Procedure At a Glance
- Prepare Patient Sample: Centrifuge patient serum to obtain plasma/serum.
- Prepare Red Blood Cells (RBCs): Make a 2-5% suspension of donor or reagent RBCs in normal saline.
- Combine Serum & RBCs: Add patient serum to the prepared RBC suspension.
- Incubation: Incubate the mixture at 37°C to allow any antibodies present in the serum to bind to the RBCs.
- Wash RBCs: Wash the RBCs thoroughly with normal saline to remove unbound antibodies. This is a critical step.
- Add Antihuman Globulin (AHG): Add the AHG (Coombs reagent) to the washed RBCs.
- Centrifuge & Observe: Centrifuge the mixture and gently resuspend the cell button. Observe for agglutination (clumping of RBCs), which indicates a positive result.
- Quality Control: Add Coombs Control Cells (CCC) to negative tubes to ensure the AHG was active and the washing was sufficient.
Introduction
The indirect antiglobulin test (IAT), also known as the indirect Coombs test, plays a crucial role in ensuring safe and compatible blood transfusions. This vital laboratory procedure delves into the world of antibodies, protecting against potentially disastrous immune reactions between your patient's blood and donor red blood cells (RBCs).
Key points of indirect antiglobulin test (IAT)
- The indirect Coombs test (IAT) detects antibodies against RBCs circulating in the serum, not directly bound to the RBCs themselves.
- It identifies a wider range of antibodies than the direct antiglobulin (Coombs) test (DAT), including weak antibodies that may not cause visible agglutination.
- The indirect Coombs test (IAT) is essential for both pre-transfusion compatibility testing and prenatal antibody screening in pregnant women.
- By identifying incompatible antibodies, the indirect Coombs test (IAT) helps prevent potentially life-threatening hemolytic transfusion reactions.
- Different types of antihuman globulin (AHG) reagents can be used in the indirect Coombs test (IAT), offering varying degrees of sensitivity and specificity for different antibody types.
- Interpreting indirect Coombs test (IAT) results is crucial for making informed clinical decisions in blood transfusion and managing autoimmune hemolytic anemia.
Central Role of Indirect Coombs Test in Immunohematology
The indirect Coombs test (IAT) is a cornerstone of blood bank practices, playing a crucial role in various immunohematological tests, including
- Antibody screening: Identifying the presence of any antibodies against red blood cells (RBCs) in a patient's serum, potentially causing transfusion reactions.
- Antibody identification: Characterizing the specific type of antibody present, guiding compatible blood selection for transfusions.
- Antigen typing: Determining the blood group antigens present on RBCs, ensuring accurate crossmatching and transfusion compatibility.
- Crossmatching: Checking for compatibility between a recipient's serum and donor RBCs, minimizing the risk of hemolytic reactions.
Principle of Indirect Coombs Test (IAT)
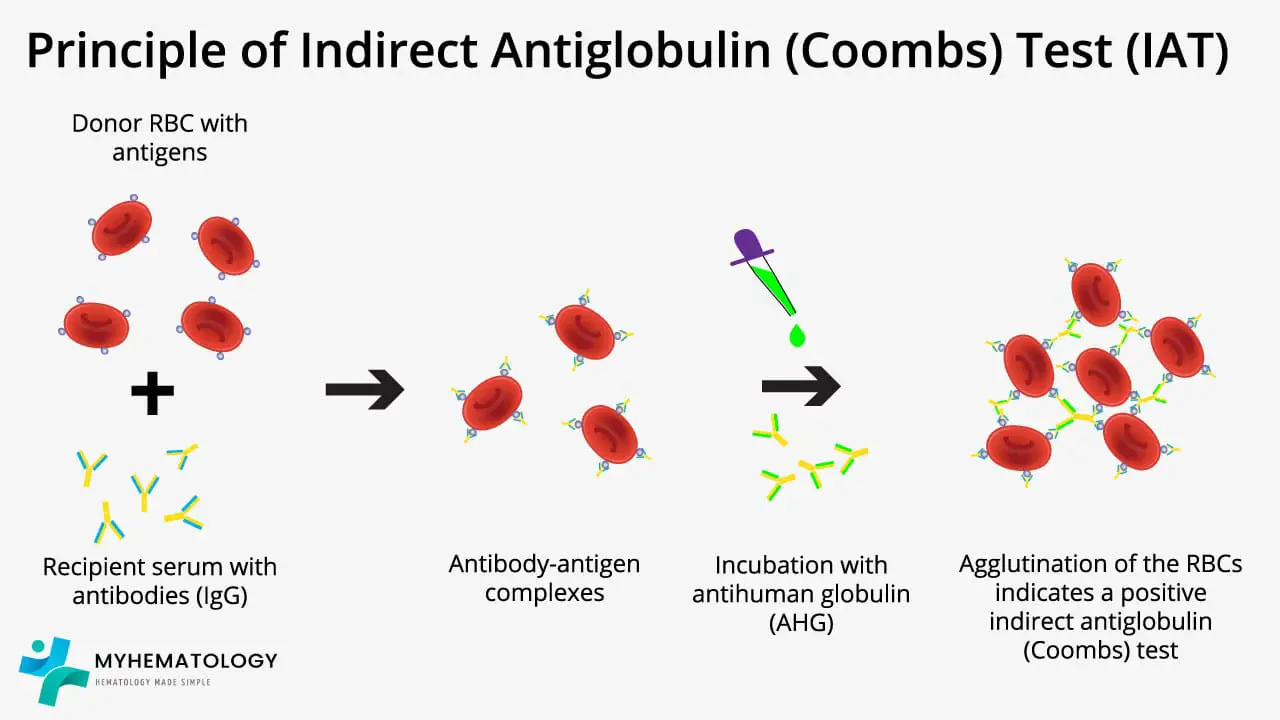
Unlike its direct counterpart - direct antiglobulin test (DAT), which scrutinizes red blood cells for attached antibodies, the indirect Coombs test (IAT) investigates the serum surrounding them for the presence of free globulin like IgG, IgM or C3d.
