Procedure At A Glance
Perls' Prussian blue stain is used to histochemically detect and visualize ferric iron (Fe3+) deposits in cells and tissues.
- Fix slides in absolute methanol for 10 minutes and dry.
- Submerge slides in the working solution (2% potassium ferrocyanide + 2% HCl) at room temperature or in a 50°C water bath for 10 minutes.
- Rinse for 3-5 minutes with tap water, then with distilled water.
- Counterstain with 1% aqueous safranin and rinse again.
- Dry, mount with Depex, and cover with a coverslip.
Introduction: Why this stain matters
When a hematologist needs to know whether the body has too little iron, too much iron, or iron sitting in the wrong place, the Prussian blue stain is usually the first answer. The Perls' Prussian blue stain (Perls' stain for short) turns ferric iron deposits a vivid blue under the microscope, making invisible stores suddenly readable. That single color change drives real decisions: confirming iron deficiency, diagnosing hemochromatosis, monitoring transfusion-dependent patients, and classifying certain bone marrow disorders [1].
Principle of Perls' Prussian Blue Stain
Iron in tissues exists in two storage forms. Ferritin is the soluble, well-behaved storage protein. Hemosiderin is what forms when ferritin runs out of room — an insoluble, yellow-brown granule made of partly degraded ferritin and lipid, found mainly inside macrophages of the liver, spleen, and bone marrow.
The chemistry is simple. Hydrochloric acid releases ferric iron (Fe³⁺) from hemosiderin. The free Fe³⁺ then reacts with potassium ferrocyanide to form an insoluble bright blue pigment, ferric ferrocyanide — also known as Prussian blue [1]:
4 Fe³⁺ + 3 K₄[Fe(CN)₆] → Fe₄[Fe(CN)₆]₃ + 12 K⁺
Perls' detects only ferric iron (Fe³⁺).
Ferrous iron (Fe²⁺) is detected by a related method called Turnbull blue. The two stains are complementary. Aggregated hemosiderin stains strongly; small amounts of cytoplasmic ferritin generally do not.
Where Perls' Prussian blue stain is used
Perls' stain shows up across hematology and histopathology:
Diagnosing iron overload. In hereditary hemochromatosis, liver biopsy with Perls' reveals dense iron deposition in hepatocytes, often in an acinar pattern. In transfusion-dependent patients (β-thalassemia, sickle cell disease, lower-risk MDS), the stain shows secondary iron loading in macrophages.
Confirming iron deficiency. A bone marrow trephine that shows zero blue staining in macrophage iron stores is strong evidence of iron deficiency. This remains useful when serum ferritin is unreliable, for example in inflammation.
Classifying MDS with ring sideroblasts. Iron staining of a bone marrow aspirate identifies sideroblasts and ring sideroblasts — a key step in subtyping myelodysplastic syndromes.
Research applications. Iron metabolism studies, developmental biology, and investigations of iron dysregulation in neurodegenerative disease all use Perls' or its variants, often combined with ferritin immunohistochemistry.
Materials
- 2% Potassium Ferrocyanide ((K4Fe(CN)6.3H2O) (aqueous)
- 2% HCl (aqueous)
- 1% aqueous safranin (counterstain; nuclear fast red is also acceptable)
- Air dried peripheral blood or bone marrow smear (test sample) plus a known positive control slide
- Distilled water
- Absolute methanol
- Coplin jars
- Mounting medium (e.g., Depex) and coverslips
Protocol
- Label one slide as the positive control and another with the patient's name or registration number.
- Fix both slides in absolute methanol for 10 minutes, then air-dry.
- In a Coplin jar, prepare the working solution by mixing equal volumes of 2% potassium ferrocyanide and 2% hydrochloric acid (typically 30 mL of each, 1:1 v/v). Use immediately.
- Submerge the fixed, dried slides in the working solution.
- Incubate for 10 minutes at room temperature, or in a 50 °C water bath if a stronger reaction is required.
- Rinse under slow-running tap water for 3 to 5 minutes, then in distilled water.
- Counterstain with 1% safranin for 30 to 60 seconds. Rinse again in tap water followed by distilled water.
- Wipe the back and edges of the slide with a Kimwipe. Avoid touching the smear itself.
- Dry the slide. Use a hair dryer on the lowest setting in short bursts (no more than 10 seconds at a time) or air-dry at a tilt.
- Mount with Depex and apply a coverslip over the area of interest.
