Key Takeaways
Drug-induced immune hemolytic anemia are caused by drugs that trigger an immune response to attack red blood cells (RBCs), leading to hemolytic anemia.
- Mechanism ▾: Drug-Induced Immune Hemolytic Anemia occurs when a drug triggers an immune response via hapten, neo-antigen, or autoimmune models leading to antibody-mediated destruction of red blood cells.
- Signs & Symptoms ▾: Patients typically present with a clinical triad of jaundice, dark urine, and splenomegaly, often accompanied by systemic symptoms such as fatigue and tachycardia.
- Complications ▾: Fulminant cases can rapidly progress to life-threatening multi-organ failure, including acute kidney injury, disseminated intravascular coagulation (DIC), and cardiovascular shock.
- Diagnosis ▾: The diagnostic process of Drug-Induced Immune Hemolytic Anemia centers on a positive Direct Antiglobulin Test (DAT) and markers of active hemolysis, supplemented by specialized drug-dependent antibody testing.
- Differential Diagnosis ▾: Clinicians must distinguish Drug-Induced Immune Hemolytic Anemia from other immune conditions like Warm AIHA and non-immune triggers like G6PD deficiency by evaluating the eluate results and peripheral smear.
- Treatment & Management ▾: The cornerstone of management is the immediate and permanent discontinuation of the offending drug, supported by vigorous hydration, renal protection, and, in severe cases, plasma exchange.
*Click ▾ for more information
Introduction
Drug-induced immune hemolytic anemia is a rare condition where a medication triggers your body's immune system to attack your own red blood cells (RBCs). This attack leads to the premature destruction of RBCs, a process called hemolysis.
Hemolysis refers to the breakdown of red blood cells (RBCs) and the release of their contents into the surrounding fluid, typically blood plasma. This breakdown can occur within the bloodstream (intravascular hemolysis) or outside the bloodstream in organs like the spleen (extravascular hemolysis). Hemolysis can cause various issues as RBCs are essential for carrying oxygen throughout your body.
Hemolysis is significant for several reasons:
- Reduced Oxygen Delivery: RBCs carry oxygen throughout the body bound to a molecule called hemoglobin. When RBCs rupture, less oxygen is available to be delivered to tissues, potentially leading to fatigue, weakness, and shortness of breath.
- Kidney Complications: Hemoglobin released from lysed RBCs can overload the kidneys, leading to potential kidney damage or failure if left untreated.
- Jaundice: Hemoglobin is broken down into a yellow pigment called bilirubin. When excessive hemolysis occurs, the liver can become overwhelmed by the amount of bilirubin, causing a yellowish discoloration of the skin and eyes known as jaundice.
Intravascular vs Extravascular Hemolysis
Hemolysis, the destruction of red blood cells (RBCs), can occur in two main locations:
- Extravascular Hemolysis: This happens outside the bloodstream, primarily in organs like the spleen and liver. These organs contain specialized cells called macrophages that are responsible for clearing away old or damaged RBCs. In extravascular hemolysis, these macrophages remove and break down the RBCs in a controlled manner.
- Intravascular Hemolysis: This occurs within the bloodstream itself. Here, the RBCs are directly lysed (broken open) by various factors, releasing their contents directly into the plasma. This can be a much more rapid and aggressive form of hemolysis compared to the extravascular process.
