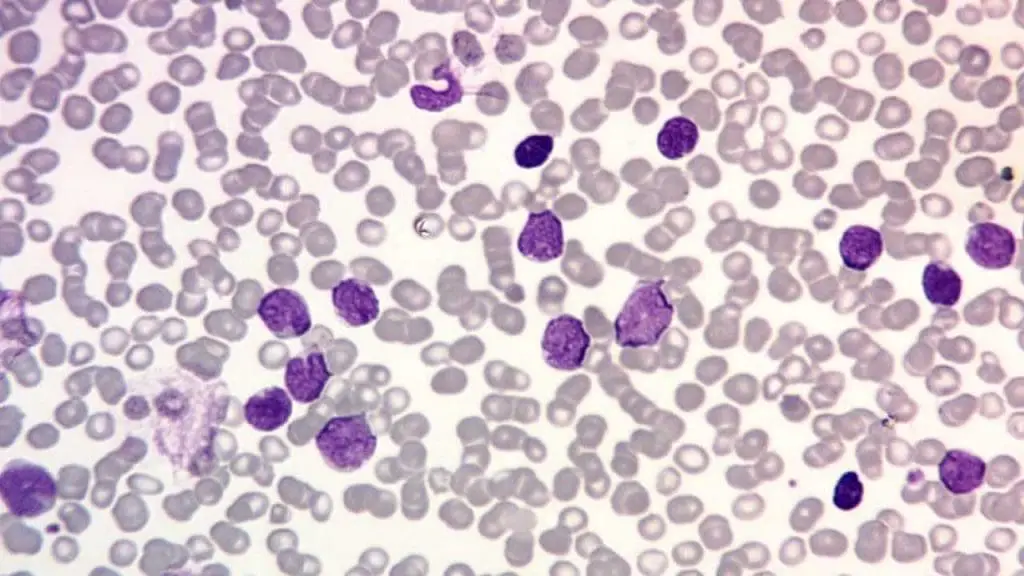
- B-cell ALL: More common in children, characterized by immature B-lymphocytes in the blood and bone marrow. Subtypes include:
- Early B-cell ALL: Diagnosed at an early stage of B-cell development.
- Late B-cell ALL: Diagnosed at a later stage of B-cell development.
- Philadelphia chromosome-positive (Ph+): Associated with a specific genetic abnormality and requires targeted therapy.
- Burkitt-type ALL: Aggressive subtype with unique features.
- T-cell ALL: Less common, affects T-lymphocytes. Subtypes include:
- Early T-cell precursor ALL: Affects very early T-cell precursors.
- Cortical T-cell ALL: Affects more mature T-cell precursors.
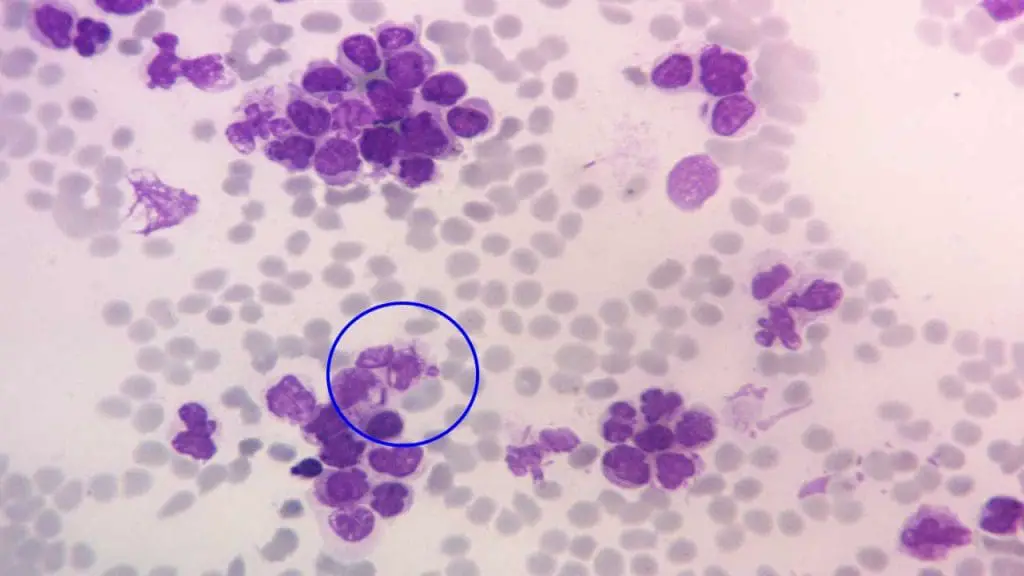
Acute Myeloid Leukemia (AML)
- Classification based on French-American-British (FAB) system: Categorizes AML into subtypes based on microscopic features of the blasts. For e.g.
- Acute myeloblastic leukemia with minimal differentiation (M0)
- Acute promyelocytic leukemia (APL) (M3)
- Genetic-based classification: Newer system identifies specific genetic mutations that influence treatment and prognosis. For e.g.
- AML with recurrent genetic abnormalities
- AML with myelodysplasia-related changes
- Therapy-related AML
Comparative Nuances: Pediatric vs. Adult Acute Leukemia
The following table summarizes the key distinctions across Acute Lymphoblastic Leukemia (ALL) and Acute Myeloid Leukemia (AML).
| Feature | Pediatric Nuance (<18 years) | Adult Nuance (>40 years) |
| Most Common Type | ALL (~80% of cases) | AML (incidence increases with age) |
| Origin of Disease | Almost always de novo (arising fresh). | Frequently secondary (arising from MDS or prior chemotherapy). |
| Common ALL Cytogenetics | t(12;21) ETV6-RUNX1 and High Hyperdiploidy (Favorable). | t(9;22) BCR-ABL1 (Philadelphia+) and Hypodiploidy (Unfavorable). |
| Common AML Mutations | KMT2A (MLL) and NUP98 rearrangements are more frequent. | NPM1, DNMT3A, and TET2 mutations (associated with clonal hematopoiesis). |
| Genetic Landscape | “Quiet” genome with few somatic mutations but large structural fusions. | Higher mutational burden with complex, age-related epigenetic changes. |
| Treatment Response | Highly sensitive to chemotherapy; excellent clearance of MRD. | Higher rates of chemo-resistance and treatment-related toxicity. |
| Prognosis (5-yr Survival) | Excellent: >90% for ALL; ~65-70% for AML. | Guarded: ~30-40% for ALL; ~15-25% for AML (varies by age). |
Chronic Lymphocytic Leukemia (CLL)
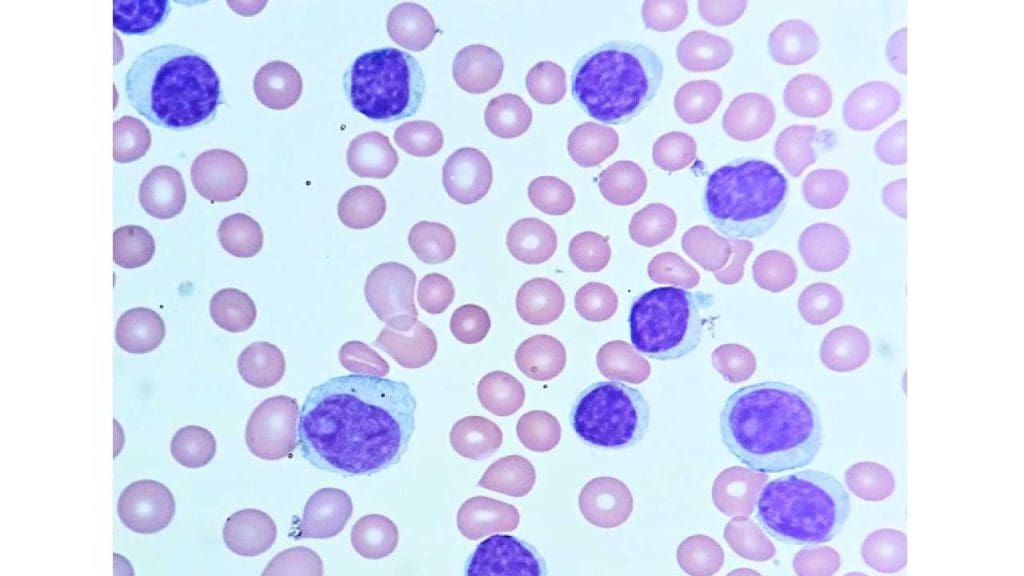
- Most common chronic leukemia, affecting mature B-lymphocytes.
