TL;DR
Neutropenic sepsis is a medical emergency defined by a fever (≥ 38.3°C (101°F)) and an ANC < 0.5 X 109/L. It requires a “door-to-needle” time of < 60 minutes for the first dose of broad-spectrum antibiotics.
- Pathophysiology ▾: It is driven by a “Perfect Storm” of a depleted immune system and breached mucosal barriers, allowing endogenous flora to invade.
- Clinical Presentation ▾: Fever may be the only sign; classic inflammatory signs like pus or swelling are often absent.
- Investigations ▾: Perform a rapid “source hunt” with paired blood cultures and baseline organ profiling, but never delay the first dose of antibiotics while awaiting results.
- Management ▾: Follow the Sepsis Six. Use antipseudomonal beta-lactams (e.g., Piperacillin-Tazobactam) as first-line therapy.
- Prevention ▾: G-CSF and prophylactic antimicrobials are key for high-risk patients.
*Click ▾ for more information
Introduction
Neutropenic sepsis (often referred to interchangeably with “febrile neutropenia” in clinical practice) represents one of the most significant and life-threatening complications in hematology and oncology. It is a unique clinical entity where the body’s primary defense mechanism against infection, the neutrophil, is severely depleted.
The most important concept to grasp is the blunted inflammatory response. Because these patients lack sufficient white blood cells to mount a typical immune response, classic signs of infection (such as pus formation, swelling, or localized pain) are frequently absent. Consequently, fever may be the only warning sign of a rapidly progressing, systemic infection.
Defining the Parameters
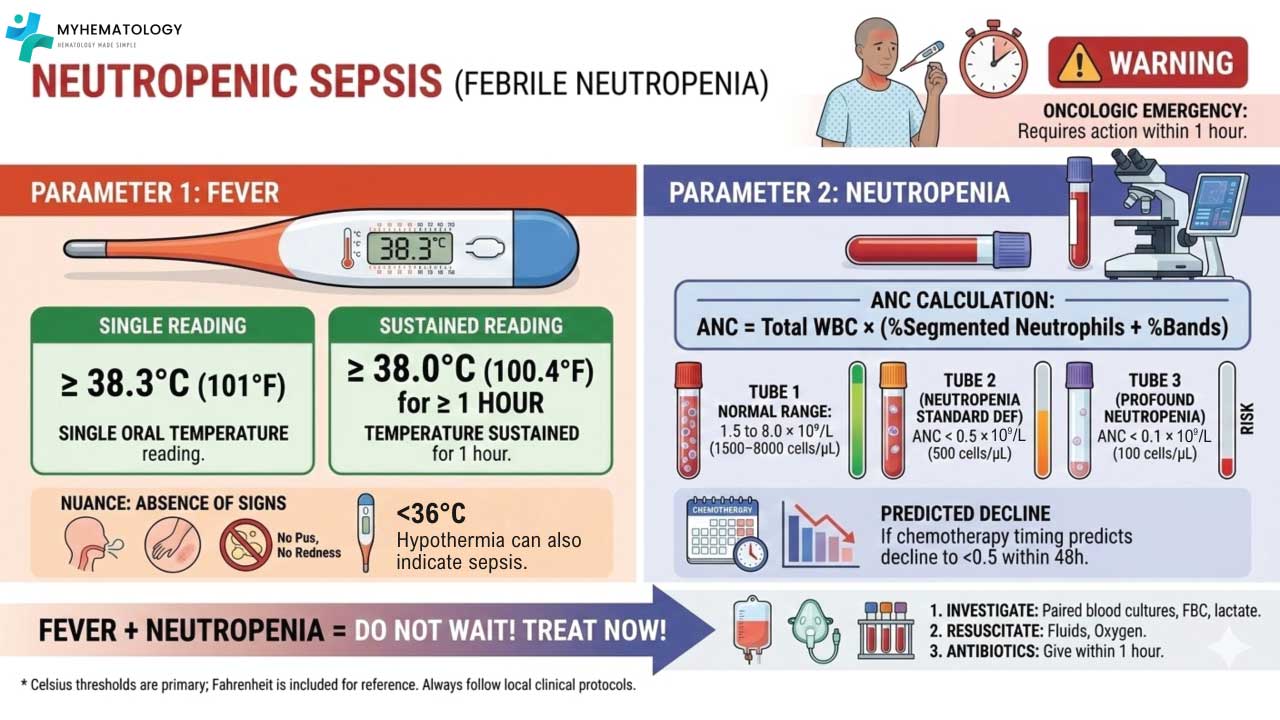
To ensure standardized care and prompt intervention, the medical community relies on precise definitions for both the hematologic and systemic components of this condition:
- Neutropenia: Defined as an Absolute Neutrophil Count (ANC) of < 0.5 X 109/L, or an ANC that is expected to fall below this threshold within the next 48 hours.
- Profound Neutropenia: An ANC of < 0.1 X 109/L carries a significantly higher risk of severe bacterial and fungal infections.
- Fever: In the context of neutropenia, fever is defined as:
- A single oral temperature of ≥ 38.3°C (101°F), OR
- A sustained temperature of ≥ 38.0°C (100.4°F) over a period of at least one hour.
