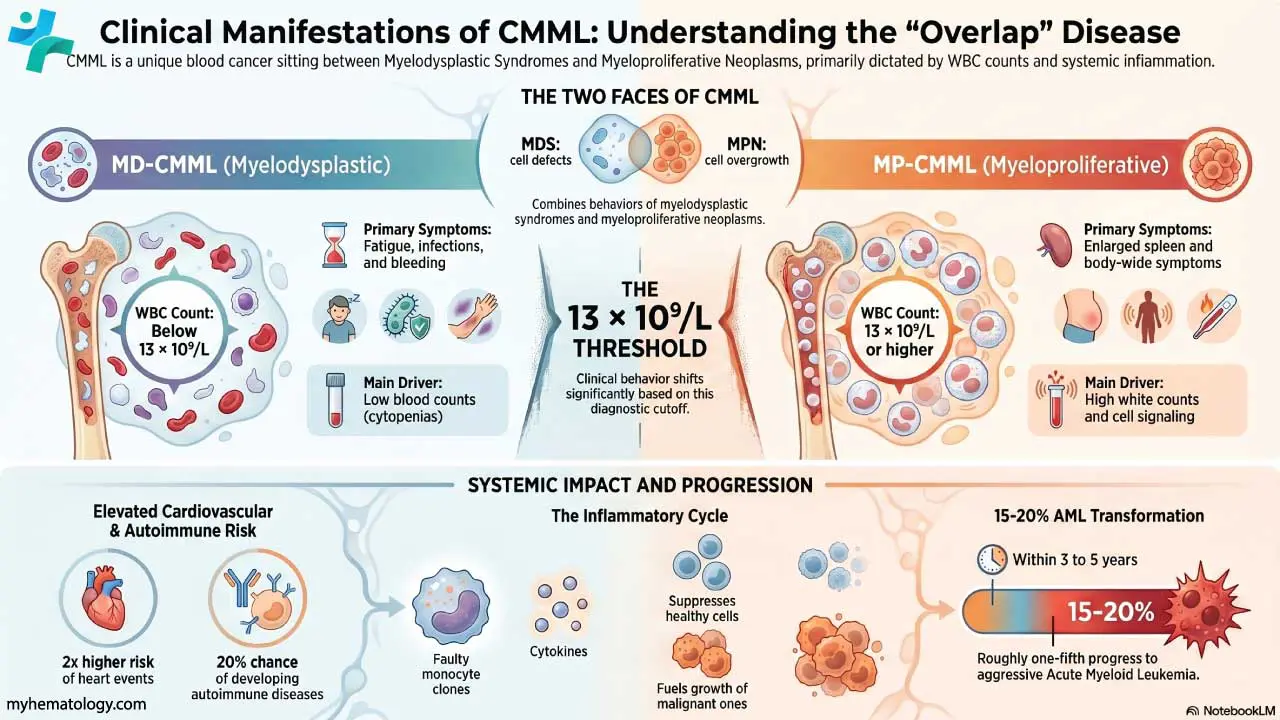
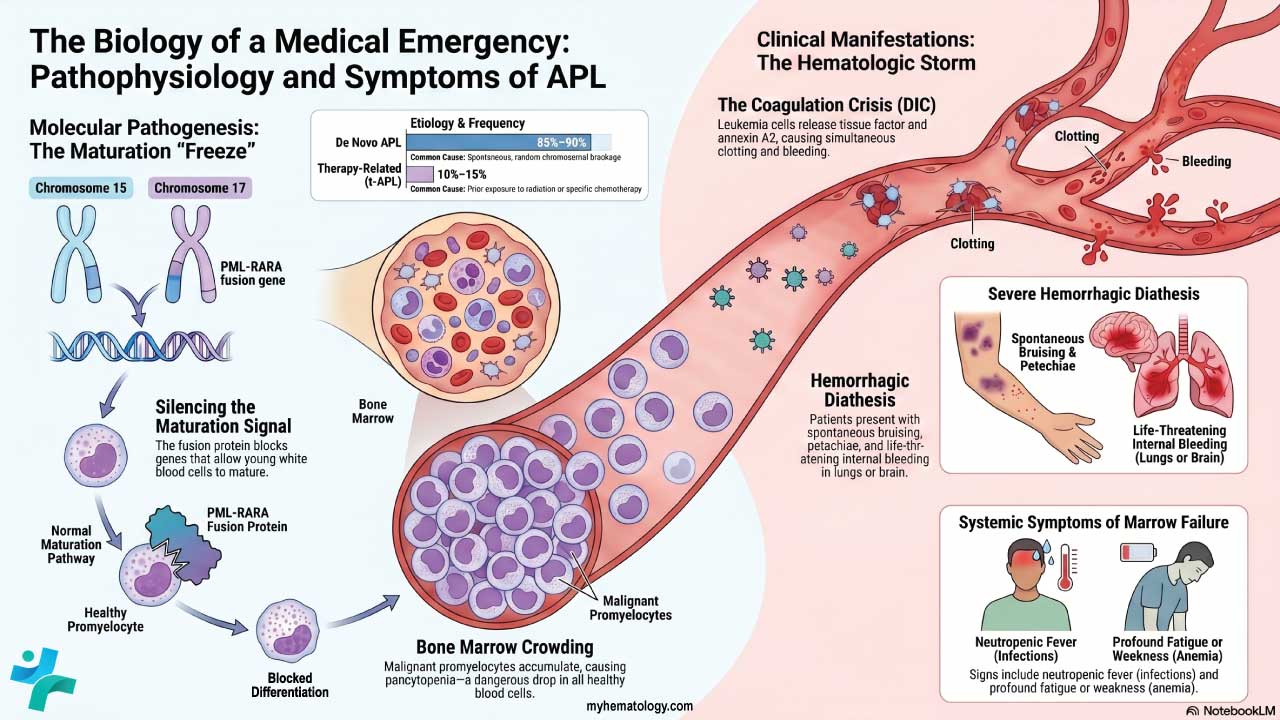
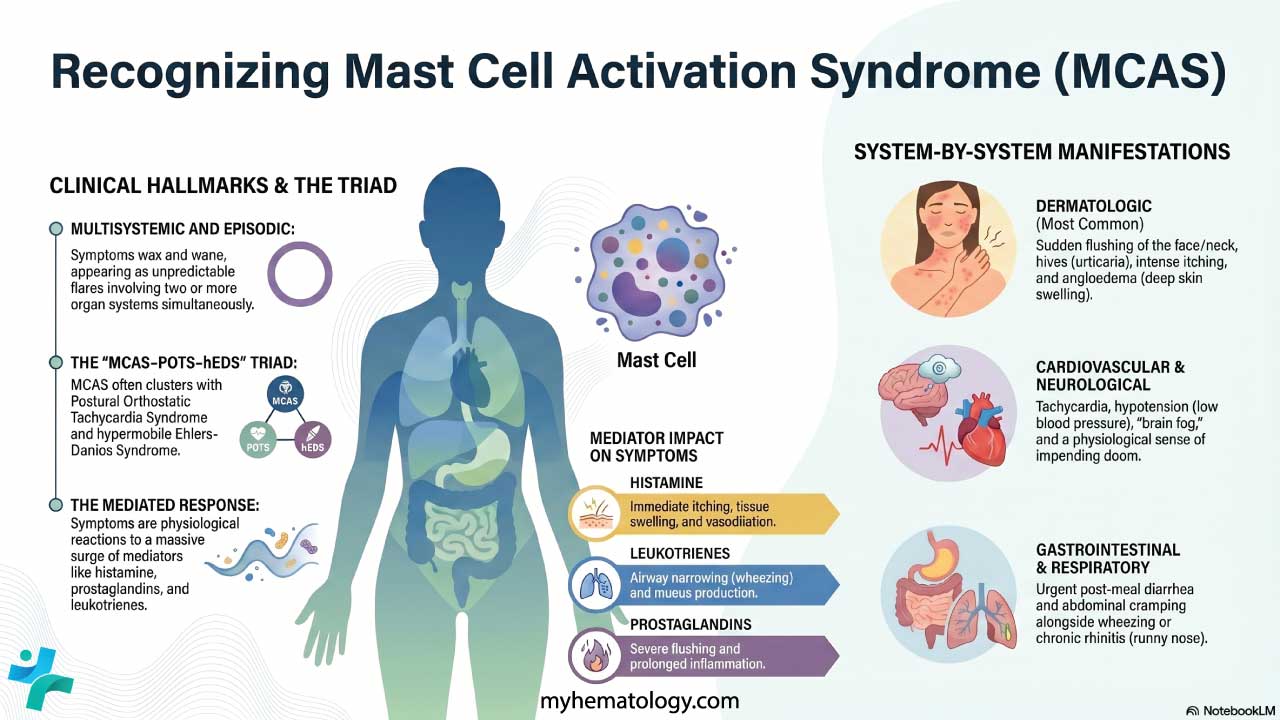
Procedure-At-A-Glance
May-Grünwald Giemsa (MGG) is a two-step Romanowsky stain used worldwide for blood smears, bone marrow aspirates, and cytology samples, and is recommended by the ICSH for bone marrow morphology.