- The slide is now ready to view.
Safety note
Potassium ferrocyanide is generally low toxicity, but mixing it with strong acid — especially under heat or if allowed to evaporate — can release small amounts of hydrogen cyanide. Always work in a fume hood or well-ventilated area, wear gloves and eye protection, and dispose of waste as hazardous (see end of article) [1].
Interpretation: How to Read the Slide
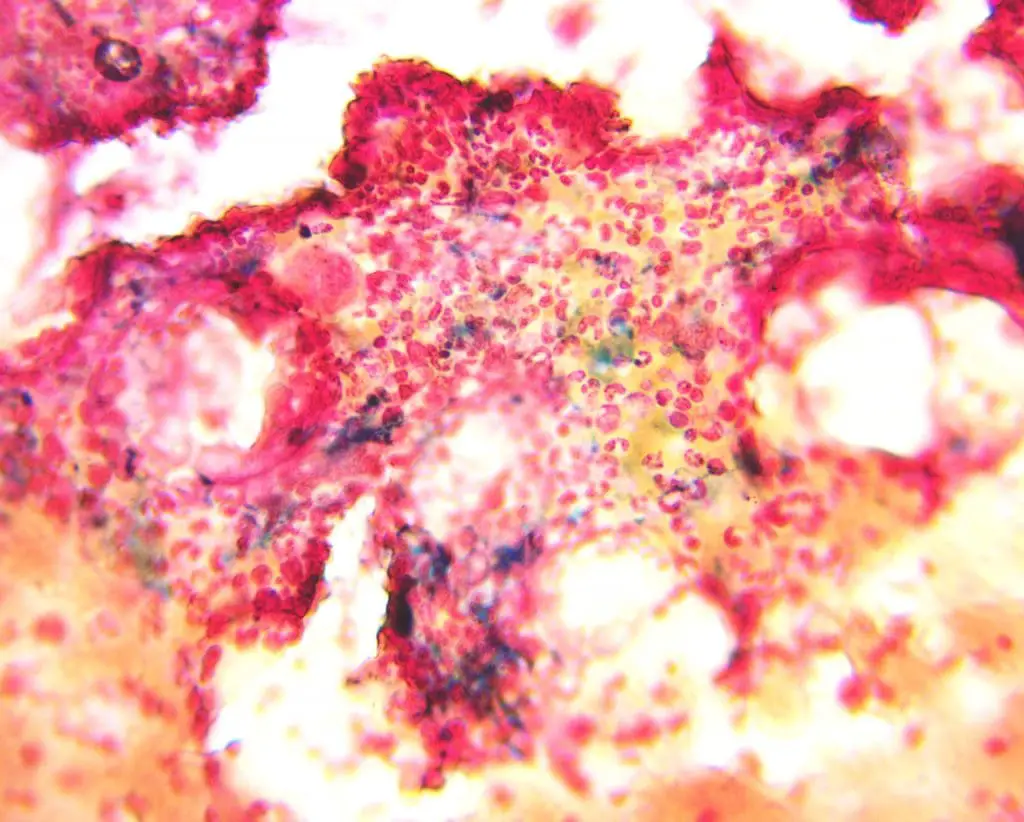
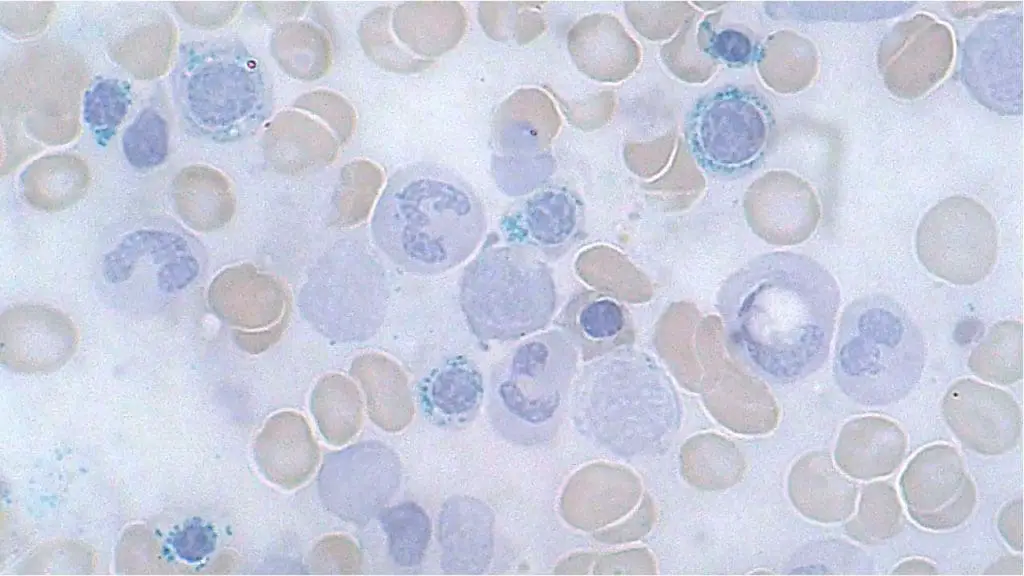
Start at low power (×10) to scan the smear or section for any blue. In a bone marrow aspirate, look first at the marrow particles and macrophages — that's where storage iron lives. Move to ×40 to grade intensity. Switch to ×100 oil immersion to confirm sideroblasts and ring sideroblasts, since you need to count granules and verify their position around the nucleus.