Neutropenic Sepsis occurs when a patient with neutropenia develops signs of systemic inflammatory response or organ dysfunction secondary to a suspected or proven infection.
The “Medical Emergency” Framework
Neutropenic sepsis is a time-critical emergency. Without neutrophils to contain bacteria, pathogens can translocate from the gut or skin into the bloodstream, leading to septic shock and multi-organ failure within hours.
Historically, the mortality rate for untreated neutropenic sepsis was as high as 70%. While modern intensive care and broad-spectrum antibiotics have reduced neutropenic sepsis mortality rate to between 2% and 21% (depending on the underlying malignancy and risk factors), the prognosis remains strictly dependent on the speed of antibiotic administration.
Epidemiology and Impact
- Incidence: Neutropenic sepsis is most common in patients undergoing intensive chemotherapy for hematologic malignancies (such as Acute Myeloid Leukemia), where the incidence of febrile neutropenia (neutropenic sepsis) can exceed 80%. In patients treated for solid tumors, the incidence is lower, typically between 10% and 50%.
- Burden: Beyond the risk of death, neutropenic sepsis often leads to dose reductions or delays in life-saving chemotherapy, potentially compromising the long-term cure of the patient’s underlying cancer.
Etiology of Neutropenic Sepsis
The etiology of neutropenic sepsis is broad, but for clinical purposes, it is generally categorized into treatment-related (iatrogenic), primary hematologic disorders, and secondary/idiosyncratic causes.
Iatrogenic: The Role of Cytotoxic Therapy
The most common cause of neutropenic sepsis in clinical practice is Systemic Anti-Cancer Therapy (SACT). Cytotoxic chemotherapy works by targeting rapidly dividing cells; unfortunately, hematopoietic stem cells and progenitor cells in the bone marrow are collateral damage.
- The Concept of the “Nadir”: Nadir is the point at which the blood count reaches its lowest level following treatment. For most conventional chemotherapy, the nadir occurs between 7 to 14 days post-administration. Recovery typically occurs by day 21 or 28, which dictates the “cycle” length of chemotherapy.
- High-Risk Regimens: Certain treatments carry a > 20% risk of causing neutropenic sepsis. These include:
- Induction for Acute Myeloid Leukemia (AML): Often results in profound, prolonged neutropenia (<0.1 X 109/L for weeks).
- Conditioning for Stem Cell Transplant: High-dose melphalan or BEAM regimens.
- Dose-Intense Lymphoma Regimens: Such as CODOX-M/IVAC.
- Other Iatrogenic Causes: While less common than chemotherapy, radiotherapy (especially to large areas of bone marrow like the pelvis or spine) and certain biologic/targeted therapies (e.g., Rituximab, Palbociclib) can also suppress neutrophil production.
Primary Bone Marrow Disorders
In these cases, the neutropenia is not a side effect of treatment but a direct result of the underlying disease process.
- Infiltration (Myelophthisis): Malignant cells (Leukemia, Lymphoma, or metastatic solid tumors like breast or prostate cancer) physically “crowd out” healthy hematopoietic tissue.
- Ineffective Hematopoiesis: In Myelodysplastic Syndromes (MDS), the bone marrow may be hypercellular, but the cells produced are dysplastic and fail to mature or enter the peripheral blood.
- Aplastic Anemia: A global failure of the bone marrow to produce all three cell lines (pancytopenia), often due to an autoimmune T-cell mediated attack on stem cells.
Idiosyncratic and Non-Malignant Causes
There are non-chemotherapy drugs that cause agranulocytosis. These are often idiosyncratic (unpredictable and not dose-dependent).
- Common Offending Medications:
- Antithyroid drugs: Carbimazole, Propylthiouracil.
- Antipsychotics: Clozapine (requires mandatory CBC monitoring).
- Antibiotics: Sulfonamides, Penicillins (rarely), and Vancomycin.
- Anti-inflammatories: Sulfasalazine, Gold salts.
- Viral Infections: Viruses can cause transient neutropenia through direct marrow suppression or peripheral destruction. Examples include HIV, CMV, EBV, and Hepatitis.
- Nutritional Deficiencies: Severe Vitamin B12 or Folate deficiency can lead to megaloblastic anemia and associated neutropenia due to impaired DNA synthesis.
| Risk Level | Incidence of Febrile Neutropenia | Examples |
| High Risk | > 20% | AML Induction, High-dose therapy for Lymphoma |
| Intermediate | 10 – 20% | Many breast/lung cancer regimens (e.g., Docetaxel) |
| Low Risk | < 10% | Weekly Paclitaxel, 5-Fluorouracil (5-FU) |
Pathophysiology of Neutropenic Sepsis
The development of neutropenic sepsis involves a complex interplay between the host’s weakened immune system, the toxicity of medical treatments, and the opportunistic nature of the human microbiome.
The Loss of the First Line of Defense
Neutrophils are the “first responders” of the innate immune system. Under normal conditions, they are the first cells to migrate to a site of infection via chemotaxis, where they neutralize pathogens through phagocytosis, the release of reactive oxygen species (ROS), and Neutrophil Extracellular Traps (NETs).