- Slow progression, often diagnosed in older adults.
- Subtypes based on genetic characteristics and disease course.
Chronic Myeloid Leukemia (CML)
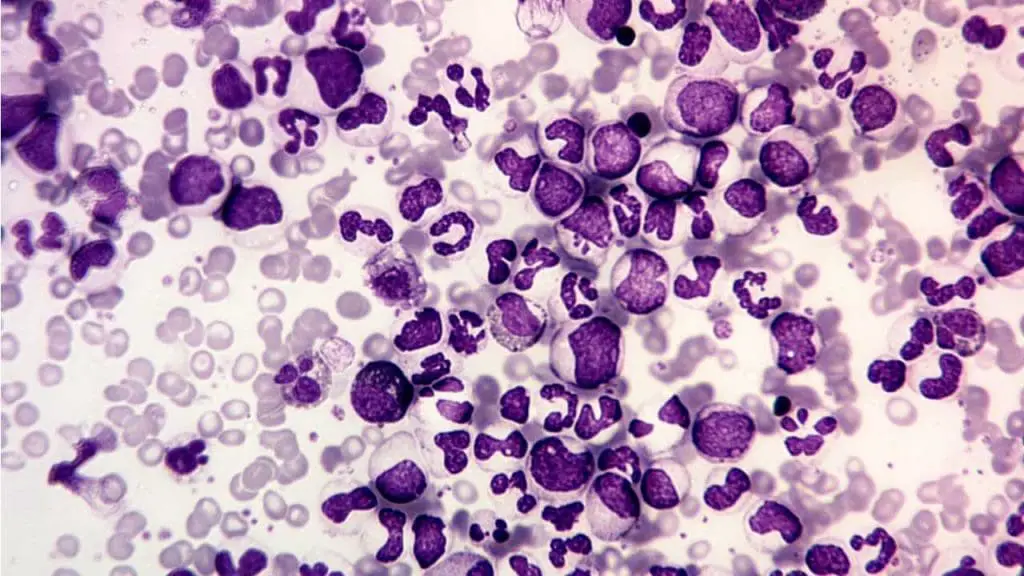
- Characterized by the Philadelphia chromosome, causing uncontrolled growth of myeloid progenitor cells.
- Chronic phase with manageable symptoms but can progress to accelerated and blast crisis phases without treatment.
- Targeted therapy available for long-term management.
Less Common Types
- Hairy cell leukemia: Slow-growing leukemia affecting B-cells with hair-like projections.
- Adult T-cell leukemia-lymphoma (ATLL): Aggressive T-cell leukemia associated with human T-lymphotropic virus type 1 (HTLV-1) infection.
- Other rare types: Myelodysplastic syndrome (MDS) and myeloproliferative neoplasms (MPN) can sometimes transform into acute leukemias.
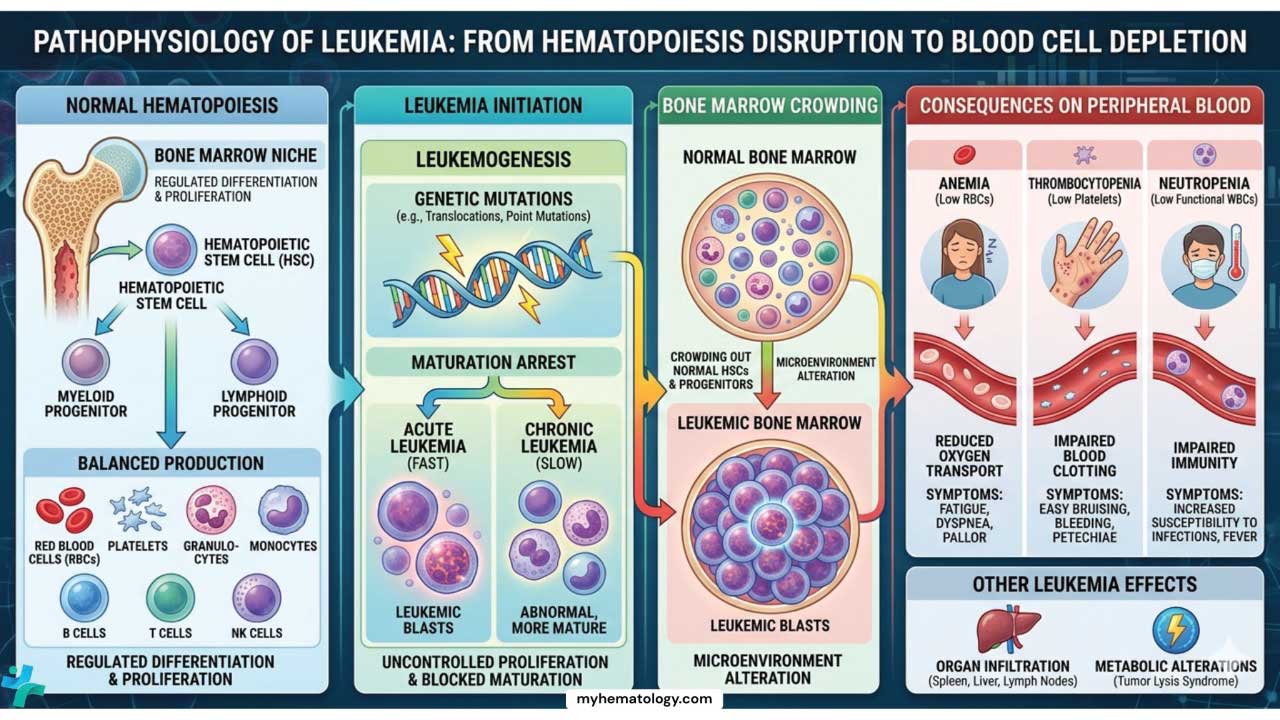
Leukemia Pathophysiology
Normal blood cell development in the bone marrow (Hematopoiesis)
The journey of a blood cell begins with the versatile hematopoietic stem cell residing in the bone marrow. This remarkable cell acts as the mother of all blood cells, possessing the unique ability to both duplicate itself and differentiates into various specialized cells. Its initial differentiation creates two distinct paths: the myeloid and lymphoid lineages through the common myeloid progenitors and common lymphoid progenitors, respectively.
