Key Takeaways
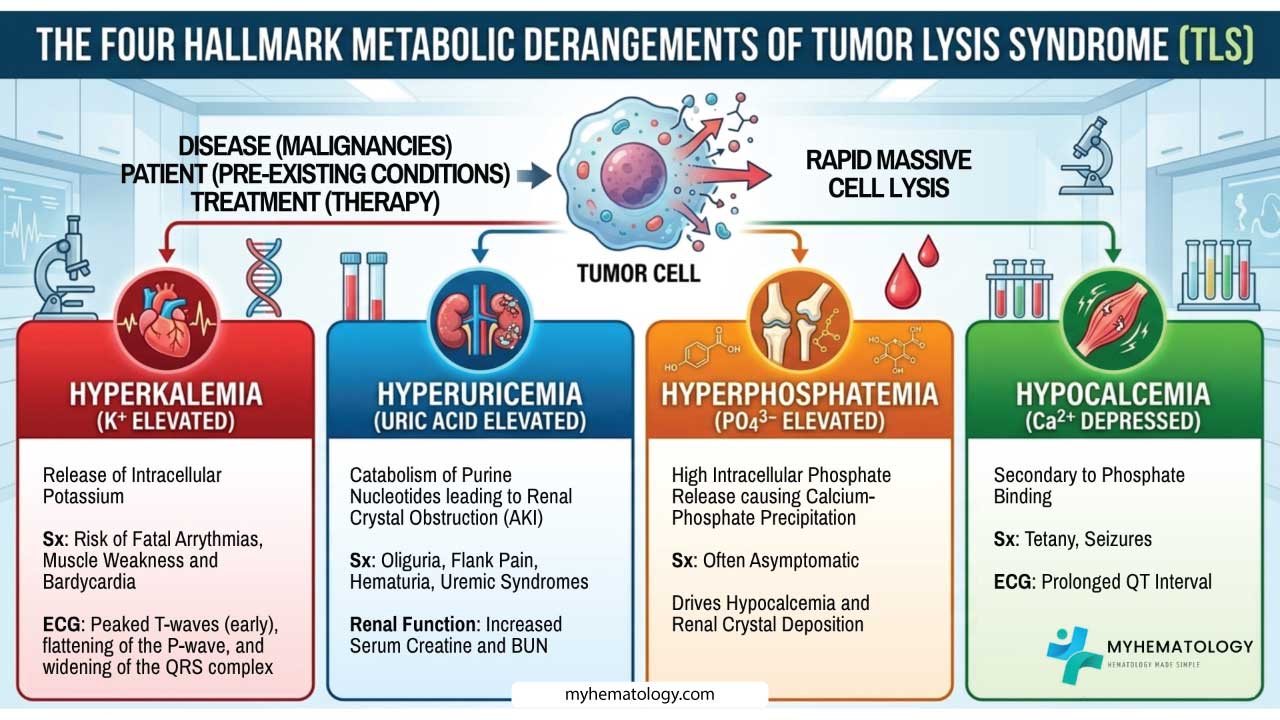
Tumor Lysis Syndrome or TLS is a life-threatening oncologic emergency caused by the rapid, massive lysis of malignant cells. This releases intracellular contents (potassium, phosphorus, and nucleic acids) into the systemic circulation, overwhelming homeostatic mechanisms.
- Pathophysiology ▾: Defined by four hallmark derangements (Metabolic Tetrad):
- Hyperuricemia: From purine catabolism (leads to renal crystal obstruction).
- Hyperkalemia: From direct cytoplasmic release (risk of fatal arrhythmias).
- Hyperphosphatemia: From organic phosphate release (causes calcium-phosphate precipitation).
- Hypocalcemia: Secondary to phosphate binding (causes tetany and seizures).
- Etiology and Risk Factors ▾:
- Disease: Highest risk in high-turnover hematologic malignancies like Burkitt lymphoma and Acute Lymphoblastic Leukemia (ALL).
- Patient: Pre-existing renal impairment, dehydration, and high baseline uric acid.
- Treatment: Typically triggered by cytotoxic chemotherapy, but can occur spontaneously or with targeted therapies (e.g., Venetoclax).
- Diagnostic Criteria (Cairo-Bishop) ▾:
- Laboratory Tumor Lysis Syndrome (LTLS): Two or more metabolic abnormalities occurring within 3 days before or 7 days after therapy.
- Clinical Tumor Lysis Syndrome (CTLS): LTLS plus significant end-organ damage (creatinine elevation, seizure, or cardiac arrhythmia).
- Clinical Manifestations ▾:
- Neuromuscular: Muscle cramps, tetany (Chvostek/Trousseau signs), and seizures.
- Cardiac: Palpitations, syncope, and ECG changes (peaked T-waves, prolonged QT).
- Renal: Oliguria, flank pain, and hematuria.
- Prevention and Risk Stratification ▾:
- Low Risk: Monitoring and oral/IV hydration.
- Intermediate Risk: IV hydration (2.5 - 3.0 L/m2/day) and Allopurinol.
- High Risk: Aggressive IV hydration and prophylactic Rasburicase.
- Management of Established Tumor Lysis Syndrome (TLS) ▾:
- Rasburicase: The drug of choice to rapidly clear existing uric acid (contraindicated in G6PD deficiency).
- Electrolyte Correction: Aggressive management of hyperkalemia; asymptomatic hypocalcemia should not be treated to avoid metastatic calcification.
- Dialysis: Indicated for refractory hyperkalemia, severe hyperphosphatemia, or volume overload.
- Differential Diagnosis ▾: Must be distinguished from prerenal azotemia, obstructive uropathy, pseudohyperkalemia (in high WBC counts), and hypercalcemia of malignancy.
- Prognosis ▾: While Laboratory Tumor Lysis Syndrome is highly manageable, Clinical Tumor Lysis Syndrome carries a high mortality rate (20 - 50%). Early recognition and maintaining renal function are the primary determinants of survival.