Intravascular vs Extravascular Hemolysis Summary
| Feature | Intravascular Hemolysis | Extravascular Hemolysis |
| Site of destruction | Within bloodstream | Outside bloodstream (primarily spleen) |
| Cause | Direct attack on RBC membrane by complement or immune cells | Phagocytosis of RBCs coated with antibodies by macrophages in the spleen |
| Initiating factors | Severe infections, toxins, incompatible blood transfusions | Autoimmune diseases, hereditary RBC defects |
| Hemoglobin release | Rapid release of free hemoglobin and iron into plasma | Gradual release of hemoglobin following RBC breakdown by macrophages |
| Clinical presentation | More severe symptoms due to rapid hemolysis (hemoglobinuria, shock) | Less severe symptoms initially, may progress to jaundice and anemia |
| Examples | Severe transfusion reactions, hemolytic uremic syndrome | Autoimmune hemolytic anemia (AIHA), hereditary spherocytosis |
Mechanism Models of Drug-Induced Immune Hemolytic Anemia
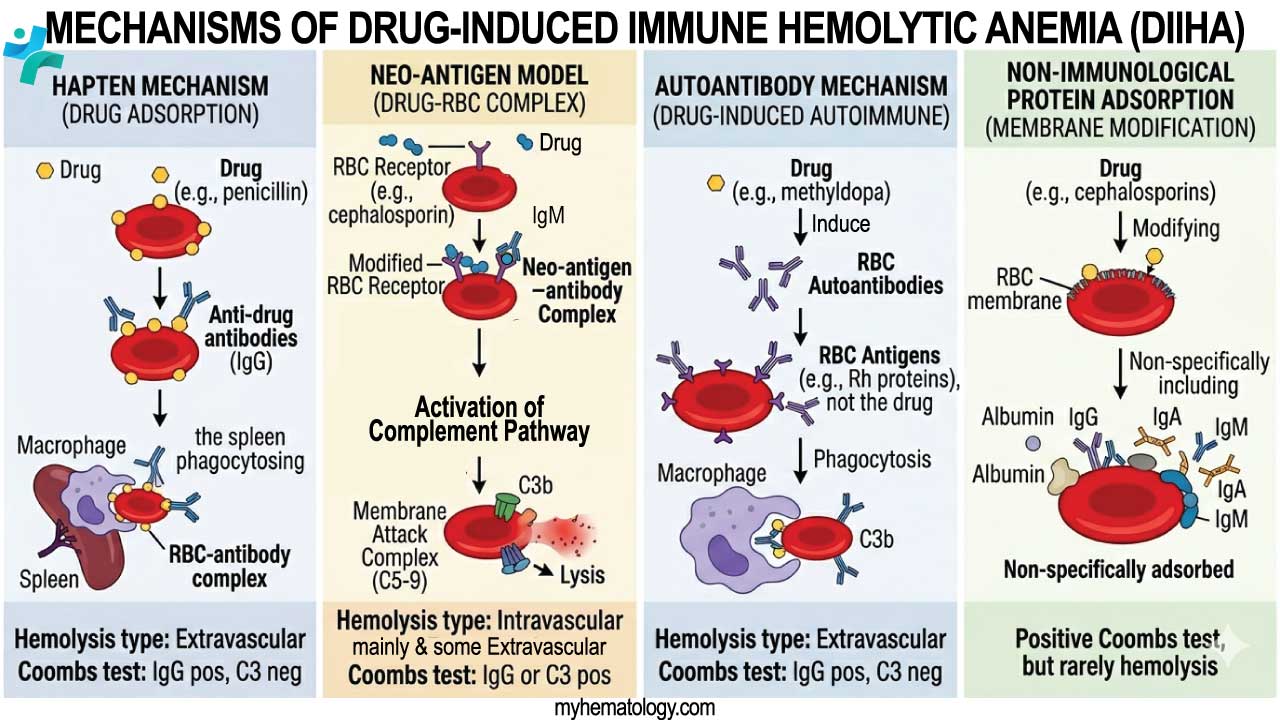
Drug-Dependent: The Hapten (Drug Adsorption) Model
This is the classic "Penicillin-type" mechanism in Drug-Induced Immune Hemolytic Anemia. It occurs when a drug binds firmly (covalently) to the proteins on the RBC membrane. Because the drug is covalently bound, the DAT is strongly positive for IgG, but the eluate is negative (unless the drug is added back into the testing system).
- The Molecular Event: The drug molecule (hapten) is too small to be seen by the immune system on its own. However, once it coats the RBC, the drug-protein combination becomes an immunogenic target.
- The Antibody: Typically IgG. These antibodies recognize the drug itself or the drug-membrane junction.
- Hemolysis Type: Primarily Extravascular. Macrophages in the spleen recognize the IgG-coated RBCs and "nibble" at the membrane, leading to spherocytosis and eventual destruction.
Drug-Dependent: The Neo-antigen (Drug-Induced) Model
Formerly often referred to as the "Immune Complex" model, the modern "Neo-antigen" theory is the mechanism for drugs like Ceftriaxone or Piperacillin. This is the most dangerous form of Drug-Induced Immune Hemolytic Anemia. It can cause sudden, life-threatening hemolysis, hemoglobinuria, and acute renal failure.
- The Molecular Event: The drug does not bind covalently. Instead, it forms a loose, non-covalent association with a specific RBC membrane protein (like Rh or Glycophorin). This creates a "new" antigen also known as a "Neo-antigen."
- The Antibody: Often IgM (though IgG occurs). These antibodies only bind to the RBC when the drug is physically sitting in that specific "pocket" on the membrane.
- Hemolysis Type: Intravascular. These antibodies are potent activators of the complement cascade. This leads to the formation of the Membrane Attack Complex (MAC) and rapid RBC lysis within the vessels.
Drug-Independent: The Autoimmune Model
In this Drug-Induced Immune Hemolytic Anemia model, the drug acts as a "trigger" that causes the immune system to lose self-tolerance. The most famous example is Methyldopa, though Immune Checkpoint Inhibitors (ICIs) are the modern equivalent. This is the only model where the eluate is positive. Even after we stop the drug, the DAT may remain positive for months because the immune system has been "reprogrammed" to attack itself.
- The Molecular Event: The drug somehow interferes with T-cell or B-cell regulation. The resulting antibodies are "true" autoantibodies; they target intrinsic RBC antigens (usually the Rh complex) and do not require the drug to be present for binding.