A few interpretive principles:
- A positive reaction is a discrete, granular blue. Hemosiderin stains coarse and chunky; do not confuse it with diffuse blue artifact.
- A negative reaction in bone marrow particles strongly suggests iron deficiency, provided the positive control slide stained correctly the same day.
- Ring sideroblasts require five or more iron granules forming a ring around at least one-third of the erythroid precursor's nucleus. The GFHC guideline recommends counting at least 100 erythroblasts before reporting a ring sideroblast percentage [1].
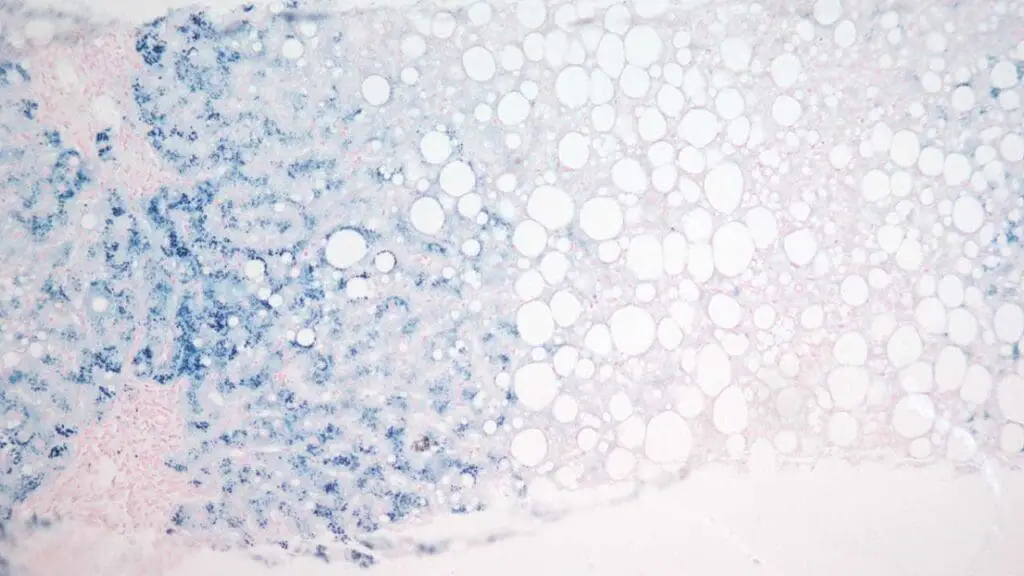
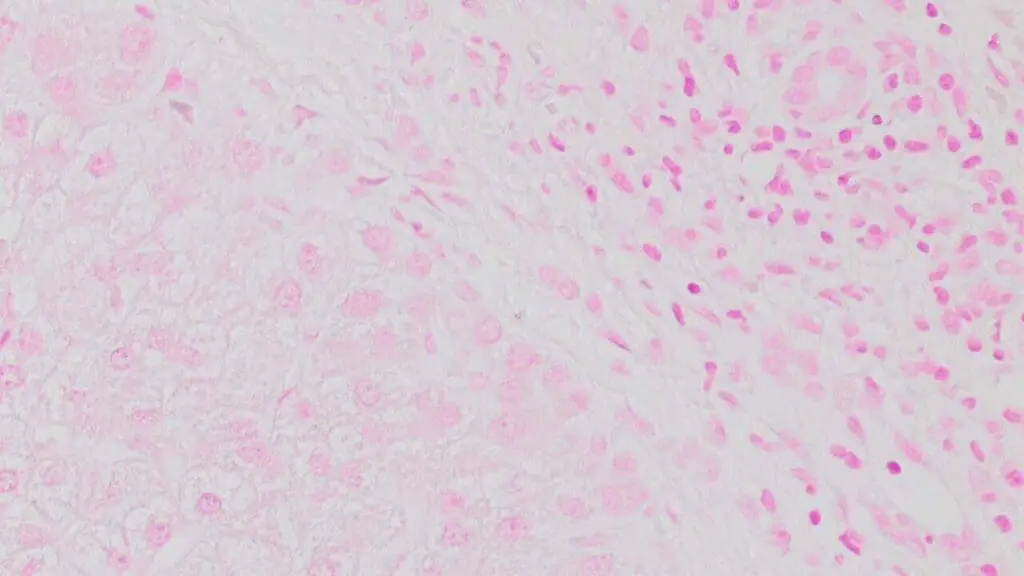
Grading summary
Grades 1 to 3 represent normal iron stores. Grades 4 to 6 indicate iron overload — most often hereditary hemochromatosis or transfusional loading in conditions such as β-thalassemia or chronically transfused MDS.