*Click ▾ for more information
Introduction
Tumor Lysis Syndrome (TLS) is one of the most critical oncologic emergencies encountered in clinical practice. At its core, Tumor Lysis Syndrome (TLS) is a constellation of metabolic disturbances caused by the rapid, massive lysis of malignant cells. When these cells rupture either spontaneously due to high tumor turnover or, more commonly, following cytotoxic therapy, they release their intracellular contents into the systemic circulation.
The kidneys and homeostatic mechanisms are overwhelmed by the sudden influx of:
- Potassium (Hyperkalemia)
- Phosphorus (Hyperphosphatemia)
- Nucleic Acids (Metabolized into Uric Acid, leading to Hyperuricemia)
This "metabolic dump" leads to a predictable tetrad of biochemical abnormalities that can progress to end-organ failure, specifically acute kidney injury (AKI), cardiac arrhythmias, and neuromuscular irritability.
The Gold Standard: Cairo-Bishop Criteria
The most widely accepted framework for defining and grading Tumor Lysis Syndrome (TLS) is the Cairo-Bishop Definition. It distinguishes between laboratory evidence and clinical impact.
Laboratory Tumor Lysis Syndrome (LTLS)
LTLS is diagnosed when two or more of the following metabolic abnormalities occur within 3 days before or 7 days after the initiation of therapy.
| Parameter | Threshold (Traditional) | Threshold (SI Units) | Change from Baseline |
| Uric Acid | ≥ 8.0 mg/dL | ≥ 476 µmol/L | 25% Increase |
| Potassium | ≥ 6.0 mEq/L | ≥ 6.0 mmol/L | 25% Increase |
| Phosphorus | ≥ 4.5 mg/dL | ≥ 1.45 mmol | 25% Increase |
| Calcium | ≤ 7.0 mg/dL | ≤ 1.75 mmol/L | 25% Decrease |
Clinical Tumor Lysis Syndrome (CTLS)
CTLS is defined as the presence of Laboratory Tumor Lysis Syndrome (TLS) plus at least one of the following clinical complications (not directly attributable to a therapeutic agent):
- Renal Insufficiency: Serum creatinine ≥ 1.5 times the upper limit of normal (ULN).
- Cardiac Arrhythmia: Or sudden cardiac death.
- Seizure: Neurological manifestations of metabolic derangement (typically hypocalcemia).
Spontaneous vs. Induced TLS
- Spontaneous TLS: Occurs prior to the start of any therapy. This is usually seen in malignancies with extremely high proliferation rates (e.g., Burkitt Lymphoma). It is characterized by high uric acid and potassium but, interestingly, often presents with low phosphorus levels because the rapidly dividing tumor cells "recycle" the released phosphate for new cell synthesis.
- Induced TLS: Triggered by treatment (chemotherapy, radiation, or targeted agents like Venetoclax). This is the more common presentation and typically manifests 12–72 hours after therapy begins.
Clinical Significance
Tumor Lysis Syndrome (TLS) is not merely a laboratory curiosity; it is a life-threatening event. Mortality rates for clinical Tumor Lysis Syndrome (TLS) can be as high as 20% to 50% if not recognized early. Prevention is the primary goal. Once a patient meets the criteria for Clinical Tumor Lysis Syndrome (TLS), they already have end-organ damage, making management significantly more complex.
The pathophysiology of Tumor Lysis Syndrome (TLS) is a masterclass in metabolic overload. While the release of potassium and phosphate is immediate, the development of acute kidney injury (AKI) is primarily driven by the catabolism of nucleic acids and the unique environment of the renal tubule.
Etiology and Risk Factors
The etiology of Tumor Lysis Syndrome (TLS) is multi-factorial, stemming from a "perfect storm" where the aggressive nature of a malignancy meets a highly effective treatment in a vulnerable patient. For clinical education, it is best to categorize these risks into disease-related, patient-related, and treatment-related factors.
Disease-Related Factors
The primary driver of Tumor Lysis Syndrome (TLS) is the biology of the tumor itself. High-risk diseases typically share three characteristics: a high proliferation rate, a large tumor burden, and extreme sensitivity to cytotoxic therapy.
- High-Risk Hematologic Malignancies:
- Burkitt Lymphoma: The "classic" example due to its near-100% proliferation rate.
- Lymphoblastic Lymphoma and Acute Lymphoblastic Leukemia (ALL): Especially with a high White Blood Cell (WBC) count (≥ 100,000/µL).
- Acute Myeloid Leukemia (AML): Specifically with high WBC counts or high LDH levels.
- Intermediate-Risk Malignancies:
- Diffuse Large B-Cell Lymphoma (DLBCL).
- Chronic Lymphocytic Leukemia (CLL) following treatment with targeted agents like Venetoclax.
- Solid Tumors (Rare): While rare, Tumor Lysis Syndrome (TLS) can occur in bulky, treatment-sensitive solid tumors such as Small Cell Lung Cancer or Germ Cell Tumors.
Patient-Related Factors
Even a "low-risk" tumor can cause Tumor Lysis Syndrome (TLS) if the patient’s homeostatic mechanisms are compromised.
- Pre-existing Renal Impairment: This is the most significant patient-specific risk. If the kidneys cannot effectively clear the sudden "metabolic dump" of uric acid and phosphate, Tumor Lysis Syndrome (TLS) develops rapidly.
- Baseline Hyperuricemia: Patients with high uric acid levels before starting treatment have a much lower threshold for developing crystal nephropathy.
- Dehydration and Oliguria: Low urine flow rates increase the concentration of solutes in the distal tubules, promoting the precipitation of uric acid and calcium-phosphate crystals.
- Splenomegaly or Bulky Lymphadenopathy: High tumor volume (≥ 10 cm mass) significantly increases the total "pool" of intracellular ions that can be released.
Treatment-Related Factors
The type and intensity of the therapy often act as the "trigger" for the syndrome.
- Cytotoxic Chemotherapy: The most common cause, typically occurring 12–72 hours after the first dose.
- Targeted Therapies: Modern agents such as Venetoclax (Bcl-2 inhibitor) in CLL or Rituximab in high-burden lymphomas have a high propensity for inducing Tumor Lysis Syndrome (TLS).
- Radiation Therapy: Particularly when treating large, sensitive masses (e.g., mediastinal lymphoma).