The indirect Coombs test (IAT) principle revolves around using RBCs, usually from a donor or a universal donor pool, and mix with the patient's serum. If any antibodies against RBCs are lurking within this serum, they latch onto the RBCs, forming invisible bonds.
When antihuman globulin (AHG) is introduced, it binds to these antibody-coated RBCs, acting like a bridge that forms visible clumps, or agglutination. This agglutination reveals the presence of anti-RBC antibodies in the patient's serum.
The power of the indirect Coombs test (IAT) lies in its versatility. It detects a wider range of antibodies than the direct Coombs test (DAT), including weak ones that might not cause visible clumping on the actual red blood cells. This makes it crucial for pre-transfusion compatibility testing, ensuring donated blood doesn't trigger an immune reaction in the recipient.
Additionally, the indirect Coombs test (IAT) plays a key role in diagnosing various immune-mediated hemolytic anemias, where antibodies attack the patient's own RBCs, leading to their premature destruction.
Materials
- Patient's serum sample
- Normal saline (0.9% NaCl) / Low ionic strength solution (LISS)
- Bovine albumin solution (30%)
- RBC suspension (3-5%):
- For compatibility testing: Use reagent RBCs of various types (A, B, O, Rh)
- For autoimmune hemolytic anemia (AIHA) investigation: Use patient's own RBCs
- Antihuman globulin (AHG) reagent: polyspecific or monospecific depending on test purpose
- Test tubes
- Centrifuge
- Coombs control red cells - IgG coated red cell (CCC)
Protocol
- Centrifuge the patient's serum sample and discard the clot.
- If using patient's own RBCs for AIHA investigation, prepare a red cell suspension by washing 5 mL patient RBCs with normal saline 3 - 4 times, discard the supernatant and resuspend RBCs to a 3-5% suspension in saline.
- Label three test tubes for each antigen type being tested:
- Test tube 1: Serum + RBCs
- Test tube 2: Serum + RBCs + Bovine Albumin (30%)
- Test tube 3: Negative Control (RBCs + LISS or saline)
- Add two drops of patient's serum and one drop of the respective RBC suspension to each tube.
- Add one drop of bovine albumin solution to the second tube only.
- Incubate all tubes at 37°C for 30 minutes or as per manufacturer's instructions.
- Wash the red cells with normal saline 3 times, discard the supernatant completely after the final wash other than the red cell button.
- Add 2 drops of polyspecific AHG onto the red cells.
- Tilt the tube and gently swirl it to mix the AHG with the red cells, ensuring they are completely resuspended.
- Place the tube in the centrifuge and spin for 15-30 seconds at 900-1000 g or as per manufacturer’s instructions.
- Gently resuspend the cell buttons and observe for agglutination macroscopically..
- Grade agglutination strength (0 to 4+) if present.
- In the absence of agglutination after initial centrifugation, add Coombs control cells (CCC) and centrifuge as per manufacturer instructions. Observe and record any agglutination with CCC, confirming test validity.