- The Antibody: IgG.
- Hemolysis Type: Extravascular. It is indistinguishable from Warm Autoimmune Hemolytic Anemia (WAIHA).
Non-immunological Protein Adsorption (NIPA)
This is a "false positive" mechanism of Drug-Induced Immune Hemolytic Anemia often seen with beta-lactamase inhibitors (e.g., Tazobactam, Clavulanate). This is a diagnostic trap. We might see a positive DAT and assume drug-induced immune hemolytic anemia, but the lack of clinical hemolysis (normal LDH, haptoglobin, and reticulocyte count) confirms it is likely NIPA.
- The Molecular Event: The drug modifies the RBC membrane in a way that allows plasma proteins (Albumin, IgG, IgA, IgM, Complement) to stick to the surface non-specifically.
- The Result: You get a Positive DAT, but there is no hemolysis.
Comparison Between Models
| Feature | Hapten (Drug Adsorption) | Neo-antigen (Drug-Induced) | Autoimmune (Drug-Independent) |
| Typical Drug | Penicillin, Piperacillin | Ceftriaxone, NSAIDs | Methyldopa, Fludarabine, ICIs |
| Binding | Covalent (Firm) | Non-covalent (Loose) | None (Targets self-antigen) |
| Hemolysis | Extravascular | Intravascular (Severe) | Extravascular |
| DAT Result | IgG positive | C3 positive (often) | IgG positive |
| Eluate Result | Negative | Negative | Positive |
| RBC Morphology | Spherocytes | Fragmented cells (Schistocytes) | Spherocytes |
Common Culprit of Drug-Induced Immune Hemolytic Anemia
Tier 1: The "Modern Big Three" (Most Common)
These three drugs account for the vast majority of drug-induced immune hemolytic anemia cases in hospitals today.
- Ceftriaxone: Currently the most significant cause of drug-induced immune hemolytic anemia worldwide. It is uniquely dangerous because it often causes severe intravascular hemolysis via the Neo-antigen mechanism. In pediatric populations, the mortality rate can be as high as 30–40%.
- Piperacillin (often with Tazobactam): A "silent" leader. Research from indicates that up to 18% of patients receiving this drug develop drug-dependent antibodies. While most remain asymptomatic, it is a frequent cause of unexplained hemoglobin drops in the ICU.
- Cefotetan: Frequently implicated in surgical prophylaxis. Because it is often given as a one-time dose, the hemolysis may occur after the patient has been discharged, leading to diagnostic delays.
Tier 2: The Oncology & Immunology Triggers (Emerging)
With the rise of immunotherapy, a new class of "Drug-Independent" drug-induced immune hemolytic anemia has climbed the hierarchy.
- Immune Checkpoint Inhibitors (ICIs): Drugs like Nivolumab and Pembrolizumab trigger drug-induced immune hemolytic anemia in about 0.5% of treated patients. Unlike Tier 1 drugs, these cause a true autoimmune reaction that can persist long after the drug is stopped.
- Fludarabine & Cladribine: These chemotherapy agents are notorious for triggering a "Drug-Independent" AIHA, particularly in patients with existing Chronic Lymphocytic Leukemia (CLL).
- Platinum-based Agents (Oxaliplatin): Often cause hemolysis via the "Immune Complex" or "Neo-antigen" model, frequently presenting during the second or third cycle of treatment.
Tier 3: The "Textbook Relics" (Historical)
These drugs are the classic examples found in older literature but are now rarely the cause of drug-induced immune hemolytic anemia in modern clinical settings.
- Methyldopa: The original "Autoimmune Model" poster child. Since it is rarely used for hypertension today (except occasionally in pregnancy), it has fallen to the bottom of the hierarchy.
- High-Dose Penicillin G: While still capable of causing drug-induced immune hemolytic anemia (Hapten model), modern dosing and the use of alternative antibiotics make this a much rarer occurrence than in the 1970s.
| Rank | Drug | Mechanism | Typical Severity | Summary |
| 1 | Ceftriaxone | Neo-antigen | Fulminant / Fatal | Can trigger DIC; most common in kids. |
| 2 | Piperacillin | Hapten / Neo-antigen | Moderate / Subclinical | High antibody frequency (18%). |
| 3 | Cefotetan | Neo-antigen | Moderate / Severe | Common in surgical prophylaxis. |
| 4 | ICIs | Drug-Independent | Chronic / Variable | Behaves like Warm AIHA; needs Rituximab. |
| 5 | NSAIDs | Neo-antigen | Moderate | Often overlooked; look for Diclofenac. |
Clinical Presentation
The clinical presentation of Drug-Induced Immune Hemolytic Anemia can range from a slow, nearly imperceptible decline in hemoglobin to a fulminant, life-threatening crisis. The manifestations are primarily dictated by whether the hemolysis is intravascular or extravascular.