- Quantitative Deficiency: When the Absolute Neutrophil Count (ANC) drops below 0.5 X 109/L, the body loses the ability to “corral” an infection. A localized bacterial entry that a healthy person would clear easily (like a micro-abrasion in the gut) becomes a systemic threat.
- Qualitative Deficiency: In many hematologic malignancies (like AML or MDS), the neutrophils that are present are often dysfunctional, further compounding the risk.
Mucosal Barrier Breakdown (Mucositis)
Cytotoxic chemotherapy does not discriminate between malignant cells and the rapidly dividing healthy cells of the gastrointestinal (GI) tract. Chemotherapy-induced mucositis causes thinning and ulceration of the mucosal lining from the mouth to the anus. This eliminates the physical barrier between the dense microbial population of the gut and the bloodstream. Consequently, the vast majority of infections in neutropenic sepsis are endogenous, caused by the patient’s own flora “crossing the line.”
The Shift in Pathogens
Historically, Gram-negative organisms were the primary concern. However, clinical patterns have shifted over the last few decades.
- Gram-Negative Bacteria: (e.g., Pseudomonas aeruginosa, Escherichia coli, Klebsiella species). These remain the most dangerous due to their potential to produce endotoxins, which trigger a rapid and often fatal septic cascade.
- Gram-Positive Bacteria: (e.g., Staphylococcus epidermidis, Viridans group streptococci). There has been a rise in these infections, largely due to the increased use of indwelling central venous catheters (Hickman lines, PICC lines) and the use of fluoroquinolone prophylaxis which selectively kills Gram-negatives.
- Fungal Pathogens: In cases of prolonged neutropenia (typically >7 days), the risk of invasive fungal infections like Candida or Aspergillus increases significantly as the bacterial competition is wiped out by broad-spectrum antibiotics.
The Sepsis Cascade in the Absence of Pus
Students often expect to see classic signs of infection, such as redness, swelling, or pus (purulence). However, pus is primarily composed of dead neutrophils.
- The Silent Infection: In a neutropenic patient, a severe pneumonia may show a “clear” chest X-ray because there are no white cells to create an infiltrate. A significant skin infection may not be red or swollen.
- Cytokine Storm: Despite the lack of neutrophils, other immune cells (macrophages and monocytes) still detect Pathogen-Associated Molecular Patterns (PAMPs). They release pro-inflammatory cytokines such as TNF-ɑ, IL-1, and IL-6. These cytokines drive the systemic inflammatory response, leading to:
- Vasodilation and capillary leak (hypotension).
- Tachycardia.
- Pyrexia (fever).
- End-organ hypoperfusion.
Symptoms and Clinical Presentation of Neutropenic Sepsis
Fever
Fever is often the first, and sometimes only, clinical indicator of neutropenic sepsis. In a patient with an ANC < 0.5, a temperature of ≥ 38.3°C is a medical emergency until proven otherwise.
- Hypothermia: Occasionally, particularly in the elderly or those in early septic shock, patients may present with hypothermia (36.0°C), which is an equally ominous sign.
The “Invisible” Infection
Because there are no neutrophils to migrate to the site of infection, the typical hallmarks of inflammation e.g. rubor (redness), tumor (swelling), and pus (purulence), may be entirely absent.
- Pneumonia: May present with hypoxia and tachypnea, but a “clear” chest on auscultation.
- Urinary Tract Infection (UTI): May present with frequency or pain, but the urine dipstick may be negative for nitrites or leukocytes because there aren’t enough bacteria or white cells to trigger the test.
- Skin/Line Infections: A central line site might look perfectly calm even if it is the source of a Staphylococcal bacteremia.
Focal Symptoms
While fever is the main clue, a “top-to-toe” assessment should be performed looking for subtle focal signs:
- Oropharynx: Sore throat, retrosternal pain (esophagitis), or white patches (Candidiasis).
- Respiratory: New cough, even if non-productive, or increased respiratory rate.
- Gastrointestinal: New-onset diarrhea (think C. difficile or typhlitis/neutropenic enterocolitis) or vague abdominal tenderness.
- Perianal Area: This is a common but overlooked site. Any pain, itching, or soreness in the perianal region in a neutropenic patient is highly suspicious for an abscess, even without visible swelling.
- Neurological: Confusion, irritability, or a “feeling of impending doom” (often an early sign of hypoperfusion).