- Fix the bone marrow aspirate smear in absolute methanol (typically for 10-15 minutes).
- Fully cover the slide with a 1:1 mixture of May-Grünwald stain and phosphate buffer and incubate (typically for 10 minutes). Decant the mixture.
- Fully cover the slide with a 1:10 dilution of Giemsa stain with phosphate buffer pH 6.8 and incubate (typically for 15 minutes). Decant the mixture.
- Wash the slide with slow running tap water and air dry.
- Mount the slide with Depex, and cover the zone of morphology with a cover slip, making it ready for microscopy.
If you have ever seen a textbook image of a blood smear with deep purple nuclei, pink red cells, and brightly stained granules, you were almost certainly looking at a Romanowsky stain. May-Grünwald Giemsa, or MGG, is one of the most important members of that family. It is the stain that lets a hematologist tell a normal blood-forming marrow apart from one with leukemia, and it is the workhorse stain in cytology labs for fine-needle aspirates and body fluids [1,2].
This article walks through what MGG is, how it works, how to perform it, and how to read it.
What MGG Is and Why It Matters
May-Grünwald Giemsa is a two-step Romanowsky stain used to color cells on a slide so each one can be examined under a microscope. It is built from two separate solutions applied in sequence: May-Grünwald (eosin and methylene blue in methanol) followed by Giemsa (eosin, methylene blue, and azure dyes). The combination produces the rich palette of blues, purples, and pinks that allows different cell types to be told apart at a glance.
In hematology, MGG is the recommended stain for bone marrow aspirate smears under the International Council for Standardization in Haematology (ICSH) guidelines [1]. It is used to diagnose leukemias such as chronic lymphocytic leukemia and chronic myeloid leukemia, to investigate anemias including aplastic anemia, and to detect bone marrow infiltration by infections or metastatic cancer. In cytology, it is used on fine-needle aspirate (FNAC) samples and on cells from cerebrospinal, pleural, and synovial fluids [2].
Principle of May-Grünwald Giemsa Stain
The MGG stain works by pairing a basic dye with an acidic dye and letting each one bind to the part of the cell with the opposite charge. This is the core of every Romanowsky stain, sometimes called the Romanowsky effect.
Here is the dye–substrate relationship, which is the single most important concept to remember:
- Basic dyes (azure B and methylene blue) are positively charged. They bind to acidic, negatively charged components of the cell, especially nucleic acids in the nucleus and RNA in the cytoplasm. These structures stain blue to deep purple.
- Acidic dyes (eosin Y) are negatively charged. They bind to basic, positively charged components, especially hemoglobin in red blood cells and basic cytoplasmic proteins. These structures stain pink to orange.
A handy way to remember it: opposites attract.
- Basic dye → acidic target → blue-purple.
- Acidic dye → basic target → pink-orange.
The two-step design is what makes MGG especially good for bone marrow. May-Grünwald goes first and brings out cytoplasmic detail. Giemsa follows and sharpens nuclear chromatin and granule colors. The end result is finer separation of immature cells and clearer differentiation of granule types than a single-step stain typically provides [2,4].
Components of the Stain
Two solutions, each with a defined chemistry:
May-Grünwald solution
- Eosin Y — acidic dye, stains basic cytoplasmic components pink to orange
- Methylene blue — basic dye, stains acidic nuclear and granule components blue to purple
Giemsa solution
- Azure B (and azure II) — oxidized methylene blue derivatives, drive the deep purple color of nuclei and azurophilic granules
- Eosin Y — same acidic dye as in May-Grünwald, reinforces cytoplasmic color
- Methylene blue chloride — interacts with the azures to produce the characteristic Romanowsky-Giemsa effect
It is the combination of azure B with eosin Y that creates the polychromatic blues, purples, and pinks Romanowsky stains are known for [4].