The lymphoid pathway leads to the development of immune warriors like lymphocytes and natural killer cells. The lymphoid progenitor cells mature into lymphoblasts, which eventually become the small lymphocytes patrolling our body, constantly vigilant against invaders.
Meanwhile, the myeloid progenitor embarks on a diverse journey giving rise to several crucial cell types. One path leads to the production of red blood cells (erythrocytes). Through a series of stages, erythroblasts mature into reticulocytes and finally functional erythrocytes, responsible for oxygen transport throughout the body.
Another branch of the myeloid lineage produces platelets, essential for blood clotting. Megakaryocytes, formed from megakaryoblasts, fragment into thousands of tiny platelets, ready to seal any breach and prevent bleeding.
Finally, the myeloid progenitor also gives rise to different types of white blood cells known as granular leukocytes (neutrophils, eosinophils, and basophils) and monocytes. Monocytes mature into macrophages, acting as scavengers and defenders within our tissues.
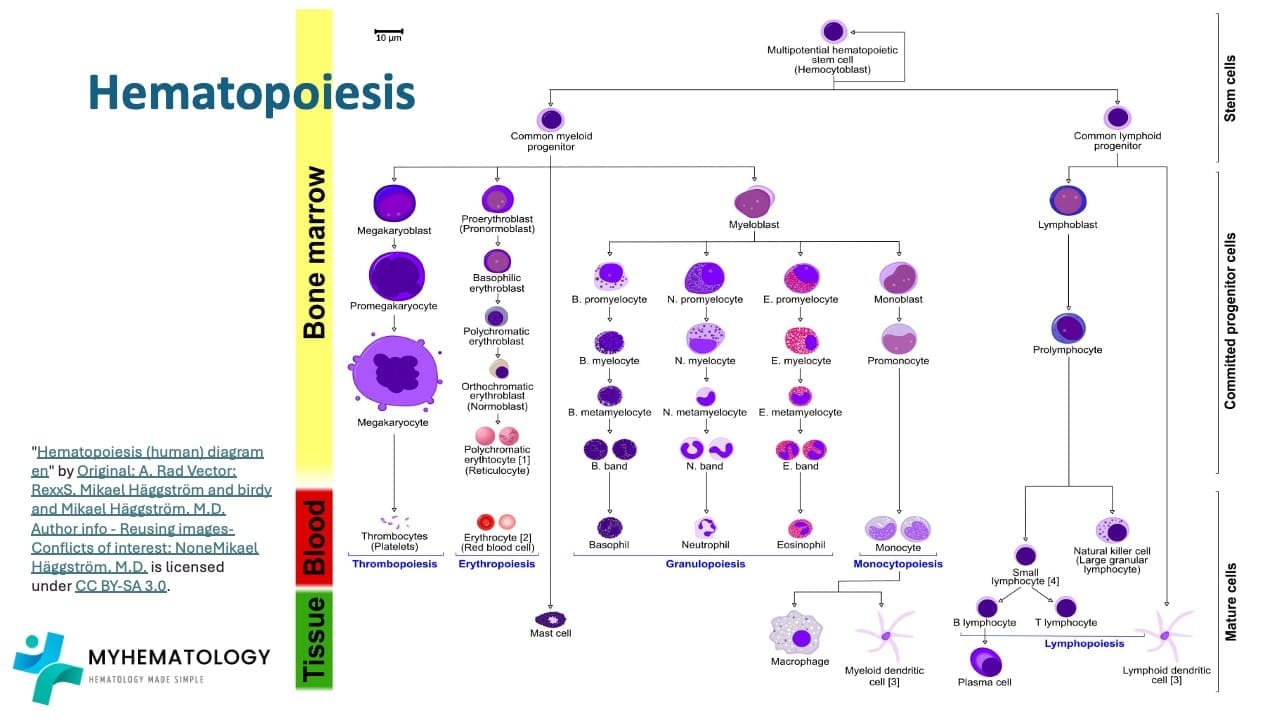
Hematopoiesis Disruption
Leukemia isn’t just an overproduction of white blood cells; it arises from a complex interplay between genetic and molecular abnormalities that disrupt the delicate balance of blood cell production.
- Chromosomal Abnormalities: Translocations, deletions, and duplications in chromosomes can lead to uncontrolled cell growth and disrupt normal gene function. For example, the Philadelphia chromosome, a hallmark of chronic myeloid leukemia (CML), results in the fusion of two genes, promoting uncontrolled proliferation.
- Mutations: Point mutations in specific genes can activate oncogenes (promoting cell division) or inactivate tumor suppressor genes (controlling cell growth), leading to uncontrolled leukemic cell proliferation. Mutations in genes like FLT3 in AML or TP53 in CLL are examples.
- Epigenetic Alterations: Chemical modifications on DNA or histones can silence tumor suppressor genes or activate oncogenes, contributing to leukemogenesis.
Domino Effect on Bone Marrow
These genetic and molecular aberrations wreak havoc on the bone marrow, the factory responsible for blood cell production.
- Uncontrolled Proliferation: Abnormal cells, driven by genetic mutations, divide uncontrollably, crowding out healthy stem cells and disrupting their differentiation into mature blood cells.
- Impaired Differentiation: Mutations can impair the normal maturation process of blood cells, leading to the production of immature and dysfunctional blasts.
- Disrupted Microenvironment: The bone marrow environment, crucial for supporting healthy blood cell development, can be altered by abnormal cells, further hindering healthy blood cell production.
Consequences of Blood Cell Depletion
The decreased production of healthy blood cells leads to a cascade of devastating consequences.
- Infections: Reduced white blood cells (neutrophils, lymphocytes) weaken the immune system, making individuals susceptible to life-threatening infections.
- Anemia: Decreased red blood cells (erythrocytes) lead to reduced oxygen delivery throughout the body, causing fatigue, weakness, and shortness of breath.
- Bleeding: Low platelet counts impair blood clotting, increasing the risk of easy bruising and bleeding.
- Organ Damage: Infiltrating leukemic cells can damage vital organs like the liver, spleen, and lymph nodes, leading to organ dysfunction.
Epidemiology and Risk Factors
Leukemia, while affecting individuals of all ages and backgrounds, exhibits distinct patterns in its occurrence, offering valuable insights into potential causes.
Age and Gender Distribution
- Childhood Leukemia: Acute lymphoblastic leukemia (ALL) is the most common childhood cancer, predominantly affecting children under five. Acute myeloid leukemia (AML) is less frequent but also occurs in children.
- Adult Leukemia: CLL and AML become more prevalent with age, with the highest rates observed in individuals over 65. CML typically affects adults between 45 and 55.