The Timeline of Onset
Understanding the "tempo" of the disease is the first step in clinical diagnosis.
- Acute/Hyperacute Onset (Hours to Days): Typical of the Neo-antigen model (e.g., Ceftriaxone). If a patient has been previously sensitized, symptoms can begin within minutes to hours of a single dose.
- Subacute/Gradual Onset (Weeks to Months): Typical of the Hapten model (high-dose Penicillin) or the Autoimmune model (Methyldopa, ICIs). Patients often present with progressive fatigue and "vague" symptoms of worsening anemia.
Classic Symptoms of Hemolysis
Regardless of the trigger, the breakdown of red blood cells produces a predictable triad of clinical signs.
- Jaundice & Scleral Icterus: As RBCs break down, heme is converted into unconjugated bilirubin. When the liver’s conjugating capacity is overwhelmed, the yellow pigment deposits in the skin and the whites of the eyes.
- Dark Urine (Hemoglobinuria): In severe intravascular hemolysis, free hemoglobin is filtered by the kidneys. This turns the urine a "coca-cola" or dark tea color. It is vital to distinguish this from hematuria (intact RBCs). Hemoglobinuria will show a positive dipstick for blood but a negative microscopic exam for RBCs.
- Splenomegaly: In extravascular hemolysis, the spleen is the primary site of RBC destruction. Constant "work hypertrophy" of the splenic macrophages can lead to a palpable, sometimes tender, enlarged spleen.
Systematic Clinical Manifestations
Clinical signs can be categorized by the body's compensatory response to falling oxygen levels.
| System | Manifestations | Underlying Pathophysiology |
| General | Pallor (conjunctival/palmar), Fatigue, Lethargy. | Reduced oxygen-carrying capacity. |
| Cardiovascular | Tachycardia, Flow murmurs, Palpitations. | Increased cardiac output to compensate for hypoxia. |
| Respiratory | Dyspnea on exertion, Tachypnea. | Air hunger due to reduced oxygen delivery to tissues. |
| Neurological | Lightheadedness, Syncope, Confusion. | Reduced cerebral oxygenation; common in elderly patients. |
| Gastrointestinal | Abdominal or back pain. | Often associated with rapid intravascular lysis (likely due to nitric oxide depletion). |
Severe and Life-Threatening Complications
In "Tier 1" drug cases (like Ceftriaxone), the clinical picture can escalate rapidly into multi-organ failure.
- Acute Kidney Injury (AKI): High levels of free hemoglobin are toxic to the renal tubules (Pigment Nephropathy). This is often preceded by flank pain and oliguria (decreased urine output).
- Disseminated Intravascular Coagulation (DIC): Massive hemolysis can trigger the coagulation cascade, leading to a paradoxical state of widespread clotting and severe bleeding.
- Hypovolemic/Cardiogenic Shock: Rapid, massive loss of RBC mass leads to cardiovascular collapse.
High-Yield Clinical "Red Flags"
When reviewing a patient’s chart, the presence of these signs should immediately trigger a medication review:
- Sudden Fever/Chills + Dark Urine: If this occurs shortly after an IV antibiotic dose, Drug-Induced Immune Hemolytic Anemia (Neo-antigen type) must be the top differential.
- Back Pain during Infusion: Similar to a hemolytic transfusion reaction, the sudden release of intracellular contents can cause severe, acute lumbar pain.
- Refractory Anemia: If a patient is being transfused but their hemoglobin fails to rise (or continues to drop), it suggests the "culprit drug" is still in the system, causing the destruction of both the patient's and the donor's RBCs.
Laboratory Investigations & Diagnosis
The laboratory diagnosis of Drug-Induced Immune Hemolytic Anemia is a specialized process that moves from general markers of hemolysis to complex, drug-specific serological testing.
The Baseline Hemolysis Panel
Before specialized drug testing begins, we must confirm that hemolysis is taking place.
- Complete Blood Count (CBC):
- Hemoglobin/Hematocrit: Typically shows a rapid, unexplained drop.
- MCV: Often elevated (macrocytosis) due to a high percentage of larger, immature reticulocytes.
- WBC & Platelets: In Drug-Induced Immune Hemolytic Anemia, these are usually normal, unless the drug is also causing concurrent immune destruction (e.g., Primary ITP or neutropenia).
- Peripheral Blood Smear:
- Spherocytes: Highly indicative of extravascular hemolysis (Hapten/Autoimmune models).
- Schistocytes/Fragmented Cells: May be seen in fulminant intravascular cases (Neo-antigen model).
- Polychromasia: Visual evidence of a robust reticulocyte response.
- Biochemical Markers:
- Lactate Dehydrogenase (LDH): Significantly elevated.
- Unconjugated Bilirubin: Elevated, correlating with jaundice.