Interpretation
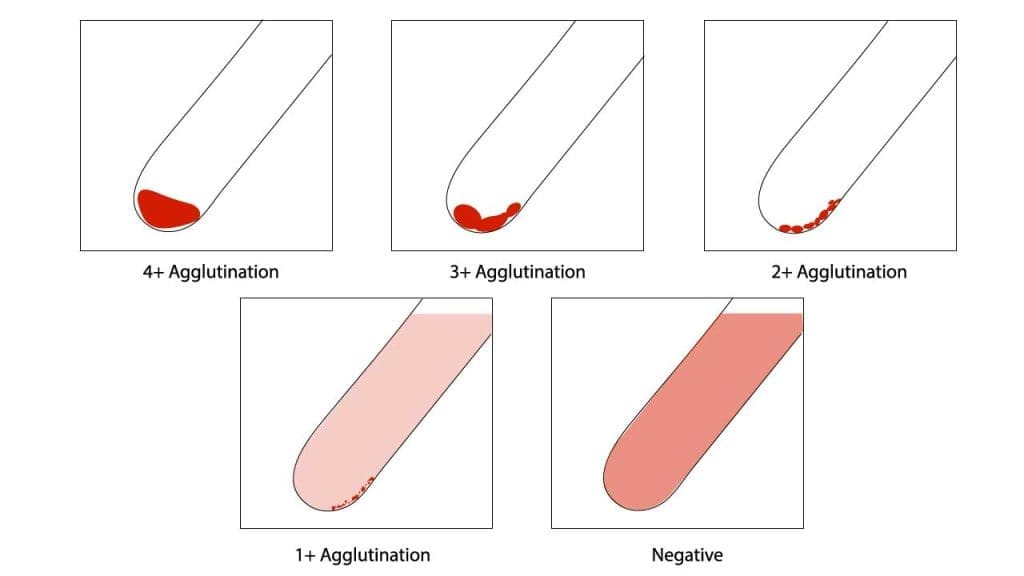
| Result | Interpretation |
| Positive agglutination or hemolysis in Test 1 only | Antibodies against the corresponding RBC type present |
| Positive agglutination or hemolysis in Test 1 and Test 2 | Non-specific agglutination due to cold agglutinins or protein interactions |
| Negative agglutination in all tubes | No detectable antibodies against the tested RBCs |
| Negative agglutination after AHG phase and after addition of CCC | The test is invalid and must be repeated |
Additional Notes
- Bovine albumin control: Helps differentiate specific antibody-mediated agglutination from non-specific agglutination due to cold agglutinins or protein interactions.
- Positive agglutination: Requires further investigation to identify the specific antibody type and clinical significance.
- Quality control: Perform Coombs control test to ensure AHG reagent reactivity and proper technique.
Function of Coombs Control Cells (CCC)
- Quality control measure to ensure the validity of the indirect antiglobulin (Coombs) test (IAT) reagents and technique.
- Consists of RBCs pre-coated with antibodies in the laboratory.
- Should agglutinate when mixed with AHG reagent, demonstrating its ability to detect antibody-coated RBCs.
- Negative CCC reaction with AHG suggests a technical error or reagent issue for example AHG was not added or inadequate, the reagent has expired or the AHG has been neutralized by free antibodies due to insufficient washing of the red cells.
Common Causes of Positive Indirect Coombs Test
A positive Indirect Antiglobulin Test (IAT) indicates the presence of unbound antibodies in the patient's serum that are capable of reacting with specific red blood cell (RBC) antigens. These antibodies are "clinically significant" if they can cause the destruction of red blood cells.
Presence of Alloantibodies (Transfusion-Related)
- Prior Transfusions: The most common reason. If a patient has received blood transfusions in the past, their immune system might have been exposed to foreign RBC antigens (from the donor blood) and produced antibodies against them. These antibodies will then be "free" in the patient's serum and react with reagent RBCs during the IAT.
- Previous Pregnancies: Similar to transfusions, exposure to fetal RBCs (if the fetus has inherited antigens from the father that are absent in the mother) during pregnancy or childbirth can sensitize the mother and lead to the production of alloantibodies.
Hemolytic Disease of the Fetus and Newborn (HDFN) / Rh Incompatibility (in pregnant women)
The IAT is crucial for screening pregnant women for antibodies (especially anti-D from Rh incompatibility, but also other blood group antibodies like anti-K, anti-Fya, anti-Jka) that could cross the placenta and cause HDFN in the fetus. A positive IAT in a pregnant woman indicates the presence of such antibodies.