Materials
You will need:
- May-Grünwald dye powder
- Giemsa dye powder
- Absolute methanol (acetone-free)
- Glycerol
- Phosphate buffer at pH 6.8
- Coplin jars or staining racks, filter paper, and a mounting medium such as DePeX
- Long coverslips for the morphology zone
A Safety Note
Methanol is flammable and toxic by ingestion or inhalation, and dye powders are respiratory irritants. Prepare stock solutions in a fume hood and wear gloves and eye protection. Methanol-containing waste must go into a labeled hazardous-waste container. DO NOT pour it down the sink.
Protocol
Specific timings vary slightly by manufacturer; check your reagent's package insert before starting. The following follows the ICSH-aligned approach used in most teaching laboratories [1,2].
Preparation of May-Grünwald stain
- Dissolve 0.3 g of May-Grünwald dye in 100 mL absolute methanol in a 250 mL conical flask.
- Warm the mixture to 50°C in a water bath for several hours, then let it to cool to room temperature.
- Stir the solution on a magnetic stirrer for 24 hours.
- Filter, label with the preparation date, and the stain is ready to use.
Preparation of Giemsa stain
- Add 1.0 g of Giemsa dye to 66 mL of glycerol in a conical flask. Warm at 50°C for 1–2 hours.
- Cool to room temperature, then add 66 mL of absolute methanol.
- Allow the mixture to dissolve over 2-3 days, mixing periodically.
- Filter before use
Staining a bone marrow aspirate smear
- Prepare a 1:1 working solution of May-Grünwald stain and phosphate buffer (pH 6.8). Mix well.
- Prepare a 1:10 dilution of Giemsa stain in phosphate buffer (pH 6.8). Mix well.
- Fix the air-dried bone marrow aspirate smear in absolute methanol for 10–15 minutes.
- Cover the slide completely with the May-Grünwald working solution and incubate for 10 minutes.
- Decant. Cover the slide completely with the Giemsa working solution and incubate for 15 minutes.
- Decant and rinse gently with slow running tap water.
- Wipe the back and edges of the slide with a lint-free wipe. Do not touch the smear itself.
- Dry the slide with a hair dryer on low (no more than 10 seconds at a time) or air-dry it tilted on a rack.
- Mount with DePeX and apply a long coverslip across the morphology zone.
- The slide is ready for microscopy.
Interpretation of MGG Stain
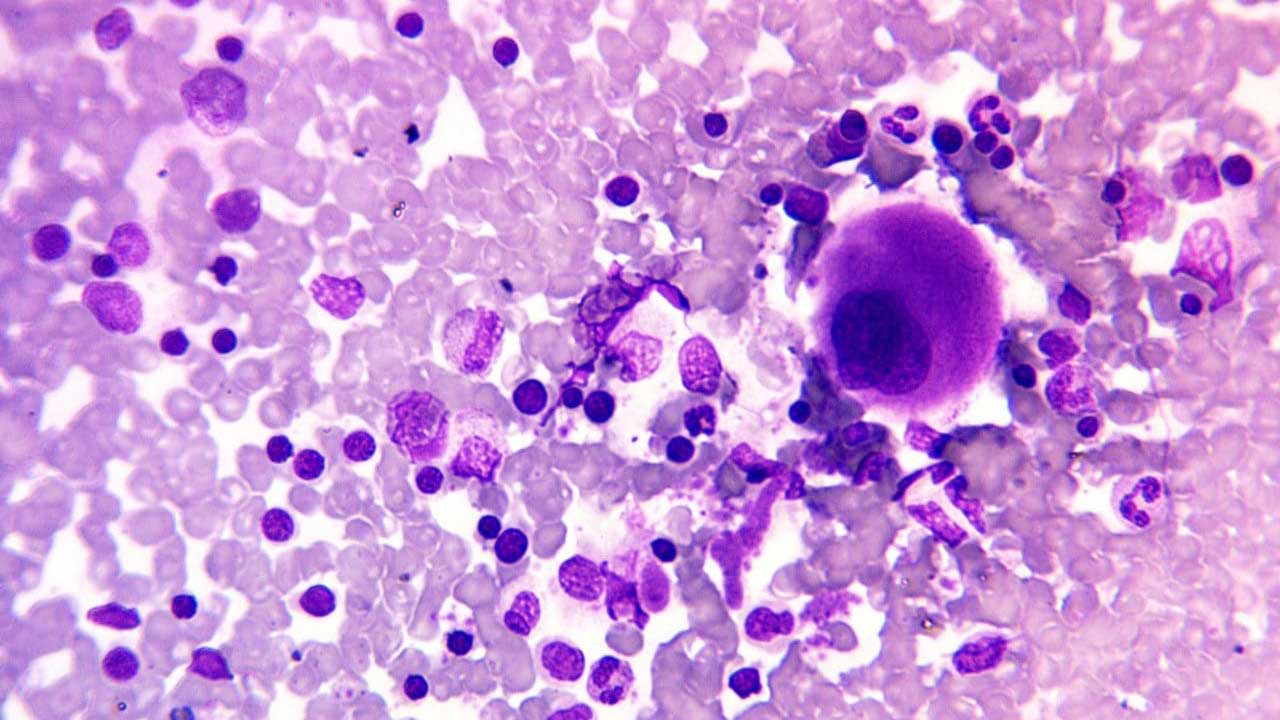
Reading a marrow smear is a layered task. Start at low power and work up. Always know the patient's clinical context before you sit down at the microscope — a marrow with many blasts means very different things in a 5-year-old with fever versus a 75-year-old being staged for myeloma.
At 10× magnification (low power)
Use this view to get a "lay of the land":