- Haptoglobin: Low or undetectable, as it binds to free hemoglobin.
Direct Antiglobulin Test (DAT) & Eluate
The "Core Serology" determines if the immune system is involved.
- DAT (Direct Coombs): Usually positive.
- IgG Positive / C3d Negative: Typical of the Hapten Model (e.g., Penicillin).
- C3d Positive / IgG Variable: Typical of the Neo-antigen Model (e.g., Ceftriaxone), as these antibodies are potent complement fixers.
- The Eluate (CRITICAL STEP):
- Positive Eluate: Suggests the drug has triggered a "Drug-Independent" autoantibody (like Methyldopa or ICIs) that targets the RBC itself.
- Negative Eluate: A hallmark of "Drug-Dependent" Drug-Induced Immune Hemolytic Anemia. This is because the drug is washed away during the elution process, leaving the antibody with nothing to bind to in the test tube.
Specialized Drug-Dependent Antibody Testing
If the patient has hemolysis, a positive DAT, and a negative eluate, the lab must perform specialized "drug-augmented" testing.
A. Testing with Drug-Treated RBCs (Hapten Style)
Used for drugs that bind firmly to the cell (e.g., Penicillin, Piperacillin).
- The lab coats healthy O-type RBCs with the suspected drug.
- The patient's serum is added.
- If agglutination occurs with drug-treated cells but not with untreated cells, the diagnosis is confirmed.
B. Testing in the Presence of a Drug Solution (Neo-antigen Style)
Used for drugs that bind loosely (e.g., Cephalosporins, NSAIDs).
- The patient's serum, healthy O-type RBCs, and a concentrated solution of the drug are mixed together in a test tube.
- Agglutination or hemolysis in this "cocktail" but not in the drug-free control confirms the diagnosis.
Summary Table
| Investigation | Hapten (Penicillin-type) | Neo-antigen (Ceftriaxone-type) | Autoimmune (ICI-type) |
| DAT Pattern | IgG (+) | C3d (+), IgG (+/-) | IgG (+) |
| Eluate | Negative | Negative | Positive |
| Drug Requirement | Needs drug-coated cells | Needs drug in solution | No drug needed in lab |
| Site of Lysis | Extravascular | Intravascular | Extravascular |
Differential Diagnosis of Drug-Induced Immune Hemolytic Anemia
The "DAT-Positive" Differentials (Immune Causes)
When a patient has laboratory markers of hemolysis and a positive Direct Antiglobulin Test (DAT), the primary task is to distinguish Drug-Induced Immune Hemolytic Anemia from other forms of Autoimmune Hemolytic Anemia (AIHA).
| Condition | DAT Pattern | Eluate Result | Key Distinguishing Feature |
| Drug-Induced Immune Hemolytic Anemia (Dependent) | IgG or C3 (+) | Negative | Hemolysis resolves quickly after drug cessation. |
| Warm AIHA (WAIHA) | IgG and/or C3 (+) | Positive | True autoantibody; often requires steroids/rituximab. |
| Cold Agglutinin (CAD) | C3 (+) only | Negative | Symptoms triggered by cold; presence of cold agglutinin titer. |
| Evans Syndrome | IgG (+) | Positive | Concurrent Primary ITP |
A negative eluate in the face of a positive DAT and active hemolysis is the "smoking gun" for Drug-Induced Immune Hemolytic Anemia (specifically the Hapten or Neo-antigen models).