Ring sideroblasts and modern MDS classification
Ring sideroblasts have always been a morphologic marker of disordered heme synthesis. Under the 2022 WHO 5th edition and the 2022 International Consensus Classification (ICC), the relevant entity is MDS with mutated SF3B1 (ICC) or MDS with low blasts and SF3B1 mutation (WHO) [2,3].
Two diagnostic thresholds matter:
- ≥15% ring sideroblasts in bone marrow erythroblasts is sufficient on morphology alone to support the diagnosis (the "morphologic substitute" clause).
- 5–14% ring sideroblasts plus a confirmed SF3B1 mutation also qualifies [3,5].
Morphology guides treatment
Lower-risk MDS with ring sideroblasts is now treated with luspatercept, a therapy that reduces transfusion dependence [6,7]. The morphologic finding on a Perls'-stained smear therefore feeds directly into a treatment decision [5].
Pathological patterns to recognize
- Heavy hepatocyte iron, acinar pattern. Suggests hereditary hemochromatosis.
- Macrophage-predominant iron overload. Suggests transfusional loading or ineffective erythropoiesis.
- Patchy or zonal staining. Can indicate localized hemorrhage or chronic congestion.
- Ring sideroblasts in the marrow. Points to MDS-SF3B1, congenital sideroblastic anemia, or a drug- or toxin-induced sideroblastic anemia (isoniazid, chloramphenicol, copper deficiency, alcohol).
- Siderocytes in peripheral blood. Mature red cells with iron granules are not normal after splenic filtering — their presence may indicate post-splenectomy state or severe dyserythropoiesis.
Troubleshooting
Pre-staining issues. Improper or prolonged fixation can degrade hemosiderin and weaken staining. Harsh decalcification chemicals leach iron and cause false negatives.
Staining issues. The working solution must be freshly prepared and slightly acidic. Under-incubation gives weak blue; over-incubation gives diffuse background. Insufficient washing leaves unreacted reagent behind; excessive washing leaches the Prussian blue out.
Post-staining issues. Some mounting media fade the blue color over months. Always include a fresh positive control to verify the stain is working before reporting a negative result.
Limitations of Perls' Prussian blue stain
Perls' is qualitative to semi-quantitative, not quantitative. It detects aggregated iron well and diffuse iron poorly. It does not distinguish hemosiderin from other rare blue-staining pigments without confirmatory tests (Turnbull blue or ferritin immunohistochemistry). For accurate iron measurement, use serum ferritin, transferrin saturation, liver T2* MRI, or atomic absorption / ICP-MS on tissue.
Waste disposal
Treat all staining waste as hazardous. Segregate solid waste (slides, filters, paper) from liquid waste, label clearly with contents and date, and follow your institution's chemical waste protocol. Never pour Perls' working solution down the drain. Contact your environmental health and safety office if neutralization is required before collection.
Frequently Asked Questions (FAQs)
What does a Prussian blue stain show?
A Prussian blue stain (also called Perls' stain) reveals ferric iron (Fe³⁺) deposits in tissues and cells. Wherever iron is stored (most often as hemosiderin in macrophages of the liver, spleen, and bone marrow) the stain produces a bright blue color. Negative areas remain unstained or take on the color of the counterstain (commonly red or pink). It is the standard histochemical test for iron stores.