- Gender Variations: ALL shows a slight male predominance in childhood, while CLL is more common in men throughout life. Conversely, AML affects men and women relatively equally.
Geographic and Ethnic Disparities
- Incidence Variations: Rates of certain leukemia types vary geographically. For example, ALL and CML show higher rates in developed nations, while AML has a higher incidence in some developing countries.
- Ethnic Differences: Genetic variations and environmental exposures contribute to ethnic disparities in leukemia risk. For instance, certain ethnicities demonstrate higher susceptibility to specific genetic mutations associated with leukemia.
Genetic Predisposition and Family History
- Inherited Mutations: Some individuals inherit genetic mutations that increase their risk of developing leukemia. Examples include Down syndrome, which elevates ALL risk, and specific gene mutations associated with familial CML.
- Family History: Having a close relative with leukemia slightly increases an individual’s risk, suggesting a possible genetic component.
Environmental Risk Factors
- Radiation Exposure: High-dose radiation exposure, either through medical procedures, occupational hazards, or accidents, can increase the risk of developing various leukemias.
- Chemicals: Exposure to certain chemicals like benzene, used in some industrial processes, has been linked to an increased risk of AML.
- Other Factors: Some studies suggest potential associations between specific infections and the development of certain leukemia types, but the evidence is still evolving.
Lifestyle Choices
- Smoking: Cigarette smoking is a well-established risk factor for AML, with increased risk proportional to smoking intensity and duration.
Clinical Presentation
Leukemia often presents with non-specific symptoms that can be easily overlooked. However, recognizing these early warning signs is crucial for prompt diagnosis and timely intervention.
General Signs and Symptoms
- Fatigue: This is the most common symptom, often described as persistent tiredness, weakness, and lack of energy. It arises from decreased oxygen delivery due to anemia caused by low red blood cells.
- Fever: Recurrent or persistent fevers can indicate infections due to a compromised immune system caused by low white blood cell counts.
- Frequent Infections: Increased susceptibility to infections like mouth sores, sore throat, and respiratory issues is a common sign due to impaired immune function.
- Easy Bruising and Bleeding: Low platelet counts can lead to easy bruising, nosebleeds, and bleeding gums, indicating impaired blood clotting.
- Lymphadenopathy: Swollen lymph nodes in the neck, armpits, or groin might occur due to the accumulation of abnormal cells within the lymphatic system.
Specific Signs and Symptoms
- Swollen Abdomen: Enlarged spleen or liver due to infiltration of leukemic cells might cause abdominal discomfort or fullness.
- Bone Pain: Infiltration of leukemic cells into the bone marrow can lead to bone pain and tenderness.
- Headaches and Seizures: Infiltration of the central nervous system by leukemic cells can cause neurological symptoms like headaches and seizures in rare cases.
- Skin Rashes: Specific types of leukemia, like hairy cell leukemia, might present with characteristic skin rashes.
Variation based on Type and Severity
- Acute Leukemias: Symptoms often progress rapidly and are more pronounced due to the aggressive nature of the disease.
- Chronic Leukemias: Symptoms may be subtle and develop slowly, sometimes even being absent initially, making early detection challenging.
Importance of Early Diagnosis and Screening
- Early diagnosis allows for prompt treatment initiation, improving the chance of successful remission and long-term survival.
- Early intervention can minimize complications like infections and bleeding.
- Screening programs targeting high-risk groups, though not currently standard practice for most leukemia types, are being explored for their potential in early detection.
Investigations and Diagnosis
Accurately diagnosing leukemia and understanding its specific characteristics are essential for guiding treatment decisions and predicting potential outcomes.
Diagnostic Workup
The diagnosis of leukemia has evolved from a purely morphological assessment to an integrated approach combining genetics, immunophenotype, and clinical history. For a comprehensive diagnostic workup, especially in the context of the WHO 5th Edition (2022) and ICC (2022) classifications, the following framework is essential.
Phase 1: Initial Screening & Morphology
The journey usually begins with a symptomatic patient (fever, fatigue, bleeding) or an incidental finding on routine labs.
- Complete Blood Count (CBC) with Differential: Look for “leukemic hiatus” (blasts and mature cells without intermediate stages), anemia, and thrombocytopenia.
- Peripheral Blood Smear (PBS):
- Blasts: Identification of lymphoblasts (scant cytoplasm, fine chromatin) vs. myeloblasts (larger, moderate cytoplasm, prominent nucleoli).
- Pathognomonic Signs: Auer rods in myeloblasts (confirming AML).
- In chronic leukemias (CML/CLL), the smear shows a spectrum of mature cells rather than a “blast crisis” (unless transformed).
Phase 2: The Confirmatory Procedure
Bone Marrow Aspiration and Trephine Biopsy remain the gold standard for definitive diagnosis and for obtaining samples for ancillary testing.
- Aspirate: Best for morphology and blast counting.
- Biopsy: Best for assessing cellularity, architecture, and identifying “dry taps” (often seen in Hairy Cell Leukemia or myelofibrosis).
- Cytochemistry: While largely superseded by flow cytometry, Myeloperoxidase (MPO) and Sudan Black B (SBB) are still useful for confirming myeloid lineage in resource-limited settings.
Phase 3: The Modern “Diagnostic Triad”
To classify leukemia according to current global standards, three pillars are required:
Immunophenotyping (Flow Cytometry)
This identifies the “Cluster of Differentiation” (CD) markers on the cell surface to determine lineage.
- Myeloid: CD13, CD33, CD117, MPO.
- B-Lymphoid: CD19, CD20, CD22, CD10, cCD79a.
- T-Lymphoid: CD2, CD3 (cytoplasmic/surface), CD5, CD7.
High-Yield CD Markers in Leukemia Immunophenotyping
| Lineage | Primary Markers | Secondary/Specific Markers | Clinical Significance |
| Stem Cell / Pan-Leukemic | CD34, HLA-DR, TdT | CD45 (LCA) | CD34 indicates immaturity (blasts); TdT is highly specific for lymphoblasts (ALL). |
| Myeloid (AML) | CD13, CD33, MPO | CD117 (c-kit), CD11b, CD15 | MPO (Myeloperoxidase) is the “gold standard” for myeloid commitment. |
| Monocytic (AML-M4/M5) | CD14, CD64 | CD11c, CD36 | High expression of CD14/CD64 helps differentiate monocytic from pure granulocytic AML. |
| B-Lymphoid (B-ALL/CLL) | CD19, cCD79a, CD22 | CD10, CD20, CD5, CD23 | CD10 (CALLA) is a key prognostic marker in pediatric ALL. CD5/CD23 co-expression is hallmark for CLL. |
| T-Lymphoid (T-ALL) | cCD3, CD7 | CD2, CD5, CD4, CD8 | Cytoplasmic CD3 (cCD3) is the most lineage-specific marker for T-cells. |
| Megakaryocytic (AML-M7) | CD41, CD61 | CD42b | Essential for diagnosing rare acute megakaryoblastic leukemia. |
| Erythroid (AML-M6) | Glycophorin A | CD71 (Transferrin receptor) | Used when the lineage appears purely erythroid on morphology. |
Cytogenetics (Karyotyping & FISH)
Detects structural chromosomal abnormalities.