Crossmatch Incompatibility (Pre-transfusion Testing)
When performing a major crossmatch (mixing patient serum with donor red blood cells), a positive IAT (specifically, an incompatible crossmatch) means the patient's serum contains antibodies that react with antigens on the donor's red blood cells. This indicates that the donor blood is not compatible for transfusion to that patient, preventing a potential hemolytic transfusion reaction.
Autoantibodies
Less commonly, a positive IAT can be due to autoantibodies – antibodies produced by the patient's immune system that react with their own red blood cell antigens. While autoantibodies are more typically detected by a positive Direct Antiglobulin Test (DAT), sometimes free autoantibodies in the serum can also cause a positive IAT. This might be seen in conditions like autoimmune hemolytic anemia.
Passively Acquired Antibodies
This occurs when antibodies are introduced into the patient's system from an external source, such as:
- Intravenous Immunoglobulin (IVIG) therapy: Contains a broad spectrum of antibodies.
- Transfusion of plasma or platelets containing donor antibodies.
Drug-Induced Antibodies
Certain medications can induce the formation of antibodies that react with red blood cell antigens, leading to a positive IAT. Examples include some antibiotics (like penicillin, cephalosporins), methyldopa, and quinidine.
Common Causes of False-Positive IAT
A false-positive Indirect Coombs Test (IAT) means that the test indicates the presence of antibodies when, in fact, there are no clinically significant red blood cell antibodies present. These can be frustrating for laboratories and clinicians as they may lead to unnecessary investigations or delays in patient care.
Technical Errors
These are among the most frequent causes and highlight the importance of meticulous technique in the blood bank.
- Inadequate Washing of Red Blood Cells: If the patient's red blood cells are not thoroughly washed before adding the Antihuman Globulin (AHG) reagent, unbound serum proteins (including non-specific immunoglobulins) can remain in the tube. These unbound proteins can neutralize the AHG reagent, leading to a false-positive reaction or, paradoxically, a false negative if too much protein is present.
- Over-centrifugation: Excessive centrifugation force can cause red blood cells to clump together non-specifically, mimicking agglutination.
- Improper Specimen Handling: Clotted samples, old samples, or samples collected in tubes with inappropriate anticoagulants can sometimes lead to non-specific agglutination.
- Contamination: Contamination of reagents or glassware can introduce substances that cause non-specific reactions.
Elevated Serum Proteins (Rouleaux Formation)
In conditions like multiple myeloma, Waldenström's macroglobulinemia, or other hypergammaglobulinemias, there is an abnormally high concentration of serum proteins (especially globulins). These proteins can cause red blood cells to stack on top of each other, resembling "stacks of coins," known as rouleaux. Rouleaux can be mistaken for true agglutination if not observed microscopically and confirmed by saline replacement (rouleaux disperses with saline, true agglutination does not).
Non-Specific Agglutination of Red Blood Cells
- Bacterial Contamination: Contaminated samples can lead to bacterial enzymes modifying red blood cell surfaces, causing them to agglutinate non-specifically.
- Cold Agglutinins (Potent IgM autoantibodies): While a positive indirect Coombs test usually implies IgG antibodies, very potent cold-reacting IgM autoantibodies might sometimes cause non-specific agglutination, particularly if incubation temperatures are not strictly maintained or if the cold autoantibody has a wide thermal amplitude.
- Abnormalities of RBCs: Certain red blood cell abnormalities or conditions (e.g., severe anemia, some parasitic infections) can make them prone to spontaneous agglutination.
Drugs/Therapeutic Monoclonal Antibodies
- Daratumumab (Darzalex): This is a significant cause of false positives in patients with multiple myeloma. Daratumumab is an anti-CD38 monoclonal antibody used to treat multiple myeloma. CD38 is expressed on both myeloma cells and red blood cells. The presence of daratumumab in the patient's serum will cause pan-agglutination (reactivity with all screening and panel cells) in the test because it binds to the CD38 antigen on the reagent red blood cells. Specialized techniques (e.g., Dithiothreitol (DTT) treatment of reagent cells) are often required to resolve this interference.
- Other Monoclonal Antibodies: Other therapeutic monoclonal antibodies, such as those targeting CD47, can also interfere with blood bank tests, leading to false positives.
- Certain Medications: While more commonly associated with drug-induced positive DAT, some drugs can also rarely lead to non-specific reactions in the test.