- Steroids: In highly sensitive lymphoid malignancies, even a "pre-phase" of corticosteroids (like Dexamethasone or Prednisone) can trigger spontaneous lysis.
Risk Stratification Table
In clinical practice, patients are categorized into risk groups to determine the level of monitoring and prophylaxis required.
| Risk Category | Examples | Recommended Prophylaxis |
| High Risk (> 5% chance) | Burkitt Lymphoma, ALL (WBC ≥ 100k), Bulky DLBCL | Aggressive hydration + Rasburicase |
| Intermediate Risk (1 - 5%) | Most AML, DLBCL (non-bulky), CLL treated with Venetoclax | Hydration + Allopurinol |
| Low Risk (< 1%) | Most solid tumors, Indolent lymphomas | Monitoring + Hydration as needed |
Pathophysiology of Tumor Lysis Syndrome
In the pathophysiology of Tumor Lysis Syndrome (TLS), we have to look beyond just the "plumbing" issue of blocked renal tubules. It is a systemic metabolic crisis where the sudden release of intracellular "pollution" overwhelms the body's homeostatic mechanisms.
The process can be broken down into three distinct phases: the cellular release, the metabolic interplay, and the end-organ damage.
The Cellular Release
Malignant cells, particularly in high-grade hematologic malignancies, are metabolic powerhouses with high turnover rates. When these cells rupture, they release their contents into the extracellular fluid (ECF) at a rate that exceeds the kidneys' excretory capacity.
- Potassium (K+): Intracellular concentration is roughly 140 - 150 mEq/L compared to the ECF's 3.5 - 5.0 mEq/L. Massive lysis leads to rapid, life-threatening hyperkalemia.
- Phosphorus (PO43-): Malignant lymphoblasts can contain up to four times more organic phosphate than a normal mature lymphocyte. This massive load is the primary driver of secondary complications.
- Nucleic Acids: Rapidly dividing cells have a high DNA/RNA content. As discussed, these are catabolized into uric acid. In humans, uric acid is the final product of purine metabolism because we lack the enzyme urate oxidase (uricase), which would normally convert uric acid into the highly soluble metabolite, allantoin.
The Metabolic Interplay
The most dangerous aspect of Tumor Lysis Syndrome (TLS) pathophysiology isn't just the individual high levels, but how they interact.
As serum phosphate levels rise, the "solubility product" ([Ca2+] x [PO43-]) is exceeded. When this product (measured in mg2/dL2) exceeds approximately 60 to 70, calcium and phosphate begin to precipitate into solid crystals.
This leads to secondary hypocalcemia, as free ionized calcium is "pulled" out of the blood to form these precipitates. Unlike uric acid, which is more of a late-stage or induced problem, hyperphosphatemia is often the most aggressive component in treated TLS.
While uric acid is often the primary focus, a secondary mechanism of acute kidney injury in Tumor Lysis Syndrome (TLS) involves calcium phosphate precipitation (nephrocalcinosis). Unlike uric acid, calcium phosphate is less soluble in alkaline environments.
These complexes can deposit in the renal interstitium and tubules, contributing to "stone-like" obstruction and parenchymal damage. This is a key reason why aggressive urine alkalinization, once a standard practice, is now approached with caution, as it may worsen calcium phosphate deposition.
Uric Acid Crystal Nephropathy
The transition from hyperuricemia to renal failure occurs within the nephron. This process is governed by two physical factors: concentration and pH.
Uric acid has very low solubility in aqueous solutions. In the systemic circulation (pH ~7.4), it exists primarily as the more soluble urate ion. However, as the filtrate moves through the nephron, the environment changes.
The pH of the tubular fluid drops significantly in the distal tubule and collecting ducts (often reaching a pH of 5.0 to 6.0). In this acidic environment, urate is converted back into its protonated form, uric acid, which is significantly less soluble.
When the concentration of uric acid exceeds its solubility limit in the distal tubule, it precipitates into needle-like crystals. These crystals cause:
- Intratubular Obstruction: As previously mentioned, uric acid crystals (in acidic distal tubules) and calcium-phosphate crystals (in alkaline/neutral environments) physically block the flow of urine.
- Renal Vasoconstriction: High levels of uric acid can inhibit the release of nitric oxide and activate the renin-angiotensin system, leading to profound renal vasoconstriction and reduced medullary blood flow.
- Inflammatory Cascade: The presence of these crystals isn't passive. They trigger the NLRP3 inflammasome, leading to the release of pro-inflammatory cytokines like IL-1β. This causes local tissue inflammation, edema, and further tubular damage.
Systemic Inflammatory Response (SIRS)
In severe cases, the massive release of intracellular proteins and cytokines (like TNF-α) can mimic a systemic inflammatory response syndrome or even lead to Disseminated Intravascular Coagulation (DIC). This explains why some patients appear "septic" even in the absence of a documented infection.
Summary of the Metabolic Tetrad
| Abnormality | Primary Mechanism | Critical Consequence |
| Hyperkalemia | Direct release from cytoplasm | Cardiac arrhythmias / Ventricular fibrillation |
| Hyperuricemia | Purine catabolism (DNA/RNA) | Intratubular crystal obstruction (AKI) |
| Hyperphosphatemia | Release of organic phosphates | Calcium-phosphate precipitation in tissues |
| Hypocalcemia | Complexing with phosphate | Tetany, seizures, prolonged QT interval |
Clinical Manifestations of TLS
The clinical manifestations of Tumor Lysis Syndrome (TLS) are the outward signs of the internal "metabolic storm." They are direct physiological consequences of the metabolic tetrad: Hyperkalemia, Hyperuricemia, Hyperphosphatemia, and Hypocalcemia.
Clinical TLS (CTLS) is diagnosed when laboratory abnormalities are accompanied by significant end-organ damage.
Neuromuscular Manifestations
These are primarily driven by hypocalcemia (secondary to hyperphosphatemia) and, to a lesser extent, hypermagnesemia if renal failure is present.
- Irritability and Tetany: Involuntary muscle contractions or "twitches."
- Classic Physical Signs:
- Chvostek’s Sign: Tapping the facial nerve (in front of the ear) triggers twitching of the facial muscles.