- Recurrent Translocations: t(15;17) for APL, t(8;21) for AML, and t(9;22) (Philadelphia chromosome) for CML/ALL.
- FISH: Useful for rapid detection of specific fusions when conventional karyotyping takes too long or fails.
Molecular Genetics (NGS & PCR)
Identifies point mutations that define risk and target therapy.
- Key Mutations: NPM1, FLT3-ITD, CEBPA, ASXL1, and IDH1/2.
- High-Yield: The presence of an NPM1 mutation can now define AML even if the blast count is below 20% in certain classifications.
Current Standards: The “Blast Threshold” Debate
A major update in 2022 involved the blast percentage required for an AML diagnosis. Traditionally set at 20%, the new guidelines allow for lower counts if specific genetic drivers are present.
Comparison: WHO 2022 vs. ICC 2022
| Feature | WHO 5th Ed (2022) | ICC (2022) |
| Genetic Drivers | Eliminates 20% threshold for defining mutations (except BCR::ABL1 & CEBPA). | Sets a 10% threshold for most recurrent genetic abnormalities. |
| MDS/AML Category | Does not use this term; maintains “MDS with increased blasts.” | Introduced MDS/AML for cases with 10–19% blasts. |
| Secondary AML | Classified as “Myeloid neoplasms, secondary.” | Uses “Diagnostic qualifiers” for prior MDS or therapy. |
Supportive & Staging Investigations
Beyond the marrow, systemic assessment is vital:
- Lumbar Puncture: Mandatory in ALL to rule out CNS involvement.
- Biochemistry: LDH (marker of cell turnover), Urate (risk of Tumor Lysis Syndrome), and Coagulation screen (PT/APTT/Fibrinogen) to screen for DIC, especially in APL.
- Imaging: Chest X-ray or CT to look for mediastinal masses (common in T-ALL) or hepatosplenomegaly.
Minimal Residual Disease (MRD) Testing
Minimal Residual Disease (MRD) increasingly referred to as Measurable Residual Disease represents the small volume of leukemic cells that remain in a patient’s body after they have achieved “Complete Remission” (CR) by traditional morphologic standards (i.e., <5% blasts in the bone marrow).
MRD is the single strongest predictor of relapse in both pediatric and adult acute leukemias.
Traditional microscopy can detect 1 leukemic cell among 100 normal cells (10⁻²). Modern MRD technologies are significantly more sensitive, allowing us to find “the needle in the haystack.”
| Method | Sensitivity | Primary Targets | Key Advantages |
| Flow Cytometry (MFC) | 10⁻⁴ to 10⁻⁵ | Leukemia-Associated Immunophenotypes (LAIP) | Fast (24h), applicable to >90% of patients. |
| RT-qPCR | 10⁻⁵ to 10⁻⁶ | Fusion transcripts (e.g., BCR::ABL1, PML::RARA) | Highly standardized, “Gold Standard” for specific translocations. |
| Next-Gen Sequencing (NGS) | 10⁻⁶ | Ig/TCR rearrangements or gene mutations (NPM1, FLT3) | Highest sensitivity; can detect clonal evolution/new mutations. |
MRD testing is no longer just for research; it directly dictates the “intensity” of a patient’s treatment plan.
- Risk Stratification: In Pediatric ALL, MRD status at Day 29 (end of induction) is the primary factor used to decide if a child needs more intensive chemotherapy or a stem cell transplant.
- Early Warning System: In CML, a “Molecular Relapse” (rising BCR::ABL1 levels) often occurs months before cells are visible under a microscope, allowing for a timely switch in Tyrosine Kinase Inhibitor (TKI) therapy.
- Transplant Decisions: For AML patients in their first CR, those who remain MRD-positive often derive the greatest benefit from an Allogeneic Stem Cell Transplant compared to those who are MRD-negative.
Current 2025/2026 Guidelines (ELN-DAVID)
According to the latest ELN-DAVID (2025/2026) consensus update, several shifts have occurred in how we handle MRD:
- Specimen Quality: “First-pull” bone marrow aspirates are mandatory to avoid hemodilution, which can lead to false-negative MRD results.
- Threshold Refinement: For NPM1-mutated AML, the threshold for bone marrow positivity has been tightened to 0.1% to enable even earlier clinical intervention.
- Standardized Terminology: Moving away from “negative” vs. “positive” toward a tiered reporting system: Optimal, Warning, and High Risk of Treatment Failure.
Prognosis and Survival Rates
- Prognosis varies significantly depending on several factors
- Type of leukemia: Each type has a different prognosis, with acute leukemias generally having lower survival rates compared to chronic types.
- Age: Younger patients often have better outcomes compared to older patients.
- Genetic mutations: Certain mutations can influence prognosis and response to treatment.
- Overall health: Underlying medical conditions can impact treatment tolerance and outcomes.
- Survival rates: While constantly improving, they vary based on the factors mentioned above. For example, the 5-year survival rate for childhood ALL is around 90%, while for adult AML, it is approximately 30%.
Treatment and Management
The management of leukemia has transitioned from “one-size-fits-all” cytotoxic chemotherapy to a highly personalized, risk-stratified approach. The strategy is defined by the integration of molecular targets, measurable residual disease (MRD) monitoring, and a major shift toward active treatment for older or less-fit patients.
The Three Phases
For acute leukemias (AML and ALL), treatment is traditionally structured into distinct phases to maximize the probability of a cure while managing toxicity.
| Phase | Primary Goal | Typical Duration | Key Interventions |
| Induction | Achieve Complete Remission (CR) (blasts <5%, normal blood counts). | 4–6 weeks | Intensive chemo (e.g., 7+3 for AML) or targeted “cocktails.” |
| Consolidation | Eliminate “hidden” leukemic cells (MRD) to prevent relapse. | 4–8 months | High-dose Cytarabine (HiDAC), targeted agents, or Stem Cell Transplant. |
| Maintenance | Prolong remission; specifically used in ALL and APL. | 2–3 years | Low-dose oral chemo (Methotrexate/6-MP) or TKIs. |
Lineage-Specific Strategies (2026 Updates)
Treatment protocols are strictly dictated by the lineage (Myeloid vs. Lymphoid) and the speed of progression (Acute vs. Chronic).
Acute Myeloid Leukemia (AML)
- The “Fitness over Age” Shift: The 2026 ASH Guidelines emphasize that chronological age is no longer a barrier. “Younger” older adults (sexagenarians) are now encouraged toward intensive therapy if fit.
- Targeted Induction: Treatment is now “mutation-first.” If a patient is FLT3-positive, Midostaurin or Gilteritinib is added to frontline chemo. For older/unfit patients, the combination of Venetoclax + Azacitidine has become the global standard, offering high response rates without the need for intense hospitalization.
Acute Lymphoblastic Leukemia (ALL)
- Pediatric-Inspired Protocols: For Adolescents and Young Adults (AYAs), pediatric regimens (heavy on Asparaginase) are superior to traditional adult protocols.
- The Immunotherapy Revolution: Drugs like Blinatumomab (BiTE) and Inotuzumab are increasingly moving to the frontline. They “bridge” patients to transplant with much lower toxicity than traditional chemo.