Human Anti-Animal Antibodies or Heterophile Antibodies
These are antibodies produced by the patient that react with animal-derived components present in some reagents (e.g., if the AHG reagent contains animal proteins). While less common with modern highly purified reagents, they can cause false positives.
When a false-positive IAT is suspected, the blood bank will typically perform additional steps to investigate, such as repeating the test with meticulously washed cells, performing a saline replacement procedure for rouleaux, using alternative reagents, or employing specialized techniques to resolve drug interference.
Alternatives to the Spin-Tube Method
While the traditional spin-tube indirect Coombs test (IAT) remains the gold standard for its sensitivity and reliability, several alternative methods offer advantages in terms of efficiency and automation:
- Solid-phase red cell adherence (SPRCA): RBCs are adhered to a solid surface, simplifying washing steps and enabling automated reading.
- Column agglutination: RBCs and reagents flow through a column, offering rapid results and ease of use.
- Gel card techniques: Miniaturized versions of the indirect Coombs test (IAT) using gel cards in microplates, enabling high throughput and standardization.
Frequently Asked Questions (FAQs)
What is the difference between direct antiglobulin (Coombs) test (DAT) and indirect Coombs test (IAT)?
Both direct antiglobulin (Coombs) test (DAT) and indirect Coombs test (IAT) are blood tests used in immunohematology to detect antibodies related to red blood cells (RBCs). However, they differ in what they are looking for:
- DAT (Direct Antiglobulin (Coombs) Test): This test checks for antibodies that are already attached to the surface of RBCs. This can happen due to autoimmune diseases or past blood transfusions. A positive direct antiglobulin (Coombs) test (DAT) indicates that RBC destruction might be occurring.
- IAT (Indirect Coombs Test): This test looks for free antibodies in the blood plasma that can potentially bind to RBCs. These antibodies haven't attached yet but could cause problems during a blood transfusion or in a newborn with hemolytic disease.
| Feature | Direct Antiglobulin (Coombs) Test (DAT) | Indirect Antiglobulin (Coombs) Test (IAT) |
|---|---|---|
| What it detects | Antibodies bound to RBCs | Free antibodies in blood plasma |
| Indicates | Current RBC destruction | Potential for future RBC destruction |
| Use cases | Autoimmune hemolytic anemia, transfusion reactions | Blood transfusion compatibility testing, hemolytic disease of the newborn |
What is indirect Coombs test used to detect?
IAT (Indirect Coombs Test) is used to detect free antibodies in the blood plasma that can potentially bind to red blood cells (RBCs). These antibodies haven't attached to RBCs yet, but their presence indicates a potential for future problems.
What is the purpose of indirect Coombs test?
The indirect antiglobulin test (IAT), also known as the indirect Coombs test, has two main purposes:
- Blood Transfusion Compatibility Testing: Before receiving a blood transfusion, an indirect Coombs test (IAT) is performed to ensure compatibility between the recipient's blood and the donor's blood. The test detects pre-existing antibodies in the recipient's plasma that could attack the donor's red blood cells. A positive indirect antiglobulin (Coombs) test (IAT) indicates incompatibility, and the recipient would need blood from a different donor with compatible blood type. This helps prevent transfusion reactions that can be serious or even fatal.
- Hemolytic Disease of the Fetus and Newborn (HDFN): In pregnant women, an indirect Coombs test (IAT) can be used to screen for HDFN. This condition occurs when a Rh-negative mother (lacks the Rh factor protein on red blood cells) carries a Rh-positive baby (inherits the Rh factor from the father). The mother's immune system may develop antibodies against the baby's Rh-positive red blood cells, potentially leading to anemia or other complications in the baby. A positive indirect Coombs test (IAT) in a pregnant woman might indicate a need for further monitoring and potential interventions to protect the baby.
Why is IAT used for antibody screening?
The indirect Coombs test (IAT) is a valuable tool for antibody screening because it offers several key advantages:
- High Sensitivity: The indirect Coombs test (IAT) is very sensitive in detecting antibodies against red blood cells (RBCs). Even small amounts of antibodies can be picked up by the test, which is crucial for preventing potential transfusion reactions.
- Antibody Specificity: While the initial indirect Coombs test (IAT) screening might not identify the specific antigen the antibody targets, further testing using the indirect Coombs test (IAT) with different types of RBCs (called an identification panel) can pinpoint the specific antigen-antibody interaction. This information is essential for selecting compatible blood for transfusion.