- Trousseau’s Sign: Inflating a blood pressure cuff above systolic pressure for 3 minutes triggers carpal spasms.
- Seizures: Massive metabolic shifts can lower the seizure threshold, leading to generalized tonic-clonic activity.
- Muscle Weakness/Paralysis: Typically a result of severe hyperkalemia, which interferes with the resting membrane potential of muscle cells.
Cardiovascular Manifestations
The heart is exceptionally sensitive to shifts in potassium and calcium. These manifestations are the most frequent cause of sudden death in Tumor Lysis Syndrome (TLS).
- Palpitations and Syncope: Often the first sign of an underlying arrhythmia.
- Arrhythmias:
- Hyperkalemia can lead to bradycardia, heart block, and eventually ventricular fibrillation (cardiac arrest).
- Hypocalcemia can cause heart failure or exacerbate arrhythmias.
- ECG Changes:
- Hyperkalemia (K+ ↑): Peaked T-waves (early), flattening of the P-wave, and widening of the QRS complex (late "sine-wave" pattern).
- Hypocalcemia (Ca2+ ↓): Prolongation of the QT interval, which can predispose the patient to Torsades de Pointes.
Renal Manifestations
Renal symptoms result from the mechanical and inflammatory damage caused by uric acid and calcium-phosphate crystals.
- Oliguria/Anuria: A significant drop in urine output is often the first "red flag" of impending renal failure.
- Flank Pain: Caused by the distension of the renal capsule due to intra-tubular obstruction and inflammation.
- Hematuria: While less common than in nephrolithiasis, "sludging" of crystals can cause microscopic or gross hematuria.
- Fluid Overload: As the kidneys fail to excrete water, patients may present with peripheral edema, hypertension, or pulmonary rales.
Gastrointestinal and Systemic Symptoms
These are often non-specific but are critical for early recognition in a clinical setting.
- Nausea, Vomiting, and Anorexia: Often early indicators of rising metabolic waste products (uremia).
- Lethargy and Fatigue: A general sense of malaise often precedes more severe neurological or cardiac events.
- Cloudy Urine: Occasionally, patients or nurses may report "milky" or cloudy urine, which is a visible sign of massive crystalluria (especially uric acid).
Linking Ions to Symptoms
| Metabolic Derangement | Clinical "Screams" |
| Hyperkalemia | Peaked T-waves, muscle weakness, bradycardia, cardiac arrest. |
| Hypocalcemia | Tetany, Chvostek/Trousseau signs, seizures, prolonged QT interval. |
| Hyperphosphatemia | Often asymptomatic itself, but drives hypocalcemia and renal crystal deposition. |
| Hyperuricemia | Oliguria, flank pain, hematuria, uremic symptoms (nausea/lethargy). |
In Spontaneous Tumor Lysis Syndrome (TLS) (before treatment), the patient might present with flank pain and oliguria but without the high phosphate levels seen in treatment-induced Tumor Lysis Syndrome (TLS). If you see high uric acid and high phosphate together, the "bomb" has usually been triggered by therapy.
Laboratory Investigations and Monitoring of TLS
The Essential "TLS Panel"
A standard Tumor Lysis Syndrome (TLS) workup includes the metabolic tetrad plus markers of tumor burden and renal function.
- Uric Acid: The primary marker of purine catabolism. Levels ≥ 8.0 mg/dL (or a 25% increase from baseline) meet the Cairo-Bishop criteria.
- Potassium (K+): The most time-sensitive marker. Even a "high-normal" value that is rising rapidly should trigger immediate concern.
- Phosphorus (PO43-): Significant elevations (≥ 4.5 mg/dL) often occur 24 - 48 hours after treatment.
- Calcium (Ca2+): Always measure ionized calcium if possible, as total calcium can be misleading in patients with low albumin. Hypocalcemia (≤ 7.0 mg/dL) is typically secondary to hyperphosphatemia.
- Lactate Dehydrogenase (LDH): While not a direct part of the diagnostic criteria, LDH is a critical surrogate marker for tumor burden and proliferation rate. A skyrocketing LDH is often a "canary in the coal mine" for impending Tumor Lysis Syndrome (TLS).
Renal Function and Monitoring
- Serum Creatinine and BUN: Used to detect the onset of Acute Kidney Injury (AKI). A ≥ 0.3 mg/dL absolute increase or a 1.5x increase from baseline.
- Urinalysis and Microscopy:
- Crystalluria: Looking for uric acid crystals (pleomorphic, diamond, or rhomboid shapes).
- Urine pH: Historically used to monitor alkalinization therapy, though this is less common now.
- Urine Output Monitoring: Strict "Ins and Outs" (I/Os) are mandatory. A drop in urine output (< 0.5 mL/kg/hr) often precedes a rise in creatinine.
Frequency of Monitoring (The Monitoring Protocol)
The frequency of lab draws is determined by the patient's risk stratification. Monitoring typically continues for 3–7 days post-treatment.
| Risk Category | Lab Frequency (Uric acid, K+, PO4, Ca2+, Cr) |
| High Risk | Every 4 to 6 hours |
| Intermediate Risk | Every 8 to 12 hours |
| Low Risk | Daily (or as clinically indicated) |
*If a patient is receiving Rasburicase, blood samples for uric acid must be collected in pre-chilled tubes and transported on ice. Rasburicase remains active ex vivo at room temperature and will break down the uric acid in the test tube, leading to a falsely low (undetectable) result.
Ancillary Investigations
- Electrocardiogram (ECG): Mandatory for any patient with hyperkalemia or significant hypocalcemia.
- Look for: Peaked T-waves, PR prolongation, or QRS widening (K+); prolonged QT interval (Ca2+).
- Cardiac Telemetry: Recommended for high-risk patients or those with existing electrolyte derangements.
- Renal Ultrasound: Useful if the diagnosis of Tumor Lysis Syndrome (TLS) is unclear, to rule out obstructive uropathy from bulky tumor masses (lymphadenopathy) versus intratubular crystal precipitation.
Laboratory "Red Flags"
According to the Cairo-Bishop criteria, a 25% change from baseline in any of the four key electrolytes, even if the value remains within the "normal" laboratory range, is considered a positive laboratory marker for Tumor Lysis Syndrome (TLS).