Chronic Leukemias (CML and CLL)
- CML: Managed primarily with Tyrosine Kinase Inhibitors (TKIs) like Imatinib or Nilotinib. The goal is a “Deep Molecular Response,” which in some cases allows for Treatment-Free Remission (TFR).
- CLL: Chemoimmunotherapy (FCR/BR) is largely obsolete. Management now focuses on BTK inhibitors (Zanubrutinib) and BCL-2 inhibitors (Venetoclax).
The “Targeted” Arsenal
Modern management utilizes the immune system and specific genetic vulnerabilities rather than just killing all dividing cells.
- CAR-T Cell Therapy: Engineered T-cells programmed to find and kill CD19+ or CD22+ B-cells. This is now a standard of care for relapsed/refractory B-ALL.
- Bispecific T-cell Engagers (BiTEs): Proteins that act as a “hook,” pulling the patient’s own T-cells directly into contact with the leukemic blasts.
Supportive Care & Oncologic Emergencies
Success in leukemia treatment is as much about managing complications as it is about killing the cancer.
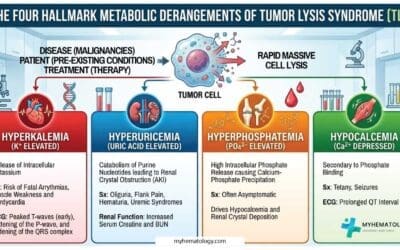
Tumor Lysis Syndrome (TLS)
Rapid cell death releases massive amounts of intracellular contents (Potassium, Phosphate, Uric Acid).
- Prophylaxis: Aggressive IV hydration and Allopurinol.
- Treatment: Rasburicase is used for high-risk patients to rapidly break down existing uric acid and protect the kidneys.
Febrile Neutropenia
A medical emergency in leukemia. Due to the lack of neutrophils, patients cannot mount a typical inflammatory response (pus/swelling).
- Protocol: Any fever > 38.0°C in a neutropenic patient requires immediate (within 1 hour) administration of broad-spectrum IV antibiotics (e.g., Piperacillin/Tazobactam).
Frequently Asked Questions (FAQs)
What is leukemia caused by?
The exact cause of leukemia is unknown in most cases. However, researchers have identified several contributing factors that seem to play a role in its development:
Genetic abnormalities: Certain genetic mutations inherited from parents or acquired during life can disrupt blood cell development and lead to uncontrolled growth of abnormal cells, potentially leading to leukemia.
Environmental factors: Exposure to some chemicals like benzene (found in gasoline and some industrial processes) and radiation (high doses from medical procedures or accidents) can increase the risk of developing certain types of leukemia.
Other factors: While the evidence is less conclusive, some studies suggest potential links between specific infections and the development of certain leukemia types, and certain medical conditions like Down syndrome can also increase the risk.
Who is most at risk for leukemia?
While anyone can develop leukemia, certain groups have a higher risk than others. The specific risk factors can vary depending on the type of leukemia.
- Age:
- Children under 5: More susceptible to acute lymphoblastic leukemia (ALL).
- Adults over 65: More susceptible to chronic lymphocytic leukemia (CLL) and acute myeloid leukemia (AML).
- Gender: Men has slightly higher risk compared to women.
- Family history: Having a close relative (parent, sibling, child) with leukemia slightly increases an individual’s risk, suggesting a possible genetic component.
- Genetic conditions: Down syndrome is associated with an increased risk of ALL. Other specific genetic mutations can also increase susceptibility.
- Environmental factors:
- High-dose radiation exposure: From medical procedures, accidents, or occupational settings.
- Exposure to certain chemicals: Benzene (found in gasoline and some industrial processes).
- Smoking: Significantly increases the risk of AML.
Is leukemia curable if caught early?
Whether leukemia is curable when caught early depends on several factors, including:
- The specific type of leukemia: Different types have different prognoses (chances of recovery) and response rates to treatment. Generally, acute leukemias have a higher chance of cure compared to chronic leukemias.
- The stage of the disease at diagnosis: Early detection plays a crucial role. Earlier stages often respond better to treatment and offer a higher chance of complete remission (disappearance of all signs of cancer).
- Individual characteristics: Factors like age, overall health, and response to treatment also influence the outcome.
While it’s not entirely accurate to say all leukemias can be cured even with early detection, here’s a general breakdown:
- Acute Leukemias
- Chronic Leukemias
- CLL: While not typically considered curable, early diagnosis allows for long-term disease control through management strategies.
- CML: While not technically curable, early detection allows for effective treatment with targeted therapies that can achieve deep remission and significantly prolong life expectancy.
What are red flags of leukemia?
Leukemia can present with various symptoms, but some stand out as potential red flags that warrant prompt medical attention. These symptoms often indicate a more critical condition and require immediate evaluation:
Rapidly worsening symptoms
- Fast and progressive fatigue: Feeling extremely tired and weak, even with minimal activity, and experiencing a rapid decline in energy levels.
- Severe and persistent infections: Frequent infections that are difficult to treat or recur quickly, suggesting a significantly compromised immune system.
- Significant bleeding: Unprovoked bleeding episodes, such as heavy nosebleeds, prolonged bleeding from minor cuts, or bleeding gums without obvious cause.
- Rapid weight loss: Unexplained and rapid weight loss, not attributed to dietary changes or exercise.
Alarming physical changes
- Enlarged lymph nodes: Swollen lymph nodes, particularly in the neck, armpits, or groin, that are firm, painless, and rapidly growing.
- Swollen abdomen: A noticeable enlargement of the abdomen due to an enlarged spleen or liver, often accompanied by discomfort or pain.
- Bone pain: Persistent and severe pain in the bones or joints, especially without any apparent injury or trauma.
- Neurological symptoms: Headaches, seizures, or changes in mental clarity, which could indicate central nervous system involvement in some cases.
These red flags are not exclusive to leukemia and can occur due to other conditions. However, their presence, especially when multiple symptoms occur together or worsen rapidly, necessitates seeking immediate medical attention for proper diagnosis and prompt intervention.
How long can a person live with leukemia?
Acute Leukemias
- Childhood ALL: Despite remaining the leading cause of death by disease for children past infancy in the United States, childhood cancer mortality dropped by over 50% between 1975 and 2020. This positive trend is reflected in improved survival rates for Acute Lymphoblastic Leukemia (ALL), where the 5-year survival rate for children under 15 climbed from 60% to nearly 90%, and for adolescents aged 15-19, skyrocketed from 28% to over 75%. These advancements offer hope for continued progress in treating childhood cancer.
- Adult AML: The 5-year relative survival rate for people 20 and older with AML is 28%. This means that 28 out of every 100 people diagnosed with AML at this age will be alive 5 years after their diagnosis. For people younger than 20, the 5-year relative survival rate is 69%.
Chronic Leukemias
- CLL: While there is no current cure for chronic lymphocytic leukemia (CLL), the outlook is encouraging. Around 87% of individuals diagnosed with CLL go on to live for at least 5 years. This signifies that many people with CLL can live well and manage the disease for an extended period.