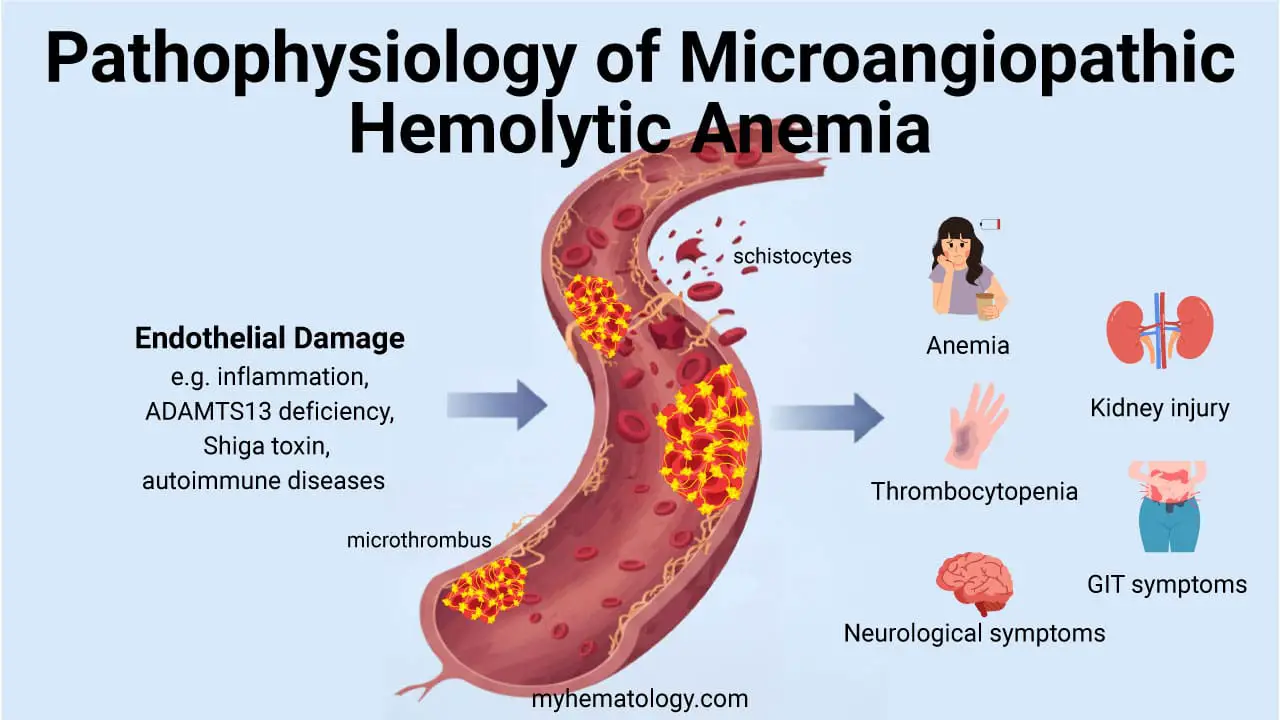
The "Schistocyte" Differentials (Microangiopathic)
If the peripheral smear shows schistocytes (fragmented cells), the differential must shift toward life-threatening Microangiopathic Hemolytic Anemias (MAHA).
- TTP (Thrombotic Thrombocytopenic Purpura): TTP can mimic the severe intravascular hemolysis seen in Ceftriaxone-induced DIIHA. TTP will have a negative DAT and severe thrombocytopenia (often < 30 x 109/L). Clinicians now use the Modified PLASMIC Score, which substitutes MCV for RDW-SD to better predict TTP risk before ADAMTS13 results are available.
- DIC (Disseminated Intravascular Coagulation): Severe DIIHA (Neo-antigen model) can actually trigger DIC. Look for prolonged PT/aPTT and low Fibrinogen, which are typical of DIC but not isolated Drug-Induced Autoimmmune Hemolytic Anemia.
The "Drug-Triggered" Mimics (Non-Immune)
Some patients experience hemolysis only after taking a drug, but the mechanism is not an antibody. This is the most common diagnostic pitfall.
- G6PD Deficiency: An enzymopathy, not an immune reaction. Oxidative drugs (e.g., Rasburicase, Nitrofurantoin, Primaquine) cause hemoglobin to precipitate (Heinz bodies). DAT is negative. The smear shows "Bite cells" and "Blister cells" rather than spherocytes.
- Hereditary Spherocytosis (HS): A cytoskeletal membrane defect. While HS causes spherocytes (like the Hapten model of Drug-Induced Autoimmune Hemolytic Anemia), the DAT is negative, and the patient will often have a lifelong history of intermittent jaundice or a family history of splenectomy.
Evans Syndrome vs. Drug-Induced Secondary Cytopenia
When a patient presents with both anemia and a low platelet count (e.g., 40 x 109/L), we must determine if this is:
- Evans Syndrome: A primary autoimmune failure involving WAIHA and Primary ITP.
- Drug-Induced: A single drug (like Amoxicillin or Cefoperazone) triggering antibodies against both RBCs and platelets.
Diagnostic Rule: If both cell lines recover within 5–7 days of stopping the suspected drug, it was a drug-induced secondary event. If the cytopenias persist or worsen, it points toward Evans Syndrome or an underlying lymphoproliferative disorder.
Treatment and Management
The management of Drug-Induced Immune Hemolytic Anemia requires rapid clinical decision-making. Unlike other forms of autoimmune hemolytic anemia where immunosuppression is the immediate go-to, Drug-Induced Immune Hemolytic Anemia management is primarily mechanical and supportive.
Step 1: The Golden Rule – Stop the Offending Drug
The absolute cornerstone of Drug-Induced Immune Hemolytic Anemia management is the immediate discontinuation of the suspected medication. Often, the patient is on multiple medications (e.g., a patient in the ICU on Piperacillin, Heparin, and Omeprazole).
- The Strategy: Stop all non-essential medications immediately. For essential antibiotics, switch to a structurally unrelated class (e.g., if Ceftriaxone is the suspected culprit, switch to a Macrolide or Fluoroquinolone, not another Cephalosporin).