Why is Perls' Prussian blue stain used in hematology?
It serves three main purposes: confirming iron deficiency (no blue staining in bone marrow macrophages), confirming iron overload such as hemochromatosis or transfusional iron loading (intense blue staining), and identifying ring sideroblasts in suspected myelodysplastic syndrome. The stain remains a routine part of bone marrow assessment despite the rise of molecular testing.
What is the difference between hemosiderin and ferritin?
Ferritin is the body's main iron storage protein and is soluble — it releases iron when the body needs it. Hemosiderin is what forms when cells (especially macrophages) accumulate more iron than ferritin can hold. It is insoluble, releases iron slowly, and shows up as yellow-brown granules even before staining. Perls' stain detects the iron in both, but hemosiderin produces the strong, granular blue staining typically seen on slides.
Are ring sideroblasts always abnormal?
Yes. A normal bone marrow has occasional sideroblasts with a few scattered iron granules in the cytoplasm. A ring sideroblast (five or more iron granules forming a ring around at least a third of the nucleus) is always pathological. Ring sideroblasts point toward defective heme synthesis, most commonly in MDS with SF3B1 mutation, congenital sideroblastic anemia, or anemia caused by certain drugs and toxins.
Is Perls' stain used to measure how much iron is in the body?
Not really. Perls' stain is excellent for showing where iron is and giving a graded estimate (Gale grade 0 to 6 in bone marrow), but it cannot measure iron concentration accurately. For quantitative measurements, clinicians use serum ferritin, transferrin saturation, liver MRI (T2*), or biochemical iron assays on tissue.
Is the stain dangerous to perform?
Potassium ferrocyanide on its own is low-toxicity, but it is mixed with hydrochloric acid in the working solution. Heating this mixture or letting it dry out can release small amounts of hydrogen cyanide, so the staining must be done in a fume hood or well-ventilated area, waste must be neutralized and disposed of as hazardous, and gloves and eye protection are required throughout.
What are the different stains available for iron assessment?
| Stain | Advantage | Disadvantage |
|---|---|---|
| Perls' Prussian Blue Stain Histochemical | Simple and widely available | Less sensitive than some other stains |
| Turnbull Blue (TB) Histochemical | More sensitive than PPB | More complex and time-consuming procedure |
| Hemosiderin Stain (e.g., Hale) Histochemical | Stains both hemosiderin and ferritin | Less specific for iron |
| DAB-Iron (Immunohistochemistry) Immunological | Highly specific for ferritin | Requires specialized equipment and reagents |
| Ferritin Immunofluorescence Immunological | High sensitivity and specificity | Requires specialized equipment and expertise |
Glossary of Related Medical Terms
- Ferric iron (Fe³⁺): The oxidized form of iron. The form that Perls' stain detects.
- Ferrous iron (Fe²⁺): The reduced form of iron. Detected by Turnbull blue, not by Perls'.
- Hemosiderin: A yellow-brown, insoluble iron storage complex formed when ferritin gets overloaded. The main target of Perls' stain in tissues.
- Ferritin: The body's main soluble iron storage protein. Releases iron readily when needed.
- Sideroblast: A red blood cell precursor (in the bone marrow) that contains visible iron granules.
- Ring sideroblast: A sideroblast where iron has accumulated in the mitochondria and forms a ring around at least one-third of the nucleus. Five or more such granules are required.
- Siderocyte: A mature red blood cell containing iron granules. Should not normally be present after the spleen has filtered red cells.
- Erythropoiesis: Red blood cell production in the bone marrow.
- Ineffective erythropoiesis: Red cell precursors are made but die before becoming mature red cells. Common in thalassemia and MDS.
- Hemochromatosis: A condition of pathological iron overload, either inherited (often HFE gene) or acquired.
- Myelodysplastic syndrome (MDS): A group of bone marrow failure disorders with abnormal blood cell production. Some subtypes feature ring sideroblasts.
- SF3B1 mutation: A spliceosome gene mutation strongly linked to MDS with ring sideroblasts. Now used as a defining criterion under WHO 2022.
- Counterstain: A second dye applied after the main stain to reveal background structures (commonly nuclei) so the primary stain stands out.