- Non-Agglutination Detection: Some antibodies, particularly IgG, may not cause visible clumping (agglutination) of RBCs on their own. The indirect Coombs test (IAT) overcomes this limitation by employing an antiglobulin reagent (also called Coombs' serum). This reagent acts as a bridge, binding to the patient's antibody if it's attached to an RBC, even if agglutination isn't readily apparent. This ensures detection of a wider range of antibodies that could cause problems during a transfusion.
- Safety and Efficiency: The indirect Coombs test (IAT) is a relatively simple and safe test to perform. It uses separated blood components (plasma or serum) instead of whole blood, allowing for easier manipulation and reducing the risk of errors. Furthermore, it's an efficient way to screen for multiple potential transfusion reactions compared to testing compatibility with every possible blood type.
What causes false negative indirect Coombs test?
There are a few reasons why an indirect Coombs test (IAT) might produce a false negative result, meaning it misses antibodies that are actually present in the blood plasma.
- Antibody Type: Most commercially available indirect Coombs test (IAT) tests are designed to detect antibodies against IgG, complement C3, or both on red blood cells (RBCs). If the antibody in question is of a different type, like IgM or IgA, it might not be picked up by the test. In these uncommon situations, a specific test for the suspected antibody type or a quantitative DAT (direct antiglobulin test) might be necessary.
- Low Antibody Levels: In some rare cases, autoimmune hemolytic anemia can be caused by antibody levels that fall below the detection limit of the indirect Coombs test (IAT). This threshold is typically around 150 to 500 IgG molecules per RBC.
- Technical Issues: Although less common, technical errors during the test procedure can lead to false negatives. Factors like inadequate centrifugation, delays in adding reagents, or problems with the antiglobulin reagent itself could affect the test's sensitivity.
- Interference from Rheumatoid Factor (RF): RF is an autoantibody (antibody against one's own tissues) that can sometimes interfere with the indirect Coombs test (IAT). The presence of RF can alter the interaction between the patient's antibody and the antiglobulin reagent, leading to a falsely negative result.
Interpreting indirect Coombs test (IAT) results should be done together with the patient's clinical history and other relevant tests. If a high suspicion of antibody presence exists despite a negative indirect Coombs test (IAT), further investigation or a different type of antibody test might be warranted.
What does a positive test before a blood transfusion mean?
A positive indirect Coombs test (IAT) before a blood transfusion indicates that your blood plasma contains antibodies that have the potential to react with certain antigens on donor red blood cells (RBCs). This reaction could cause a transfusion reaction, which can be serious or even fatal.
Why is AHG important in IAT?
The Antihuman Globulin (AHG) reagent is absolutely essential in the Indirect Antiglobulin Test (IAT) because it acts as a bridge or cross-linker, enabling the visualization of antigen-antibody reactions that would otherwise be undetectable. It converts an invisible antigen-antibody reaction (IgG coating on RBCs) into a visible one (agglutination), allowing for the detection of clinically significant antibodies in the Indirect Coombs Test.
The IAT primarily detects IgG antibodies. IgG antibodies are relatively small compared to red blood cells (RBCs). While they can bind to antigens on the surface of RBCs, they are typically too small to span the distance between two separate red blood cells and cause visible agglutination on their own. Even when multiple IgG molecules coat an RBC, the electrostatic repulsion between RBCs prevents them from clumping together.
When AHG is added to red blood cells that have been coated with IgG antibodies (from the patient's serum during the incubation phase), the AHG molecules bind to the Fc portion of these IgG antibodies that are attached to different red blood cells.
Because each AHG molecule has multiple binding sites, it can simultaneously bind to IgG antibodies on two or more different red blood cells. This cross-linking effect effectively bridges the gap between individual red blood cells, overcoming their natural repulsive forces and causing them to clump together. This clumping is what we observe as visible agglutination.
Disclaimer: This protocol is for educational purposes only. Local laboratory standard operating procedures take precedence. It is not intended to be a substitute for informed professional medical advice, diagnosis, or treatment. Always consult a qualified healthcare professional for clinical decision-making. While the information presented here is derived from credible medical sources and is believed to be accurate and up-to-date, it is not guaranteed to be complete or error-free. See additional information.
References
- American Association of Blood Banks (AABB). Technical Manual, 21st Edition, 2023.
- Dean L. Blood Groups and Red Cell Antigens [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2005.
- Bain BJ, Bates I, Laffan MA. Dacie and Lewis Practical Haematology: Expert Consult: Online and Print 12th Edition (Elsevier). 2016.
- Theis SR, Hashmi MF. Coombs Test. [Updated 2022 Sep 12]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK547707/