Signs of Impending Septic Shock
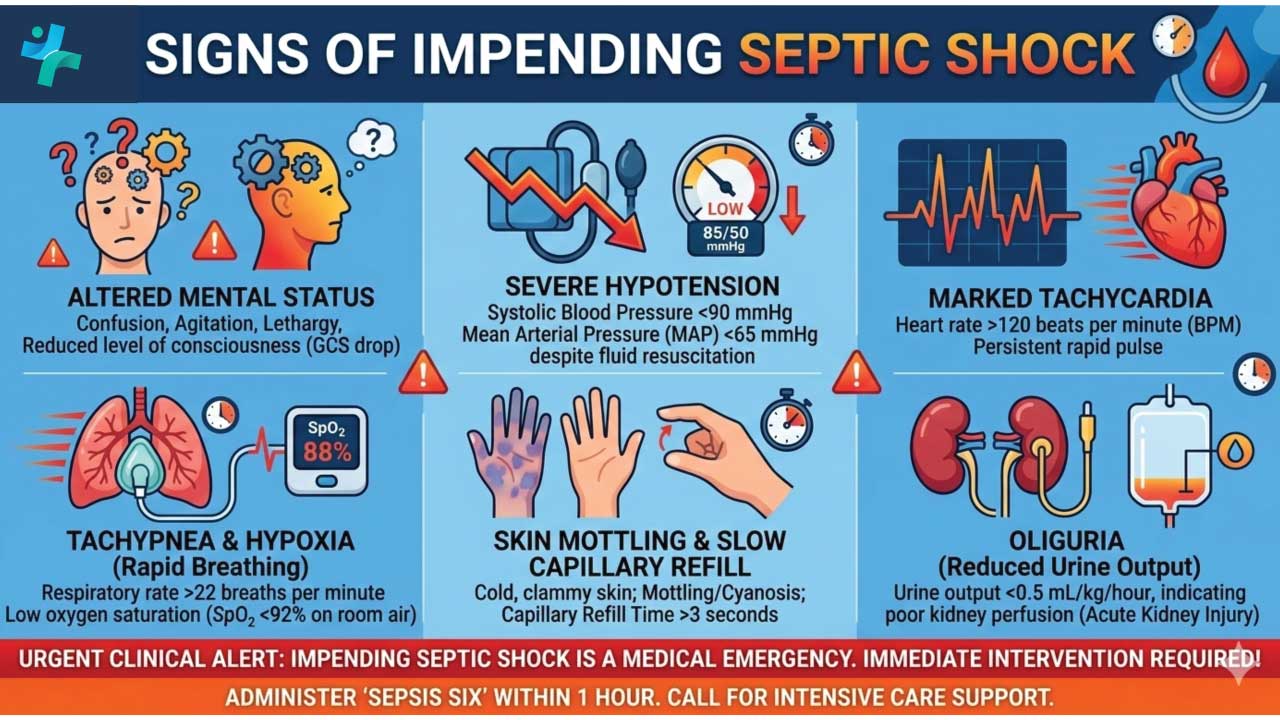
If the infection is not caught early, the patient will quickly progress to overt sepsis. We should be vigilant for the “Red Flag” signs:
- Hypotension: Systolic Blood Pressure <90 mmHg or a drop of >40 mmHg from baseline.
- Tachycardia: Heart rate >100 bpm.
- Tachypnea: Respiratory rate >20 breaths/min.
- Oliguria: Reduced urine output (indicating renal hypoperfusion).
- Altered Mental Status: Confusion or lethargy.
If a neutropenic patient says they “just don’t feel right,” believe them. In the absence of white cells, subjective malaise is a high-sensitivity marker for objective deterioration.
Summary of Checklist for Clinical Examination
| System | What to Look For (Subtle) |
| Skin | Look at IV sites, PICC/Hickman line exits, and recent biopsy sites. |
| Mouth | Check for mucositis, ulcers, or thrush. |
| Lungs | Check respiratory rate and oxygen saturations (more reliable than stethoscopes here). |
| Abdomen | Listen for bowel sounds; palpate for right lower quadrant tenderness (Typhlitis). |
| Perianal | Visual inspection only. Never perform a digital rectal exam (DRE) in a neutropenic patient due to the risk of bacterial translocation. |
Laboratory Investigations and Imaging
The most critical takeaway is the “Investigate, but don’t wait” rule. All investigations should be performed rapidly, but the first dose of broad-spectrum antibiotics should never be delayed for more than 60 minutes while waiting for imaging or lab results.
The Essential Blood Panel
The goal here is twofold: to confirm the diagnosis of neutropenia and to assess the degree of systemic organ dysfunction.
- Complete Blood Count (CBC) with Differential: This is the diagnostic cornerstone.
- Students must look specifically at the Absolute Neutrophil Count (ANC), not just the total White Cell Count (WBC).
- Calculation: ANC = WBC X (% Neutrophils + % Bands)
- Renal Profile (Urea & Electrolytes): Crucial for baseline kidney function, as many empiric antibiotics (like Gentamicin or Vancomycin) are nephrotoxic and require dose adjustment.
- Liver Function Tests (LFTs): To screen for hepatic involvement or congestion.
- C-Reactive Protein (CRP) / Procalcitonin: While these are markers of inflammation, they may be sluggish to rise in a neutropenic patient. They are most useful as a “baseline” to track the trend of recovery rather than as a tool to rule out infection.
- Coagulation Screen & Lactate: Essential for identifying Disseminated Intravascular Coagulation (DIC) or lactic acidosis, which are markers of severe septic shock.
Microbiological Investigations
In approximately 70% of neutropenic sepsis cases, a clear microbiological cause is never identified. However, we must “cast a wide net” to find the culprit.
- Paired Blood Cultures (The Gold Standard):
- Peripheral Culture: From a fresh venipuncture.