- Is the marrow hypercellular (crowded with cells) or hypocellular (sparse)?
- Are the cell trails coming off the marrow particles normal, diluted, or unusually rich?
- How many megakaryocytes are present, and do they look normal?
- Are there abnormal clumps of cells suggesting infiltration by lymphoma or metastatic cancer?
At 40× magnification (high power)
Now move closer and look at lineages:
- Is erythropoiesis (red cell production) normoblastic, megaloblastic, or dysplastic?
- Are all maturation stages of the myeloid and lymphoid lineages present, including blasts?
- Are plasma cells and macrophages normal in number and appearance?
- Is there evidence of metastatic cells — for example, clusters that do not belong in marrow?
At 100× magnification (oil immersion)
Use oil immersion to confirm fine cytoplasmic and nuclear features. Look for inclusions like Auer rods — needle-shaped reddish-purple structures inside the cytoplasm of some leukemic myeloid cells. Auer rods are made of fused azurophilic granules. Their presence confirms the cell is myeloid and strongly supports a diagnosis of acute myeloid leukemia (AML); densely packed multiple Auer rods ("faggot cells") are characteristic of acute promyelocytic leukemia.
Troubleshooting
- Insufficient staining time
- Dilute or expired stain
- Buffer too acidic
- Inadequate methanol fixation
- Over-washing
- Increase staining time
- Check working dilution
- Adjust buffer toward 6.8–7.2
- Use fresh filtered stain
- Ensure full methanol fixation
- Reduce wash
- Excessive staining time
- Stain too concentrated
- Buffer too alkaline
- Insufficient washing
- Decrease staining time
- Dilute stain further
- Adjust buffer toward 6.8
- Lengthen wash
- Uneven smear
- Incomplete drying before fixation
- Partial slide submersion
- Precipitate
- Improve smear technique
- Air-dry fully
- Fully submerge slide
- Clean glassware
- Filter stain
- Delayed fixation
- Over-drying
- Contaminated reagents
- Dye precipitation
- Fix promptly with fresh methanol
- Avoid excess drying
- Use fresh distilled water
- Filter Giemsa just before use
- Buffer too alkaline
- Insufficient eosin uptake
- Adjust buffer toward 6.8
- Increase May-Grünwald time
A practical quality-control habit
Run a known control slide (a normal peripheral blood smear) in each batch. If the control looks right, your patient slide should too.
How MGG Compares with Related Methods
The original MGG method is over a century old, but the field around it has not stood still.
- Leishman stain is a single-step Romanowsky stain widely used for peripheral blood smears. It is faster and simpler than MGG. For bone marrow, MGG is generally preferred because the two-step design tends to give clearer separation of cytoplasmic and nuclear detail [1].