Disclaimer: This protocol is for educational purposes only. Local laboratory standard operating procedures take precedence. It is not intended to be a substitute for informed professional medical advice, diagnosis, or treatment. Always consult a qualified healthcare professional for clinical decision-making. While the information presented here is derived from credible medical sources and is believed to be accurate and up-to-date, it is not guaranteed to be complete or error-free. See additional information.
References
- Lours, C., Cottin, L., Wiber, M., Andrieu, V., Baccini, V., Baseggio, L., Brouzes, C., Chatelain, B., Daliphard, S., Fenneteau, O., Geneviève, F., Girard, S., Leymarie, V., Maloum, K., Rieu, J. B., Sebahoun, G., Sudaka, I., Troussard, X., Wagner-Ballon, O., Wuilleme, S., … Lesesve, J. F. (2022). Perls' Stain Guidelines from the French-Speaking Cellular Hematology Group (GFHC). Diagnostics (Basel, Switzerland), 12(7), 1698. https://doi.org/10.3390/diagnostics12071698
- Khoury, J. D., Solary, E., Abla, O., Akkari, Y., Alaggio, R., Apperley, J. F., Bejar, R., Berti, E., Busque, L., Chan, J. K. C., Chen, W., Chen, X., Chng, W. J., Choi, J. K., Colmenero, I., Coupland, S. E., Cross, N. C. P., De Jong, D., Elghetany, M. T., Takahashi, E., … Hochhaus, A. (2022). The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia, 36(7), 1703–1719. https://doi.org/10.1038/s41375-022-01613-1
- Arber, D. A., Orazi, A., Hasserjian, R. P., Borowitz, M. J., Calvo, K. R., Kvasnicka, H. M., Wang, S. A., Bagg, A., Barbui, T., Branford, S., Bueso-Ramos, C. E., Cortes, J. E., Dal Cin, P., DiNardo, C. D., Dombret, H., Duncavage, E. J., Ebert, B. L., Estey, E. H., Facchetti, F., Foucar, K., … Tefferi, A. (2022). International Consensus Classification of Myeloid Neoplasms and Acute Leukemias: integrating morphologic, clinical, and genomic data. Blood, 140(11), 1200–1228. https://doi.org/10.1182/blood.2022015850
- Bain, B. J. and Leach, M. (2025). Blood cells: A practical guide (7th ed.). Wiley-Blackwell.
- Mortuza, S., Chin-Yee, B., James, T. E., Chin-Yee, I. H., Hedley, B. D., Ho, J. M., Saini, L., Lazo-Langner, A., Schenkel, L., Bhai, P., Sadikovic, B., Keow, J., Sangle, N., & Hsia, C. C. (2024). Myelodysplastic Neoplasms (MDS) with Ring Sideroblasts or SF3B1 Mutations: The Improved Clinical Utility of World Health Organization and International Consensus Classification 2022 Definitions, a Single-Centre Retrospective Chart Review. Current oncology (Toronto, Ont.), 31(4), 1762–1773. https://doi.org/10.3390/curroncol31040134
- Della Porta, M. G., Garcia-Manero, G., Santini, V., Zeidan, A. M., Komrokji, R. S., Shortt, J., Valcárcel, D., Jonasova, A., Dimicoli-Salazar, S., Tiong, I. S., Lin, C. C., Li, J., Zhang, J., Pilot, R., Kreitz, S., Pozharskaya, V., Keeperman, K. L., Rose, S., Prebet, T., Lai, Y., … Platzbecker, U. (2024). Luspatercept versus epoetin alfa in erythropoiesis-stimulating agent-naive, transfusion-dependent, lower-risk myelodysplastic syndromes (COMMANDS): primary analysis of a phase 3, open-label, randomised, controlled trial. The Lancet. Haematology, 11(9), e646–e658. https://doi.org/10.1016/S2352-3026(24)00203-5
- Fenaux, P., Platzbecker, U., Mufti, G. J., Garcia-Manero, G., Buckstein, R., Santini, V., Díez-Campelo, M., Finelli, C., Cazzola, M., Ilhan, O., Sekeres, M. A., Falantes, J. F., Arrizabalaga, B., Salvi, F., Giai, V., Vyas, P., Bowen, D., Selleslag, D., DeZern, A. E., Jurcic, J. G., … List, A. F. (2020). Luspatercept in Patients with Lower-Risk Myelodysplastic Syndromes. The New England journal of medicine, 382(2), 140–151. https://doi.org/10.1056/NEJMoa1908892