- Central Culture: From every lumen of an indwelling catheter (Hickman, PICC, or Port-a-Cath).
- Why? If the central line culture grows bacteria significantly faster than the peripheral one (Differential Time to Positivity), the line itself is likely the source.
- Urine Culture: Obtain even if the patient has no urinary symptoms.
- Stool Culture: If the patient has diarrhea, specifically test for Clostridium difficile and enteric pathogens.
- Viral Swabs: Throat and nose swabs for respiratory viruses (e.g., Influenza, COVID-19, RSV) as these can mimic bacterial sepsis.
- The Forbidden Procedure: Digital Rectal Examinations (DRE) and the use of rectal thermometers/suppositories are strictly contraindicated. The risk of causing mucosal micro-trauma and subsequent bacterial translocation into the bloodstream far outweighs the diagnostic benefit. DO NOT PERFORM.
Imaging
As discussed in pathophysiology, a neutropenic patient’s chest X-ray can look deceptively normal because they lack the cells to form an “opacification” or “infiltrate.”
- Chest X-Ray (CXR): Performed for all febrile neutropenic patients. It serves as a baseline, though it has low sensitivity in the early stages.
- High-Resolution CT (HRCT) Chest: If the fever persists despite 48 – 72 hours of antibiotics, an HRCT is the next step.
- The “Halo Sign”: A nodule surrounded by a “halo” of ground-glass opacity which is a classic (though not pathognomonic) sign of Invasive Pulmonary Aspergillosis.
- CT Abdomen/Pelvis: Indicated if there is abdominal pain, looking for signs of Neutropenic Enterocolitis (Typhlitis), characterized by bowel wall thickening.
Differential Diagnosis
Not every fever in a neutropenic patient is caused by an infection. However, the “Golden Rule” is that we treat all of them as sepsis until proven otherwise.
The differential diagnosis is most useful when the patient is not responding to initial antibiotics or when the fever has a very specific temporal relationship with a drug or procedure.
Neoplastic (Tumor) Fever
The underlying malignancy itself can cause significant pyrexia. This is particularly common in:
- Lymphomas: (Hodgkin’s and Non-Hodgkin’s) often present with “B-symptoms,” including drenching night sweats and cyclical fevers (Pel-Ebstein fevers).
- Acute Leukemias: Rapidly proliferating blasts can release pyrogenic cytokines.
- Solid Tumors: Especially renal cell carcinoma and hepatic metastases.
Distinguishing feature: Tumor fever often lacks the systemic toxicity (hypotension, tachycardia) seen in sepsis, and patients may feel relatively “well” despite the high temperature.
Drug-Induced Fever
Many medications used in hematology can trigger a febrile response.
- Cytarabine (Ara-C) Syndrome: Common in AML treatment; presents with fever, rash, conjunctivitis, and bone pain.
- Antibiotics: Paradoxically, the very drugs we use to treat sepsis (e.g., Vancomycin, Beta-lactams) can cause a drug fever, often accompanied by a skin rash or eosinophilia.
- Monoclonal Antibodies: Agents like Rituximab or Gemtuzumab frequently cause infusion-related pyrexia due to cytokine release.
Transfusion-Related Reactions
Hematology patients are frequently transfusion-dependent.
- Febrile Non-Hemolytic Transfusion Reaction (FNHTR): A common, benign rise in temperature during or shortly after a blood/platelet transfusion caused by cytokines in the donor product.
- TRALI (Transfusion-Related Acute Lung Injury): Presents with fever, hypoxia, and lung infiltrates, mimicking neutropenic pneumonia.
Thromboembolism (VTE)
Cancer is a hypercoagulable state.
- Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE): A PE can present with sudden onset tachycardia, tachypnea, and fever. In a neutropenic patient, these signs are easily confused with sepsis.
Key Investigation: A D-dimer is often chronically elevated in cancer patients; therefore, a CT Pulmonary Angiogram (CTPA) is usually required if clinical suspicion is high.
Specialized Hematologic Conditions
- Engraftment Syndrome: Occurs in patients recovering from a stem cell transplant as their new white cells begin to function, causing a massive “pro-inflammatory” surge that mimics sepsis.
- Graft-versus-Host Disease (GVHD): Acute GVHD can present with fever, skin rash, and diarrhea, mimicking a GI-source infection.
Sepsis vs Tumor Fever
| Feature | Neutropenic Sepsis | Tumor Fever |
| Onset | Often abrupt | Often gradual/evening spikes |
| Toxicity | High (patient looks “sick”) | Low (patient looks stable) |
| CRP/Lactate | Usually significantly elevated | Often normal or mildly elevated |
| Hemodynamics | Unstable (hypotension) | Stable |
| Antibiotic Response | Usually responds within 48h | No response |
Treatment and Acute Management
The “Golden Hour”
The primary goal in neutropenic sepsis is to administer the first dose of broad-spectrum antibiotics within 60 minutes of the patient’s arrival at the hospital (or within 60 minutes of the first documented fever for an inpatient).
In the absence of neutrophils, bacteria can double in concentration every 20-30 minutes. A delay of even a few hours significantly increases the risk of septic shock and death.