- Wright stain and Wright-Giemsa are the standard alternatives in many North American laboratories. ICSH guidelines accept both Wright-Giemsa and MGG as suitable for bone marrow smears [1].
- Modified ultrafast Giemsa (MUFG) is a rapid variant developed for fine-needle aspirate cytology, where quick on-site reads matter. Studies have shown comparable diagnostic quality to standard MGG with substantially shorter staining time [3].
- Immunohistochemistry and flow cytometry complement morphology rather than replace it. They identify proteins and cell-surface markers, but they cannot show a cell's shape, granulation, or chromatin pattern.
- Digital morphology and AI are increasingly applied to MGG-stained slides. Systems such as CellaVision pre-classify cells and present them to the hematologist for review, and emerging AI tools assist in flagging dysplastic features in suspected myelodysplastic neoplasms. The stain has not changed; the way we look at it has.
Frequently Asked Questions (FAQs)
What is the May-Grünwald Giemsa (MGG) stain used for?
May-Grünwald Giemsa (MGG) is a two-step Romanowsky stain used to color blood cells, bone marrow cells, and cells in body fluids so they can be examined under a microscope. It is used in three main settings: hematology (counting and classifying white blood cells, diagnosing leukemia and anemia), cytology (looking at cells from fine-needle aspiration biopsies and fluids like cerebrospinal or pleural fluid), and histology (assessing bone marrow biopsies). It is recommended by the International Council for Standardization in Haematology (ICSH) as one of the routine stains for bone marrow aspirate smears.
Why is May-Grünwald Giemsa (MGG) stain better than Leishman stain for bone marrow analysis?
Both are Romanowsky stains and both work well on peripheral blood. MGG is favored for bone marrow because its two-step design separates cytoplasmic and nuclear staining: May-Grünwald handles cytoplasmic detail first, and Giemsa then sharpens nuclear and chromatin contrast. The result is generally cleaner differentiation of immature cell types and granule patterns, which matters when assessing complex marrow samples. Leishman is simpler and faster, and it remains adequate for routine peripheral blood smears.
Why does the buffer have to be pH 6.8?
The buffer pH controls how the acidic and basic dyes ionize, which in turn controls how strongly each dye binds to its target. At pH 6.8 the balance produces pink-orange red blood cells, blue-purple nuclei, and the right shades of granule color. If the buffer is too alkaline, red blood cells turn bluish or greenish because the basic dye binds them too strongly. If it is too acidic, nuclear detail looks washed out. pH drift is the most common reason an MGG slide does not look right.
What does an Auer rod look like, and why does it matter?
An Auer rod is a needle-shaped or rod-shaped reddish-purple structure inside the cytoplasm of certain leukemic cells. It is made of fused azurophilic granules. When you see one, the cell is definitely from the myeloid lineage, and Auer rods strongly support a diagnosis of acute myeloid leukemia (AML). Multiple Auer rods clustered in one cell (so-called "faggot cells") are typical of acute promyelocytic leukemia (APL), a subtype that needs urgent treatment.
Is MGG staining still relevant in the era of flow cytometry and digital pathology?
Yes. Flow cytometry and molecular tests give powerful information about cell surface markers and genetics, but they do not show what a cell looks like. Morphology on an MGG-stained smear is still the first step in diagnosing most blood disorders, and international guidelines continue to recommend it. Modern labs increasingly pair MGG with digital imaging systems (such as CellaVision) and AI-assisted pre-classification, but the underlying stain has not changed.
Are there faster versions of the MGG stain?
Yes. Modified ultrafast Giemsa (MUFG) is a rapid variant developed for fine-needle aspiration cytology, where on-site results can guide whether enough material has been collected. Studies comparing MUFG with standard MGG report comparable diagnostic quality with markedly shorter staining times, which is useful in busy cytology services. Standard MGG remains the reference method for bone marrow aspirates.
Glossary of Related Medical Terms
- Romanowsky stain — A family of blood and bone marrow stains that combine an acidic dye (eosin) with an oxidized basic dye (methylene blue and its azures) to produce the characteristic blue, purple, and pink colors. MGG, Wright, Leishman, and Giemsa are all members.
- Azure B — An oxidation product of methylene blue. It is the key basic dye responsible for the deep purple color seen in nuclei and certain granules.