Differential Diagnosis
In the clinical setting, Tumor Lysis Syndrome (TLS) is often a "diagnosis of exclusion" or a diagnosis made by recognizing a pattern of laboratory trends. The differential diagnosis can be divided into three primary categories: other causes of Acute Kidney Injury (AKI), laboratory artifacts, and contrasting metabolic states.
Other Causes of AKI in Oncology
Patients with high-grade malignancies are often medically fragile, and their renal failure may not always be due to crystal nephropathy.
- Prerenal Azotemia: Oncology patients are frequently dehydrated due to poor oral intake, nausea/vomiting, or fever. Prerenal AKI typically shows a high BUN-to-creatinine ratio (> 20:1) and low urinary sodium (FENa < 1%), without the explosive rise in PO43- and uric acid seen in TLS.
- Nephrotoxic Medications: Many chemotherapeutic agents (e.g., Cisplatin, Methotrexate) and supportive meds (e.g., NSAIDs, certain antibiotics, IV contrast) are directly nephrotoxic. These usually follow a specific administration timeline and lack the characteristic metabolic tetrad.
- Obstructive Uropathy (Postrenal): Bulky tumors, such as retroperitoneal lymphadenopathy in lymphoma or pelvic masses, can physically compress the ureters. Hydronephrosis will be visible on a renal ultrasound. While Tumor Lysis Syndrome (TLS) causes "internal" obstruction (tubular), this is "external" obstruction.
Pseudohyperkalemia
In patients with extremely high white blood cell counts (e.g., hyperleukocytic AML or CLL), laboratory results can be deceptive.
When blood is drawn into a test tube, the fragile, crowded leukemic cells can rupture mechanically during the draw or during centrifugation. This releases potassium into the serum inside the tube, showing a high value that does not reflect the patient's actual physiology.
If the patient has a sky-high K+ but a normal ECG and no other Tumor Lysis Syndrome (TLS) markers, suspect pseudohyperkalemia.
Draw a plasma potassium (heparinized tube) and transport it to the lab by hand (avoiding pneumatic tubes) to minimize mechanical lysis
Spontaneous TLS vs. Treatment-Induced TLS
While both fall under the umbrella of "Tumor Lysis Syndrome (TLS)," they present differently and require different levels of suspicion.
| Feature | Spontaneous TLS | Treatment-Induced TLS |
| Trigger | High tumor turnover alone | Cytotoxic therapy (chemo, radiation) |
| Uric Acid | Extremely High | High |
| Phosphate | Often Normal/Low | High |
| Clinical Context | Present at diagnosis | 12 - 72 hours after therapy starts |
Why the difference in Phosphate? In spontaneous TLS, the rapidly dividing tumor cells often "re-uptake" the released phosphorus to build new cells, whereas, in treatment-induced TLS, the cells are dying en masse and cannot recycle the ions.
Hypercalcemia of Malignancy vs. Tumor Lysis Syndrome (TLS)
This is a critical distinction for students, as the management is diametrically opposed.
- Hypercalcemia of Malignancy: Common in solid tumors (lung, breast) and Multiple Myeloma. It is driven by PTHrP (parathyroid hormone-related protein) or bone resorption.
- Tumor Lysis Syndrome (TLS): Characterized by hypocalcemia (low calcium).
If an oncology patient presents with high calcium, they likely have a different metabolic complication (Hypercalcemia of Malignancy). If they have low calcium and high phosphate, they are likely in Tumor Lysis Syndrome (TLS).
Sepsis and Septic Shock
Sepsis can cause AKI and metabolic acidosis, which can shift potassium out of cells, mimicking some features of Tumor Lysis Syndrome (TLS).
Sepsis typically presents with hypotension and high lactate, but lacks the specific "triad" of hyperuricemia and hyperphosphatemia found in Tumor Lysis Syndrome (TLS). However, remember that sepsis and Tumor Lysis Syndrome (TLS) often coexist in the same patient.
Summary Table for Differential Diagnosis
| Condition | K+ | PO43− | Uric Acid | Ca2+ |
| Tumor Lysis Syndrome | ↑ | ↑ | ↑ | ↓ |
| Spontaneous TLS | ↑ | ↔/↓ | ↑↑ | ↔/↓ |
| Pseudohyperkalemia | ↑ | ↔ | ↔ | ↔ |
| Hypercalcemia of Malignancy | ↔ | ↔ | ↔ | ↑↑ |
| Prerenal AKI | ↔/↑ | ↔ | ↔ | ↔ |
Risk Stratification and Prevention of TLS
The cornerstone of managing Tumor Lysis Syndrome (TLS) is prevention. Because the mortality rate for established clinical Tumor Lysis Syndrome (TLS) is high, the standard of care is to risk-stratify every patient before initiating therapy and apply a prophylactic regimen tailored to that risk.
Risk Stratification
The most widely used system is based on the Howard et al. (2011) consensus guidelines, which classify patients into High, Intermediate, or Low risk based on three variables: the type of malignancy, the extent of tumor burden, and the patient's renal function.
| Risk Group | Typical Malignancies | Key Clinical/Lab Markers |
| High Risk (> 5% probability) | Burkitt Lymphoma; Lymphoblastic Lymphoma; ALL (WBC ≥ 100 x 109/L); AML (WBC ≥ 100 x 109/L). | Bulky disease (≥ 10 cm); LDH > 2X Upper Limit of Normal (ULN); Pre-existing renal dysfunction. |
| Intermediate Risk (1 - 5% probability) | DLBCL; ALL (WBC 25 - 100 x 109/L); AML (WBC 25 - 100 x 109/L); CLL (treated with Venetoclax). | LDH > 2X ULN but non-bulky disease. |
| Low Risk (< 1% probability) | Most solid tumors; Indolent Lymphomas (Follicular, Marginal Zone); Multiple Myeloma; CML (Chronic phase). | Normal renal function; LDH <2X ULN; Non-bulky disease. |
Prevention Strategies
The goal of prophylaxis is to maximize the excretion of metabolic byproducts and minimize the formation of uric acid and calcium-phosphate crystals.