Do not wait for the ANC result if there is a high clinical suspicion (e.g., a patient on active chemotherapy). Treat first, confirm later.
Immediate Actions: The “Sepsis Six“
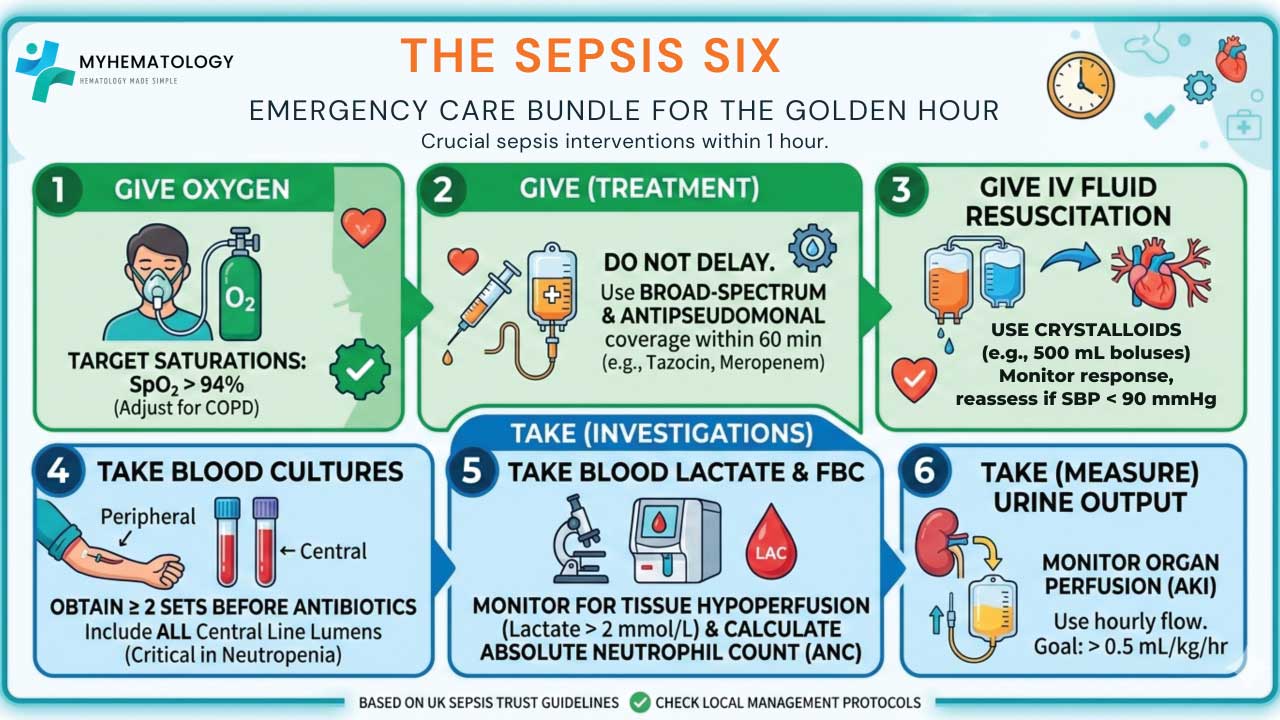
- Oxygen: Give high-flow oxygen to keep saturations > 94% (or 88 – 92% in COPD).
- Blood Cultures: Take at least two sets (Peripheral + Central Line).
- IV Antibiotics: Start empirical broad-spectrum therapy immediately.
- IV Fluids: Rapid fluid resuscitation with crystalloids (e.g., 500 mL boluses) if the patient is hypotensive or has a lactate > 2 mmol/L.
- Check Lactate: High lactate indicates tissue hypoperfusion.
- Monitor Urine Output: Accurate fluid balance is essential to monitor for acute kidney injury (AKI).
Empirical Antibiotic Choice
The choice of antibiotic must cover Pseudomonas aeruginosa, as this is the most rapidly lethal pathogen in neutropenic patients.
- First-line Monotherapy: * Piperacillin-Tazobactam (Tazocin) 4.5g IV (Standard of care in many centers).
- Cefepime or Ceftazidime (if penicillin-allergic).
- Meropenem or Imipenem (usually reserved for patients already colonized with ESBL-producing bacteria or those who are hemodynamically unstable).
- When to add Vancomycin/Teicoplanin (Gram-positive cover):
- Suspected catheter-related infection (redness at the exit site).
- Hemodynamic instability/septic shock.
- Known colonization with MRSA.
- Severe mucositis.
Risk Stratification: MASCC vs. CISNE
Not all patients require a two-week hospital stay. We use scoring systems to decide who can be safely treated with oral antibiotics at home.
- MASCC Score: A score of ≥ 21 indicates low risk (e.g., solid tumor, no hypotension, no dehydration, mild symptoms). These patients may potentially be discharged on oral Ciprofloxacin and Co-amoxiclav.
- CISNE Score: Specifically designed for patients with solid tumors to identify those at low risk of complications.
Escalation and Adjuncts
- Antifungal Therapy: If the neutropenic sepsis patient remains febrile after 4 – 7 days of broad-spectrum antibiotics and the source is unknown, empiric antifungal therapy (e.g., Caspofungin or Liposomal Amphotericin B) should be considered.