- Expected Outcome: In drug-dependent Drug-Induced Immune Hemolytic Anemia (Hapten or Neo-antigen models), hemolysis typically begins to slow within 24 to 48 hours of drug clearance, though the DAT may remain positive for weeks.
Step 2: Acute Stabilization and the Transfusion Dilemma
In severe cases, profound anemia leads to hemodynamic instability, necessitating immediate packed red blood cell (pRBC) transfusion. However, this presents a classic clinical hurdle. Because the patient's serum is filled with antibodies, the laboratory crossmatch will often show that all donor blood is "incompatible" (pan-reactive).
We must never withhold life-saving blood due to a positive crossmatch in acute hemolysis. The blood bank will provide the "least incompatible" blood. Keep in mind that the transfused cells will also be destroyed if the offending drug is still circulating. Therefore, transfuse slowly and monitor closely for volume overload and worsening hemolysis.
Step 3: Advanced Clearance – Therapeutic Plasma Exchange (TPE)
This is a critical update for modern management, particularly for life-threatening intravascular hemolysis (Neo-antigen model) caused by Tier 1 drugs like Ceftriaxone. If a drug has a long half-life, is highly protein-bound, or if the patient is already experiencing acute kidney injury (delaying renal clearance of the drug), simply stopping the medication isn't fast enough.
TPE (Plasmapheresis) is used to physically remove the circulating drug, the drug-antibody complexes, and the free hemoglobin from the patient's plasma, rapidly halting the hemolytic cascade.
Step 4: Targeted Immunosuppression
The role of immunosuppression in Drug-Induced Immune Hemolytic Anemia is highly dependent on the mechanism.
- Drug-Dependent Models (Hapten/Neo-antigen): Corticosteroids (e.g., Prednisone 1-2 mg/kg) are traditionally given, but they are generally ineffective at stopping the acute destruction. They are primarily used as a temporizing measure while waiting for the drug to clear.
- Drug-Independent Models (Autoimmune): For drugs that trigger a true autoantibody (e.g., Immune Checkpoint Inhibitors, Fludarabine), the immune system will continue attacking RBCs long after the drug is gone.
- First-Line: High-dose Corticosteroids.
- Second-Line: Rituximab (anti-CD20 monoclonal antibody) is frequently required for severe or refractory cases induced by modern oncology drugs.
Step 5: Renal Protection and Supportive Care
Massive intravascular hemolysis releases toxic levels of free hemoglobin, which can precipitate in the renal tubules and cause acute tubular necrosis (ATN).
- Vigorous Hydration: Intravenous fluids are pushed to maintain high urine output and "flush" the kidneys.
- Alkalinization of Urine: Sodium bicarbonate is sometimes added to IV fluids to alkalinize the urine, which helps prevent hemoglobin from precipitating in the tubules.
- Folic Acid: 1-5 mg daily is prescribed to support the bone marrow's massive compensatory reticulocytosis.
Frequently Asked Questions (FAQs)
Can levothyroxine cause Drug-Induced Immune Hemolytic Anemia?
Levothyroxine, a synthetic thyroid hormone replacement medication, is generally not considered a common cause of Drug-Induced Immune Hemolytic Anemia. Levothyroxine doesn't have a known mechanism for direct RBC interaction.
Why is the eluate test negative in most Drug-Induced Immune hemolytic anemia cases?
In drug-dependent Drug-Induced Immune Hemolytic Anemia, the antibody only recognizes the target when the drug is present. Because the eluate testing process washes the drug away, the resulting fluid (eluate) will test negative unless the laboratory specifically re-introduces the culprit drug into the test tube.
What is the most common antibiotic that causes Drug-Induced Immune Hemolytic Anemia?
Historically, high-dose penicillins and methyldopa were the primary causes. Today, cephalosporins (specifically ceftriaxone and cefotetan) and piperacillin are the most frequently implicated antibiotics.
Can stopping the medication cure Drug-Induced Immune Hemolytic Anemia?
Yes. Discontinuing the offending drug is the first-line and most crucial step in management. In most cases, hemolysis slows significantly within a few days to weeks after the medication is completely cleared from the body, though severe cases may require supportive transfusions or therapies like plasmapheresis.
Why is ceftriaxone more dangerous than other drugs?
Ceftriaxone typically triggers an IgM-mediated response that is highly efficient at fixing complement. This leads to rapid, massive cell lysis within the bloodstream (intravascular hemolysis), which can lead to kidney failure and death within hours.
Can ITP and Drug-Induced Immune Hemolytic Anemia occur together?
Yes. When a drug triggers an immune response against both platelets and red blood cells, it can mimic Evans Syndrome. In these cases, it is vital to distinguish if the ITP is Primary ITP or secondary to the drug.
Is a positive DAT always a sign of Drug-Induced Immune Hemolytic Anemia?
No. Some drugs (like Tazobactam) cause Non-immunological Protein Adsorption (NIPA), which makes the DAT positive by "sticking" proteins to the cell, even though no actual immune attack or hemolysis is occurring.