Aggressive Hydration (The "Gold Standard")
This is the most critical intervention for all risk levels.
- Mechanism: Increases intravascular volume, enhances glomerular filtration, and dilutes the concentration of uric acid and phosphate in the renal tubules.
- Protocol: Typically 2.5 - 3.0 L/m2/day (roughly 200 mL/hr in adults).
- Goal: Maintain a high urine output (at least 100 mL/hr or > 3 mL/kg/hr in children).
- Caveat: Use with caution in patients with pre-existing heart failure or oliguric renal failure; loop diuretics (e.g., Furosemide) may be needed to maintain output once the patient is euvolemic.
Hypouricemic Agents
There are two primary pharmacological pathways to manage uric acid:
1. Allopurinol (Prevention for Low/Intermediate Risk)
- Mechanism: A xanthine oxidase inhibitor. It prevents the conversion of hypoxanthine and xanthine into uric acid.
- Limitation: It does not reduce existing uric acid. It only prevents new uric acid from being formed. It also leads to an accumulation of xanthine, which can cause "xanthine nephropathy" (though this is rare).
- Dosing: Usually 300 mg/m2/day (max 800 mg), starting 24 - 48 hours before chemo.
2. Rasburicase (Prevention for High Risk / Treatment for Established TLS)
- Mechanism: A recombinant urate oxidase enzyme. It converts uric acid into allantoin, which is five to ten times more soluble than uric acid and easily excreted.
- Advantages: It works rapidly (within 4 hours) and clears existing uric acid.
- Contraindication: G6PD deficiency. Rasburicase produces hydrogen peroxide as a byproduct; in G6PD-deficient patients, this leads to severe hemolysis and methemoglobinemia.
The Debate on Urine Alkalinization
- Historical View: Sodium bicarbonate was used to raise urine pH to >7.0 to increase uric acid solubility.
- Current View: Generally not recommended. While it increases uric acid solubility, it decreases the solubility of calcium phosphate, promoting its precipitation in the kidneys. Given the efficacy of Rasburicase, alkalinization is now considered counterproductive in most cases.
Monitoring as Prevention
Prevention also involves a rigorous laboratory schedule to detect "Laboratory TLS" before it becomes "Clinical TLS."
- Low Risk: Clinical monitoring and daily labs.
- Intermediate Risk: Hydration + Allopurinol; labs every 8 - 12 hours.
- High Risk: Hydration + Rasburicase (often a single prophylactic dose); labs every 4 - 6 hours.
Prophylactic Recommendations
| Risk Group | Hydration Strategy | Pharmacotherapy | Monitoring |
| Low | Oral or IV (as needed) | None or Allopurinol | Daily |
| Intermediate | IV (2.5 - 3.0 L/m2) | Allopurinol (7 - 10 days) | 8 - 12h |
| High | IV (3.0 L/m2) | Rasburicase | 4 - 6h |
Treatment and Management of Established TLS
When Tumor Lysis Syndrome (TLS) moves from the "Laboratory" stage to "Clinical TLS," the management shifts from prophylaxis to an aggressive, multi-disciplinary emergency response. The primary goals are to protect the heart from arrhythmias, prevent permanent renal damage, and rapidly clear metabolic toxins.
General Management Principles
- Inpatient Setting: Patients with established Tumor Lysis Syndrome (TLS) should be managed in an Intensive Care Unit (ICU) or a High Dependency Unit (HDU) for continuous cardiac telemetry and frequent lab monitoring (every 4 to 6 hours).
- Strict Fluid Balance: Hourly monitoring of "ins and outs" (I/Os) is mandatory. If urine output drops below 100 mL/hr despite adequate hydration, immediate intervention is required.
Aggressive Intravenous Hydration
Hydration remains the most effective way to "flush" the kidneys.
- Volume: 3 L/m2/day is the standard for established Tumor Lysis Syndrome (TLS).
- Fluid Choice: Isotonic saline (0.9% NaCl) is preferred. Avoid adding potassium to the IV fluids.
- Diuretics: If the patient is appropriately hydrated but remains oliguric, loop diuretics (e.g., Furosemide) may be used to maintain urine output. However, diuretics are contraindicated if the patient is dehydrated or has an obstructive uropathy.
Correcting the Metabolic Tetrad
A. Hyperuricemia (The Immediate Priority)
In established Tumor Lysis Syndrome (TLS), Allopurinol is insufficient.
- Rasburicase: This is the drug of choice. It rapidly breaks down existing uric acid into allantoin.
Monitor for hemolysis in patients of Mediterranean or African descent (screen for G6PD deficiency if possible).
B. Hyperkalemia
Management depends on the severity and ECG changes.
- Membrane Stabilization: IV Calcium Gluconate (only if ECG changes are present, like peaked T-waves).
- Intracellular Shift: IV Insulin with Glucose (D50) and/or inhaled beta-agonists (Albuterol).
- Elimination: Sodium zirconium cyclosilicate or other potassium binders.
C. Hyperphosphatemia
- Phosphate Binders: Oral binders (e.g., Sevelamer or Aluminum Hydroxide) can be used, but their utility is limited if the patient is NPO or vomiting.
Strictly eliminate phosphorus from the diet and IV fluids.
D. Hypocalcemia (The "Hands-Off" Rule)
- Treatment: Do not treat asymptomatic hypocalcemia. Administering calcium in the presence of high phosphate promotes calcium-phosphate precipitation in the kidneys and other organs (metastatic calcification).
- Exception: Treat only if the patient is symptomatic (tetany, seizures, or prolonged QT interval) using the lowest effective dose of IV calcium.
Renal Replacement Therapy (RRT)
If medical management fails to stabilize the patient, urgent hemodialysis or Continuous Renal Replacement Therapy (CRRT) is indicated.
Indications for Dialysis in Tumor Lysis Syndrome (TLS):
- Refractory Hyperkalemia: K+ > 6 mEq/L despite medical therapy.
- Severe Hyperphosphatemia: PO43- > 10 mg/dL or rapidly rising.
- Volume Overload: Pulmonary edema or inability to tolerate aggressive hydration.