- G-CSF (Granulocyte Colony-Stimulating Factor): While G-CSF (Filgrastim) is primarily used for prevention, it is sometimes used in the acute setting if the neutropenic sepsis patient has life-threatening complications (e.g., multi-organ failure) to shorten the duration of neutropenia.
- Source Control: If a central line is suspected to be the source and the neutropenic sepsis patient is unstable or the culture grows Staph. aureus or Candida, the line must be removed.
Antibiotic Selection Logic
| Scenario | Recommended Antibiotic |
| Standard Presentation | Piperacillin-Tazobactam |
| Penicillin Allergy (Mild) | Ceftazidime |
| Penicillin Allergy (Anaphylaxis) | Ciprofloxacin + Vancomycin OR Aztreonam |
| Septic Shock / Unstable | Meropenem + Vancomycin |
Complications and Prognosis of Neutropenic Sepsis
Major Complications
When the initial “Golden Hour” interventions fail to stabilize the neutropenic sepsis patient, a cascade of systemic failures often follows:
- Septic Shock and Hemodynamic Collapse: Persistent hypotension despite adequate fluid resuscitation, requiring vasopressor support (e.g., Noradrenaline). This is often driven by massive peripheral vasodilation and capillary leak.
- Acute Respiratory Distress Syndrome (ARDS): Characterized by non-cardiogenic pulmonary edema and severe hypoxia – low PaO2/FiO2 ratio on arterial blood gas.
- Acute Kidney Injury (AKI): Resulting from both systemic hypoperfusion (pre-renal) and the nephrotoxicity of certain “standard” treatments like Vancomycin, Aminoglycosides, or contrast-enhanced CT scans.
- Disseminated Intravascular Coagulation (DIC): A catastrophic breakdown of the coagulation system leading to simultaneous microvascular thrombosis and systemic hemorrhage.
- Neutropenic Enterocolitis (Typhlitis): A life-threatening inflammation of the cecum. In a neutropenic patient, this can progress to bowel perforation and peritonitis with terrifying speed.
- Secondary Invasive Fungal Infections (IFIs): As broad-spectrum antibiotics eliminate bacterial competition, opportunistic fungi like Aspergillus or Candida can take hold, often leading to high mortality rates in the persistently neutropenic patient.
Prognosis
The prognosis of neutropenic sepsis is highly variable and depends on the “triad” of the host, the pathogen, and the timing of the intervention.
| Factor | Favorable Prognosis | Poor Prognosis |
| Duration of Neutropenia | Short-lived (e.g., solid tumor) | Prolonged (>7 days) (e.g., AML) |
| Depth of Neutropenia | ANC > 0.1 X 109/L | Profound (ANC < 0.1 X 109/L) |
| Comorbidities | None/Minimal | Age >60, COPD, Heart Failure, AKI |
| Source of Infection | Identifiable/Localized | Occult or multi-drug resistant (MDR) |
| Timing of Antibiotics | Within 60 minutes | Delay > 2 hours |
| Disease Status | In remission | Relapsed/Refractory malignancy |
The Recovery Phase
Prognosis of neutropenic sepsis improves dramatically once the bone marrow begins to recover. The first sign of recovery in neutropenic sepsis is often a rise in the monocyte count (the “monocyte signal”) followed by an increase in neutrophils. Once the ANC rises above 0.5 X 109/L and the patient has been afebrile for 48 hours, the immediate threat typically subsides.
While overall neutropenic sepsis mortality in modern units is roughly 5% to 10%, this figure can soar to 40% or higher if the patient develops septic shock or requires mechanical ventilation in the ICU.
Prevention Strategies for Neutropenic Sepsis
Hematopoietic Growth Factors (G-CSF)
The use of Granulocyte Colony-Stimulating Factor (G-CSF), such as Filgrastim or Pegfilgrastim, is the most effective way to reduce the duration of neutropenia.
- Primary Prophylaxis: Administered after chemotherapy when the regimen has a predicted > 20% risk of causing febrile neutropenia.
- Secondary Prophylaxis: Used in subsequent cycles if the patient has already experienced an episode of neutropenic sepsis.
Antimicrobial Prophylaxis
- Antibacterial: Fluoroquinolones (e.g., Levofloxacin) are often prescribed during the nadir for high-risk neutropenic sepsispatients (e.g., those with AML or undergoing stem cell transplant).
- Antifungal/Antiviral: Standard for patients with prolonged neutropenia (e.g., Fluconazole for Candida and Aciclovir for HSV/VZV reactivation).
Patient Education and “Safety Netting”
The patient is the first line of defense for neutropenic sepsis prevention. They must be taught to:
- Monitor Temperature: Check twice daily or whenever feeling unwell.
- The “No-Delay” Rule: Go to the Emergency Department immediately if a fever occurs; do not wait for the morning or call a GP.
- Hygiene: Rigorous handwashing and avoidance of crowded places or people with known infections.
- Safe Diet: While strict “neutropenic diets” (avoiding all raw fruit/veg) are increasingly replaced by general “safe food handling” guidelines, patients are still advised to avoid high-risk foods like unpasteurized dairy or blue vein cheese.