- CML: Thanks to advancements in treatment, the five-year survival rate for chronic myeloid leukemia (CML) has more than doubled, with 70% of patients now surviving for five years or longer. Previously, the typical survival rate for CML was only three to five years.
What does leukemia pain feel like?
Describing leukemia pain can be challenging as the experience varies significantly depending on the individual and the source of the pain. However, here are some common types of pain associated with leukemia:
Bone pain: In leukemia, the abnormal accumulation of white blood cells in the bone marrow can cause it to expand, leading to bone pain. The long bones of the legs and arms are the most common location to experience this pain. It can be described as:
- Sharp and stabbing: This is often felt in the long bones of the arms and legs, ribs, sternum (breastbone), or back.
- Dull and aching: This might be a persistent, throbbing discomfort.
- Worse at night: Pain often intensifies at night, disrupting sleep and causing significant discomfort.
Joint pain: This can occur when leukemia cells accumulate around joints, causing inflammation and discomfort. It can feel similar to:
- Arthritis pain: Aching and stiffness in the joints, making movement difficult.
- General muscle soreness: A dull ache or tightness in the muscles surrounding the affected joint.
Does leukemia affect sperm?
Yes, leukemia can affect sperm production and quality in several ways.
- Direct damage: Leukemia cells can infiltrate the testicles, the organs responsible for sperm production. This infiltration can directly damage sperm-producing cells (spermatogonia) and disrupt spermatogenesis, the process of sperm formation.
- Chemotherapy: Many chemotherapy drugs used to treat leukemia can have side effects on sperm production. These drugs can damage spermatogonia, reduce sperm count, motility (movement), and morphology (shape).
- Hormonal imbalances: Leukemia can disrupt the production of hormones like testosterone, which plays a crucial role in sperm production and function.
Can leukemia stop you from having kids?
Leukemia itself doesn’t directly stop you from having kids, but the treatments used to fight it can impact fertility in both men and women.
- Chemotherapy, commonly used to treat leukemia, can damage egg and sperm cells, potentially reducing fertility or even causing temporary or permanent infertility.
- Radiation therapy directed to the pelvic area can also significantly reduce fertility potential.
However, fertility preservation methods like sperm banking or egg freezing before treatment can offer options for having biological children in the future.
What is silent leukemia?
The term “silent leukemia” is not a specific medical term referring to a distinct type of leukemia. However, it’s sometimes used informally to describe chronic lymphocytic leukemia (CLL) in its early stages because it often doesn’t present with noticeable symptoms.
- Slow-growing: CLL is a slow-growing cancer, meaning symptoms often develop gradually and might not be readily apparent in the early stages.
- Non-specific symptoms: Even when symptoms do arise, they can be vague and non-specific, such as fatigue, mild infections, or weight loss, which can be easily attributed to other causes.
Why does leukemia cause splenomegaly?
Leukemia causes splenomegaly, or an enlarged spleen, due to a combination of factors.
- Infiltration by abnormal cells: Leukemia involves the uncontrolled growth of abnormal white blood cells. These cells can infiltrate the spleen, which is responsible for filtering blood and removing old or damaged cells. As the abnormal cells accumulate in the spleen, it enlarges to accommodate the increased cellular load.
- Increased workload: The spleen plays a crucial role in the immune system, fighting infections and removing abnormal cells from the bloodstream. In leukemia, the increased production of abnormal white blood cells puts a significant strain on the spleen, causing it to work harder and potentially enlarge to meet the increased demand.
- Extramedullary hematopoiesis: Normally, blood cell production occurs in the bone marrow. However, in some cases of leukemia, the bone marrow becomes overwhelmed by abnormal cells, and blood cell production can start occurring in other organs, including the spleen. This additional production can contribute to the enlargement of the spleen.
Why do leukemia patients need blood transfusions?
Leukemia patients commonly need blood transfusions for these main reasons:
- To combat anemia: Leukemia and its treatments can damage the bone marrow, limiting its ability to produce healthy red blood cells. This leads to anemia, causing fatigue, weakness, and shortness of breath. Red blood cell transfusions replenish these cells, improving oxygen delivery and alleviating symptoms.
- To prevent bleeding: Leukemia often leads to thrombocytopenia, a dangerously low platelet count. Platelets are critical for blood clotting. Platelet transfusions increase platelet levels, minimizing the risk of excessive bleeding or bruising.
- To fight infections: Some leukemias result in low white blood cell counts (neutropenia), weakening the immune system. While less common, transfusions of specific white blood cells can temporarily boost the body’s ability to combat infections.
- To support treatment: Chemotherapy and some leukemia treatments further damage bone marrow function. Transfusions provide an immediate supply of essential blood cells, helping patients tolerate and recover from treatments.
The type and frequency of transfusions depend on the leukemia type, how it affects the patient’s blood cells, their treatment plan, and their response.
What is the difference between leukemia and lymphoma?
While both are forms of blood cancer, leukemia primarily affects the blood and bone marrow, characterized by the rapid overproduction of abnormal white blood cells. Lymphoma mainly targets the lymphatic system, including the lymph nodes, spleen, and thymus, and typically presents as solid tumors within these tissues rather than circulating in the bloodstream.
How is flow cytometry used in the diagnosis of leukemia?
Flow cytometry analyzes the unique cluster of differentiation (CD) markers on the surface of blood cells. By running a patient’s bone marrow or blood sample through the flow cytometer, pathologists can pinpoint the exact lineage (myeloid vs. lymphoid) and the maturity level of the leukemic blasts. This precise immunophenotyping is essential for determining the specific leukemia subtype and guiding targeted treatment plans.
Are gene editing technologies being used to treat leukemia?
Yes, precision medicine in hematology is rapidly advancing. Technologies like CRISPR and highly precise base editors are actively being researched to target and correct the specific molecular genetics and chromosomal translocations that drive leukemia. While still largely in the research and trial phases, these methods potentially offer curative therapies at the fundamental DNA level.
What does it mean to test for Minimal Residual Disease (MRD)?
MRD testing looks for microscopic traces of leukemia cells that standard morphological tests might miss after a patient has undergone chemotherapy. Achieving an “MRD-negative” status means that highly sensitive techniques cannot detect remaining cancer cells, which generally correlates with a much more favorable long-term prognosis and helps clinicians decide if further interventions, like stem cell transplants, are necessary.
Glossary of Related Medical Terms
- Auer Rods: Clumps of azurophilic granular material that form elongated needles seen in the cytoplasm of leukemic blasts, strongly indicative of Acute Myeloid Leukemia (AML).
- Base Editing: An advanced form of molecular gene editing that allows for the direct, irreversible conversion of one specific DNA base into another without causing double-strand DNA breaks. It is currently being extensively researched for targeted hematological therapies.
- Blast Cells: Immature, undifferentiated blood cells that multiply uncontrollably in acute leukemias, failing to develop into functional, mature blood cells.
- Extramedullary Hematopoiesis: The formation of blood cells outside of the bone marrow, such as in the liver or spleen, which can lead to organomegaly in certain chronic leukemias.