If a patient has Drug-Induced Immune Hemolytic Anemia once, can they ever take that drug again?
No. Re-exposure to the culprit drug can lead to a "recall" immune response that is much faster and more severe than the first episode. This should be marked as a permanent allergy in the medical record.
Glossary of Related Medical Terms
- Direct Antiglobulin Test (DAT): A blood test used to determine if red blood cells have been coated in vivo with immunoglobulin, complement, or both.
- Eluate: The solution obtained by detaching (eluting) antibodies from the surface of sensitized red blood cells so their specificity can be tested in vitro.
- Hapten: A small molecule (like a medication) that can elicit an immune response only when attached to a larger carrier protein, such as an RBC membrane protein.
- Intravascular Hemolysis: The destruction of red blood cells directly within the blood vessels, often leading to hemoglobinuria.
- Molecular Mimicry: A theoretical mechanism where a foreign substance (like a drug) structurally resembles a host tissue, causing the immune system to mistakenly attack the host's own cells.
- Polychromasia: A presentation on a peripheral blood smear where red blood cells appear larger and have a bluish tint, indicating an increased presence of immature RBCs (reticulocytes) compensating for hemolysis.
- Neo-antigen: A brand-new antigen formed when a drug loosely associates with a protein on the red blood cell membrane, creating a target that neither the drug nor the cell had alone.
- Drug-Dependent Antibody: An antibody that requires the drug to be physically present in the blood or on the cell surface to cause a reaction.
- Drug-Independent Antibody: An autoantibody triggered by a drug that continues to attack red blood cells even after the drug has been completely cleared from the system.
Disclaimer: This article is intended for informational purposes only and is specifically targeted towards medical students. It is not intended to be a substitute for informed professional medical advice, diagnosis, or treatment. While the information presented here is derived from credible medical sources and is believed to be accurate and up-to-date, it is not guaranteed to be complete or error-free. See additional information.
References
- Garratty G. Drug-induced immune hemolytic anemia. Hematology Am Soc Hematol Educ Program (2009) 2009 (1): 73–79.
- Maquet J, Lafaurie M, Michel M, Lapeyre-Mestre M, Moulis G. Drug-induced immune hemolytic anemia: detection of new signals and risk assessment in a nationwide cohort study. Blood Adv (2024) 8 (3): 817–826.
- https://medlineplus.gov/ency/article/000578.htm
- Chan Gomez J, Saleem T, Snyder S, Joseph M, Kanderi T. Drug-Induced Immune Hemolytic Anemia due to Amoxicillin-Clavulanate: A Case Report and Review. Cureus. 2020 Jun 17;12(6):e8666. doi: 10.7759/cureus.8666. PMID: 32699666; PMCID: PMC7370667.
- Sun, X. M., Liu, L. H., Wu, Q., & Wang, H. G. (2023). Cefoperazone/sulbactam-induced hemolytic anemia. Journal of postgraduate medicine, 69(1), 46–49. https://doi.org/10.4103/jpgm.JPGM_1335_20
- Maquet, J., Lafaurie, M., Michel, M., Lapeyre-Mestre, M., & Moulis, G. (2024). Drug-induced immune hemolytic anemia: detection of new signals and risk assessment in a nationwide cohort study. Blood advances, 8(3), 817–826. https://doi.org/10.1182/bloodadvances.2023009801
- Kubo, A., Murakami, S., & Iwata, T. (2024). Drug Interaction-induced Hemolytic Anemia: An Unresolved Diagnostic Process. Internal medicine (Tokyo, Japan), 63(5), 631–633. https://doi.org/10.2169/internalmedicine.2119-23
- Dei Zotti, F., Qiu, A., D'Agati, V. D., Jagnarine, S., Kyritsis, E., Miller, A., Tredicine, M., Fliginger, D., Stone, E. F., Panch, S., & Hudson, K. E. (2024). Mitigation of checkpoint inhibitor-induced autoimmune hemolytic anemia through modulation of purinergic signaling. Blood, 144(15), 1581–1594. https://doi.org/10.1182/blood.2024024230
- Jalgaonkar, S. V., & Parmar, U. I. (2023). Drug-induced hemolytic anemia due to cefoperazone-sulbactum: Challenges in reaching diagnosis. Journal of postgraduate medicine, 69(1), 9–10. https://doi.org/10.4103/jpgm.jpgm_248_22
- Onyechi, A., Ohemeng-Dapaah, J., Patel, R., Onyechi, E., Oyenuga, M., Sartaj, S., Mehta, M., Lacasse, A., & Anyadibe, I. E. (2023). Metaxalone-induced Immune Hemolytic Anemia. Journal of community hospital internal medicine perspectives, 13(5), 86–89. https://doi.org/10.55729/2000-9666.1236