Frequently Asked Questions (FAQs)
If the patient has a fever but the ANC is 0.6 X 109/L, is it still neutropenic sepsis?
Technically no, as the threshold for neutropenic sepsis is 0.5. However, if the count is expected to drop (the “impending nadir”), it should be treated with the same clinical urgency as confirmed neutropenic sepsis.
Should I wait for the blood culture results before starting antibiotics in suspected neutropenic sepsis?
Never. The delay required for cultures to grow (12 – 48 hours) would be fatal. Antibiotics must be started within the “Golden Hour” based on clinical suspicion of neutropenic sepsis.
Why can’t I perform a rectal exam in neutropenic sepsis patients?
The rectal mucosa is thin and potentially damaged by chemotherapy in neutropenic sepsis patients. A rectal exam can cause micro-tears that allow gut bacteria to enter the bloodstream directly, potentially causing the very sepsis you are trying to investigate.
Glossary of Medical Terms
- Absolute Neutrophil Count (ANC): The actual number of neutrophils present in the blood, calculated by multiplying the total white blood cell count by the percentage of neutrophils (segmented and bands).
- Bacteremia: The presence of viable bacteria within the circulating bloodstream.
- Cytotoxic: A substance or treatment (like chemotherapy) that is toxic to cells, specifically those that are rapidly dividing.
- Endogenous Flora: The microorganisms (bacteria, fungi) that naturally reside on or inside the human body, such as in the gut or on the skin.
- Hematopoiesis: The process by which the body produces new blood cells in the bone marrow.
- Iatrogenic: A condition or complication induced inadvertently by a medical treatment or diagnostic procedure.
- Mucositis: The painful inflammation and ulceration of the mucous membranes lining the digestive tract, often a side effect of chemotherapy.
- Myelophthisis: The displacement of normal bone marrow tissue by abnormal cells, such as leukemia blasts or metastatic tumor cells.
- Nadir: The point in time after chemotherapy when the blood cell counts (specifically neutrophils and platelets) reach their lowest level.
- Pancytopenia: A simultaneous deficiency of all three cellular components of the blood: red cells, white cells, and platelets.
- Pyrexia: The medical term for fever; an elevation in body temperature above the normal range.
- Septicemia: A systemic infection caused by the multiplication of microorganisms in the blood; often used interchangeably with “sepsis” in clinical settings.
- Translocation: The passage of viable bacteria from the gastrointestinal tract through the mucosal barrier into the bloodstream or lymphatic system.
- Typhlitis: Also known as neutropenic enterocolitis; a severe, necrotizing inflammation of the cecum occurring in immunocompromised patients.
Disclaimer: This article is intended for informational purposes only and is specifically targeted towards medical students. It is not intended to be a substitute for informed professional medical advice, diagnosis, or treatment. While the information presented here is derived from credible medical sources and is believed to be accurate and up-to-date, it is not guaranteed to be complete or error-free. See additional information.
References
- National Collaborating Centre for Cancer (UK). Neutropenic Sepsis: Prevention and Management of Neutropenic Sepsis in Cancer Patients. London: National Institute for Health and Clinical Excellence (NICE); 2012 Sep. (NICE Clinical Guidelines, No. 151.) 2, Diagnosis of neutropenic sepsis. Available from: https://www.ncbi.nlm.nih.gov/books/NBK373673/
- Clarke, R. T., Jenyon, T., van Hamel Parsons, V., & King, A. J. (2013). Neutropenic sepsis: management and complications. Clinical medicine (London, England), 13(2), 185–187. https://doi.org/10.7861/clinmedicine.13-2-185.
- Kochanek, M., Schalk, E., von Bergwelt-Baildon, M., Beutel, G., Buchheidt, D., Hentrich, M., Henze, L., Kiehl, M., Liebregts, T., von Lilienfeld-Toal, M., Classen, A., Mellinghoff, S., Penack, O., Piepel, C., & Böll, B. (2019). Management of sepsis in neutropenic cancer patients: 2018 guidelines from the Infectious Diseases Working Party (AGIHO) and Intensive Care Working Party (iCHOP) of the German Society of Hematology and Medical Oncology (DGHO). Annals of hematology, 98(5), 1051–1069. https://doi.org/10.1007/s00277-019-03622-0
- Guillotin, F., Aubert, L., Benguerfi, S., Munoz Calahorro, R., Vennier, A., Boutoille, D., Gastinne, T., Reignier, J., Lascarrou, J. B., Karakachoff, M., & Canet, E. (2025). Neutropenic sepsis and septic shock in ICU patients: A single-center experience over the last decade. PloS one, 20(10), e0334511. https://doi.org/10.1371/journal.pone.0334511
- Mielecka-Jarmocik, G., Szymbor, K., Balwierz, W., Skoczeń, S., Leń, M., Kania, K., & Pawińska-Wąsikowska, K. (2024). Use of Granulocyte Transfusions in the Management of Severe Infections Among Children with Neutropenia. Journal of personalized medicine, 14(11), 1107. https://doi.org/10.3390/jpm14111107