- Eosin Y — An acidic, negatively charged dye that binds to positively charged proteins like hemoglobin, giving red blood cells their pink-orange color.
- Bone marrow aspirate — A liquid sample of bone marrow, usually drawn from the back of the hip bone, used to look at individual blood-forming cells under the microscope.
- Smear (film) — A thin layer of cells spread on a glass slide so each cell can be examined separately.
- Erythropoiesis — The process of red blood cell production in the bone marrow.
- Myeloid lineage — The family of bone marrow cells that gives rise to neutrophils, eosinophils, basophils, and monocytes.
- Lymphoid lineage — The family of cells that gives rise to lymphocytes (T cells, B cells, NK cells).
- Megakaryocyte — A very large bone marrow cell that releases platelets into the blood.
- Auer rod — A needle-shaped, reddish-purple structure inside the cytoplasm of some leukemic myeloid cells. Made of fused azurophilic granules. When seen, it confirms the cell is myeloid and points toward acute myeloid leukemia (AML).
- Dysplasia — Abnormal cell shape or maturation. In bone marrow, dysplastic features suggest disorders like myelodysplastic syndromes (MDS).
- Hypercellular / hypocellular marrow — Marrow that is more crowded with cells than normal (hypercellular) or emptier than normal (hypocellular).
- Phosphate buffer — A pH-stable salt solution. In MGG, a buffer at pH 6.8 keeps the dye balance correct so colors come out as expected.
- Differential count — A count of how many of each white blood cell type are present in a sample, expressed as a percentage.
- Fine-needle aspiration cytology (FNAC) — A diagnostic procedure that uses a thin needle to sample cells from a lump or organ, often stained with MGG.
Disclaimer: This protocol is for educational purposes only. Local laboratory standard operating procedures take precedence. It is not intended to be a substitute for informed professional medical advice, diagnosis, or treatment. Always consult a qualified healthcare professional for clinical decision-making. While the information presented here is derived from credible medical sources and is believed to be accurate and up-to-date, it is not guaranteed to be complete or error-free. See additional information.
References
- Lee, S. H., Erber, W. N., Porwit, A., Tomonaga, M., Peterson, L. C., & International Council for Standardization In Hematology (2008). ICSH guidelines for the standardization of bone marrow specimens and reports. International journal of laboratory hematology, 30(5), 349–364. https://doi.org/10.1111/j.1751-553X.2008.01100.x
- Piaton, E., Fabre, M., Goubin-Versini, I., Bretz-Grenier, M. F., Courtade-Saïdi, M., Vincent, S., Belleannée, G., Thivolet, F., Boutonnat, J., Debaque, H., Fleury-Feith, J., Vielh, P., Egelé, C., Bellocq, J. P., Michiels, J. F., & Cochand-Priollet, B. (2016). Guidelines for May-Grünwald-Giemsa staining in haematology and non-gynaecological cytopathology: recommendations of the French Society of Clinical Cytology (SFCC) and of the French Association for Quality Assurance in Anatomic and Cytologic Pathology (AFAQAP). Cytopathology : official journal of the British Society for Clinical Cytology, 27(5), 359–368. https://doi.org/10.1111/cyt.12323
- Deepthi, B., Prayaga, A. K., & Rukmangadha, N. (2022). Comparison of Modified Ultrafast Giemsa Stain with the Standard May Grunwald Giemsa Stain in FNAC of Various Organs. Journal of cytology, 39(4), 174–179. https://doi.org/10.4103/joc.joc_43_22
- Horobin R. W. (2011). How Romanowsky stains work and why they remain valuable - including a proposed universal Romanowsky staining mechanism and a rational troubleshooting scheme. Biotechnic & histochemistry : official publication of the Biological Stain Commission, 86(1), 36–51. https://doi.org/10.3109/10520295.2010.515491
- Bain BJ, Bates I, Laffan MA. Dacie and Lewis Practical Haematology: Expert Consult: Online and Print 12th Edition (Elsevier). 2016.
- Wittekind D. (2003). Traditional staining for routine diagnostic pathology including the role of tannic acid. 1. Value and limitations of the hematoxylin-eosin stain. Biotechnic & histochemistry : official publication of the Biological Stain Commission, 78(5), 261–270. https://doi.org/10.1080/10520290310001633725