- Symptomatic Hypocalcemia: Specifically when induced by high phosphate.
- Profound Oliguria/Anuria: Uremic complications.
Management Summary Table
| Metabolic Abnormality | First-Line Intervention | Key Consideration |
| Uric Acid ↑ | Rasburicase (0.15 - 0.2 mg/kg) | Use chilled tubes for lab draws. |
| Potassium ↑ | Insulin/Glucose + Ca-Gluconate | Continuous cardiac monitoring. |
| Phosphate ↑ | Hydration + Phosphate Binders | Avoid calcium containing binders. |
| Calcium ↓ | Observation | Only treat if symptomatic. |
| Renal Failure | Aggressive Hydration | Low threshold for dialysis/CRRT. |
Prognosis and Follow-Up
Once the metabolic storm passes (usually within 5–7 days), focus shifts back to treating the underlying malignancy. However, any episode of Clinical Tumor Lysis Syndrome (TLS) increases the risk of chronic kidney disease later in life, so long-term renal monitoring is recommended.
Prognosis and Complications of TLS
The prognosis of Tumor Lysis Syndrome (TLS) has improved significantly over the last two decades due to better risk stratification and the introduction of recombinant urate oxidase (Rasburicase).
Prognosis: Factors Influencing Outcomes
The prognosis for a patient with Tumor Lysis Syndrome (TLS) is generally dictated by the speed of recognition and the success of early intervention.
- Mortality Rates:
- Laboratory TLS: Low mortality, as these patients typically respond to hydration and hypouricemic agents before end-organ damage occurs.
- Clinical TLS: Historically, mortality rates have been reported between 20% and 50%. Even with modern intensive care, the presence of multi-organ failure (specifically AKI and cardiac arrhythmias) carries a guarded prognosis.
- The "Double-Edged Sword" of Chemotherapy: Tumor Lysis Syndrome (TLS) is a sign that the cancer is highly sensitive to treatment (a "good" oncologic sign), but the syndrome itself can be fatal. The prognosis is best when the metabolic crisis is resolved quickly, allowing the patient to continue their full chemotherapy cycle.
- Renal Recovery: Most patients who develop AKI from Tumor Lysis Syndrome (TLS) will recover renal function if treated early. However, approximately 5 - 10% of those who require dialysis may suffer permanent renal damage or transition to chronic kidney disease (CKD).
Acute and Life-Threatening Complications
These occur during the "storm" phase (typically 12 - 72 hours post-treatment) and require immediate ICU intervention.
- Acute Kidney Injury (AKI): The most frequent complication. It is driven by uric acid crystal nephropathy and calcium-phosphate precipitation. It complicates fluid management and electrolyte clearance, creating a dangerous feedback loop.
- Lethal Arrhythmias: Severe hyperkalemia (K+) and hypocalcemia (Ca2+) can lead to ventricular tachycardia, ventricular fibrillation, or asystole. This is the most common cause of sudden death in Tumor Lysis Syndrome (TLS).
- Neurological Crisis: Hypocalcemia-induced tetany can progress to status epilepticus (continuous seizures), which can cause permanent hypoxic brain injury.
- Disseminated Intravascular Coagulation (DIC): In extreme cases, the massive release of cytokines and procoagulant factors from lysed cells can trigger a systemic coagulopathy.
Long-Term and Indirect Complications
These complications affect the patient’s overall journey through cancer treatment.
- Metastatic Calcification: When the calcium-phosphate product remains high, crystals can deposit in soft tissues, including the heart (conduction system interference) and the vasculature.
- Treatment Interruption: This is a "silent" complication. If a patient develops Tumor Lysis Syndrome (TLS), subsequent doses of chemotherapy must often be delayed or reduced in intensity. This "under-treatment" can lead to the underlying cancer becoming resistant or relapsing sooner.
- Chronic Kidney Disease (CKD): The "insult" of acute tubular necrosis during Tumor Lysis Syndrome (TLS) can leave the patient with a reduced nephron mass, making them more vulnerable to future nephrotoxic chemotherapies (like Cisplatin).
Complications at a Glance
| Organ System | Acute Complication | Long-term Consequence |
| Renal | Acute Kidney Injury (AKI) | Chronic Kidney Disease (CKD) |
| Cardiac | Ventricular Arrhythmias / Arrest | Metastatic Calcification |
| Neurological | Seizures / Tetany | Hypoxic brain injury (rare) |
| Oncologic | Metabolic Crisis | Treatment Delay / Cancer Relapse |
The most dangerous complication of Tumor Lysis Syndrome (TLS) is not just the electrolyte shift itself, but the inability to deliver the life-saving cancer treatment because the patient's body cannot handle the "debris" of the dying cells.
Frequently Asked Questions (FAQ)
Why is phosphate often normal or low in spontaneous TLS?
In spontaneous Tumor Lysis Syndrome (TLS) (before treatment), the malignant cells are dividing so rapidly that they "re-uptake" or recycle the released phosphorus to synthesize new cellular membranes and DNA. In treatment-induced Tumor Lysis Syndrome (TLS), the cells are dying en masse and cannot recycle the ions, leading to the characteristic high phosphate levels.
Can I use the "25% Rule" for all patients?
The Cairo-Bishop criteria allow for a diagnosis of Laboratory Tumor Lysis Syndrome (TLS) if there is a 25% change from baseline. However, many clinicians are cautious with this rule because a 25% increase that still falls within the "normal" range may not always be clinically significant. It is most useful as an early warning sign to escalate monitoring.
Why is Rasburicase contraindicated in G6PD deficiency?
Rasburicase breaks down uric acid into allantoin, but this chemical reaction produces hydrogen peroxide as a byproduct. Patients with G6PD deficiency cannot neutralize this oxidative stress, leading to severe hemolysis and methemoglobinemia.
Why is aggressive hydration preferred over urine alkalinization?
While alkalinization (using sodium bicarbonate) makes uric acid more soluble, it significantly decreases the solubility of calcium phosphate. This increases the risk of calcium-phosphate crystals depositing in the renal tubules (nephrocalcinosis), which can be harder to manage than uric acid crystals.
When should I suspect "Pseudohyperkalemia"?