- Flow Cytometry: A highly sensitive laboratory diagnostic technology used to analyze the physical and chemical characteristics of cells in a fluid. It is an essential tool for the immunophenotyping and precise subtyping of leukemias.
- Hematopoiesis: The normal, heavily regulated process of blood cell formation, development, and differentiation that occurs within the bone marrow.
- Minimal Residual Disease (MRD): The minute number of leukemic cells that remain in the patient’s body during or after treatment. Measuring MRD is a critical indicator for evaluating the risk of clinical relapse.
- Philadelphia Chromosome: A specific genetic abnormality in chromosome 22 of leukemia cells (hallmark in CML), resulting from a reciprocal translocation with chromosome 9, leading to the BCR-ABL fusion gene.
Disclaimer: This article is intended for informational purposes only and is specifically targeted towards medical students. It is not intended to be a substitute for informed professional medical advice, diagnosis, or treatment. While the information presented here is derived from credible medical sources and is believed to be accurate and up-to-date, it is not guaranteed to be complete or error-free. See additional information.
References
- Du M, Chen W, Liu K, Wang L, Hu Y, Mao Y, Sun X, Luo Y, Shi J, Shao K, Huang H, Ye D. The Global Burden of Leukemia and Its Attributable Factors in 204 Countries and Territories: Findings from the Global Burden of Disease 2019 Study and Projections to 2030. J Oncol. 2022 Apr 25;2022:1612702. doi: 10.1155/2022/1612702. PMID: 35509847; PMCID: PMC9061017.
- Dong Y, Shi O, Zeng Q, Lu X, Wang W, Li Y, Wang Q. Leukemia incidence trends at the global, regional, and national level between 1990 and 2017. Exp Hematol Oncol. 2020 Jun 19;9:14. doi: 10.1186/s40164-020-00170-6. PMID: 32577323; PMCID: PMC7304189.
- Hwang SM. Classification of acute myeloid leukemia. Blood Res. 2020 Jul 31;55(S1):S1-S4. doi: 10.5045/br.2020.S001. PMID: 32719169; PMCID: PMC7386892.
- Döhner H, Wei AH, Appelbaum FR, Craddock C, DiNardo CD, Dombret H, Ebert BL, Fenaux P, Godley LA, Hasserjian RP, Larson RA, Levine RL, Miyazaki Y, Niederwieser D, Ossenkoppele G, Röllig C, Sierra J, Stein EM, Tallman MS, Tien HF, Wang J, Wierzbowska A, Löwenberg B. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood. 2022 Sep 22;140(12):1345-1377. doi: 10.1182/blood.2022016867. PMID: 35797463.
- Ishii H, Yano S. New Therapeutic Strategies for Adult Acute Myeloid Leukemia. Cancers (Basel). 2022 Jun 5;14(11):2806. doi: 10.3390/cancers14112806. PMID: 35681786; PMCID: PMC9179253.
- Sekeres MA. When Blood Breaks Down: Life Lessons from Leukemia (Mit Press). 2021
- Bhushan B. AML (Acute Myeloid Leukemia): A survival guide for patients. 2021.
- Greiner J. Immunotherapies for Acute Myeloid Leukemia (MDPI AG). 2020.
- Daniel J. DeAngelo, Elias Jabbour, and Anjali Advani. Recent Advances in Managing Acute Lymphoblastic Leukemia. American Society of Clinical Oncology Educational Book 2020 :40, 330-342.
- Yilmaz M, Kantarjian H, Ravandi-Kashani F, Short NJ, Jabbour E. Philadelphia chromosome-positive acute lymphoblastic leukemia in adults: current treatments and future perspectives. Clin Adv Hematol Oncol. 2018 Mar;16(3):216-223. PMID: 29742077.
- Saleh K, Fernandez A, Pasquier F. Treatment of Philadelphia Chromosome-Positive Acute Lymphoblastic Leukemia in Adults. Cancers (Basel). 2022 Apr 1;14(7):1805. doi: 10.3390/cancers14071805. PMID: 35406576; PMCID: PMC8997772.
- Carr JH. Clinical Hematology Atlas 6th Edition (Elsevier).
- Hallek M, Cheson BD, Catovsky D, Caligaris-Cappio F, Dighiero G, Döhner H, Hillmen P, Keating MJ, Montserrat E, Rai KR, Kipps TJ; International Workshop on Chronic Lymphocytic Leukemia. Guidelines for the diagnosis and treatment of chronic lymphocytic leukemia: a report from the International Workshop on Chronic Lymphocytic Leukemia updating the National Cancer Institute-Working Group 1996 guidelines. Blood. 2008 Jun 15;111(12):5446-56. doi: 10.1182/blood-2007-06-093906. Epub 2008 Jan 23. Erratum in: Blood. 2008 Dec 15;112(13):5259. PMID: 18216293; PMCID: PMC2972576.
- National Comprehensive Cancer Network® (NCCN®). NCCN Guidelines for Patients® Chronic Lymphocytic Leukemia. 2022.
- Hallek M, Eichhorst B, Catovsky D. Chronic Lymphocytic Leukemia (Hematologic Malignancies) 1st ed. 2019 Edition (Springer).
- Sekeres, M. A., Mattison, R., Artz, A., Baer, M. R., Chua, C. C., Demichelis-Gómez, R., Egan, P. C., Fletcher, L., Foucar, C., Garcia, J. S., Gilberto, L., Gómez de León, A., Lancet, J., Loh, K. P., Malcovati, L., Marini, B., Platzbecker, U., Sorror, M. L., Tinsley-Vance, S., Treitz, J., … Brignardello-Petersen, R. (2026). American Society of Hematology 2025 guidelines for treating newly diagnosed acute myeloid leukemia in older adults. Blood advances, 10(6), 1897–1928. https://doi.org/10.1182/bloodadvances.2025017934
- DuVall, A. S., McNeer, J., Cheung, M. C., Adrianzen-Herrera, D. A., Advani, A. S., Aljurf, M., Audino, A. N., Benitez, L., Deeter, A., Dworkin, E., Hiltbrand, O. S., Isenalumhe, L. L., Li, L., Miller-Chism, C. N., Mozessohn, L., Ng, A. P., Pu, J. J., Shafer, D., Vargas Madueno, F., Viny, A. D., … Wolfson, J. A. (2026). ASH 2026 Guidelines for Frontline Management of Acute Lymphoblastic Leukemia in Adolescents and Young Adults. Blood advances, bloodadvances.2021006469. Advance online publication. https://doi.org/10.1182/bloodadvances.2021006469
- Cloos, J., Valk, P. J. M., Thiede, C., Döhner, K., Roboz, G. J., Wood, B. L., Walter, R. B., Wang, S., Wierzbowska, A., Wei, A. H., Wu, D., Vergez, F., Venditti, A., van der Reijden, B. A., van de Loosdrecht, A. A., Tiong, I. S., Thol, F. R., Subklewe, M., Roumier, C., Reuvekamp, T., … Heuser, M. (2026). 2025 update on MRD in acute myeloid leukemia: a consensus document from the ELN-DAVID MRD Working Party. Blood, 147(11), 1147–1167. https://doi.org/10.1182/blood.2025031480