Suspect this if the lab reports a critically high potassium level in a patient with a very high White Blood Cell (WBC) count but a normal ECG. The mechanical stress of the blood draw can cause fragile leukemic cells to burst inside the tube, falsely elevating the result.
Glossary of Related Medical Terms
- Allantoin: A highly water-soluble metabolic byproduct of uric acid breakdown. In humans, this is only produced when the enzyme urate oxidase (Rasburicase) is administered.
- Anuria: The total or near-total absence of urine production (typically defined as less than 50 mL per day), representing a severe stage of acute kidney injury.
- Azotemia: An elevation of blood urea nitrogen (BUN) and serum creatinine levels, indicating a decrease in the glomerular filtration rate (GFR).
- Cairo-Bishop Criteria: The universally recognized classification system used to diagnose and grade the severity of both Laboratory and Clinical Tumor Lysis Syndrome (TLS).
- Chvostek’s Sign: A clinical sign of hypocalcemia characterized by an involuntary twitching of the facial muscles when the facial nerve is tapped.
- Crystalluria: The presence of crystals in the urine. In Tumor Lysis Syndrome (TLS), this typically refers to uric acid or calcium-phosphate crystals which can cause mechanical obstruction of the nephron.
- G6PD Deficiency: A genetic condition that predisposes red blood cells to break down (hemolysis) under oxidative stress. It is a strict contraindication for the use of Rasburicase.
- Hyperleukocytosis: An extremely high white blood cell count (usually >100,000/µL), which serves as a major risk factor for the development of Tumor Lysis Syndrome (TLS) in leukemia patients.
- Hyperuricemia: An abnormally high level of uric acid in the blood (defined in Tumor Lysis Syndrome (TLS) as ≥ 8.0 mg/dL), resulting from the rapid catabolism of purine nucleic acids.
- Hypocalcemia: Low levels of calcium in the blood. In Tumor Lysis Syndrome (TLS), this is typically "secondary hypocalcemia" caused by calcium binding to excess phosphate released from lysed cells.
- Ionized Calcium: The "free" or physiologically active form of calcium in the blood. This is the preferred measurement in Tumor Lysis Syndrome (TLS) because it accurately reflects the risk of neuromuscular and cardiac complications.
- Lactate Dehydrogenase (LDH): An intracellular enzyme released during cell damage. In hematology, it is used as a surrogate marker for tumor burden and the rate of cell turnover.
- Metastatic Calcification: The deposition of calcium-phosphate crystals in soft tissues, such as the kidneys or heart, occurring when the "solubility product" of calcium and phosphate is exceeded.
- Nephrocalcinosis: A specific form of metastatic calcification where calcium salts are deposited within the renal parenchyma and tubules, leading to permanent kidney damage.
- Oliguria: A reduction in urine output (typically < 0.5 mL/kg/hr in adults), which is often the earliest clinical warning sign of impending renal failure in Tumor Lysis Syndrome (TLS).
- Rasburicase: A recombinant version of the enzyme urate oxidase that rapidly converts uric acid into the much more soluble allantoin.
- Trousseau’s Sign: A sign of latent tetany (hypocalcemia) where a carpal spasm is induced by inflating a blood pressure cuff above the systolic pressure for 3 minutes.
- Xanthine Oxidase: The key enzyme in the purine catabolic pathway that converts hypoxanthine and xanthine into uric acid. It is the target inhibited by Allopurinol.
Disclaimer: This article is intended for informational purposes only and is specifically targeted towards medical students. It is not intended to be a substitute for informed professional medical advice, diagnosis, or treatment. While the information presented here is derived from credible medical sources and is believed to be accurate and up-to-date, it is not guaranteed to be complete or error-free. See additional information.
References
- Adeyinka A, Kaur A, Bashir K. Tumor Lysis Syndrome. [Updated 2024 Oct 5]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2026 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK518985/
- Lupușoru, G., Ailincăi, I., Frățilă, G., Ungureanu, O., Andronesi, A., Lupușoru, M., Banu, M., Văcăroiu, I., Dina, C., & Sinescu, I. (2022). Tumor Lysis Syndrome: An Endless Challenge in Onco-Nephrology. Biomedicines, 10(5), 1012. https://doi.org/10.3390/biomedicines10051012
- Arnaud, M., Loiselle, M., Vaganay, C., Pons, S., Letavernier, E., Demonchy, J., Fodil, S., Nouacer, M., Placier, S., Frère, P., Arrii, E., Lion, J., Mooney, N., Itzykson, R., Djediat, C., Puissant, A., & Zafrani, L. (2022). Tumor Lysis Syndrome and AKI: Beyond Crystal Mechanisms. Journal of the American Society of Nephrology : JASN, 33(6), 1154–1171. https://doi.org/10.1681/ASN.2021070997
- Joseph, A., & Zafrani, L. (2023). How I Treat Tumor Lysis Syndrome. Clinical journal of the American Society of Nephrology : CJASN, 18(12), 1634–1636. https://doi.org/10.2215/CJN.0000000000000331
- Mirrakhimov, A. E., Voore, P., Khan, M., & Ali, A. M. (2015). Tumor lysis syndrome: A clinical review. World journal of critical care medicine, 4(2), 130–138. https://doi.org/10.5492/wjccm.v4.i2.130
- Belay, Y., Yirdaw, K., & Enawgaw, B. (2017). Tumor Lysis Syndrome in Patients with Hematological Malignancies. Journal of oncology, 2017, 9684909. https://doi.org/10.1155/2017/9684909
- Puri, I., Sharma, D., Gunturu, K. S., & Ahmed, A. A. (2020). Diagnosis and management of tumor lysis syndrome. Journal of community hospital internal medicine perspectives, 10(3), 269–272. https://doi.org/10.1080/20009666.2020.1761185
- Cheson, B. D., Heitner Enschede, S., Cerri, E., Desai, M., Potluri, J., Lamanna, N., & Tam, C. (2017). Tumor Lysis Syndrome in Chronic Lymphocytic Leukemia with Novel Targeted Agents. The oncologist, 22(11), 1283–1291. https://doi.org/10.1634/theoncologist.2017-0055