Procedure-At-A-Glance
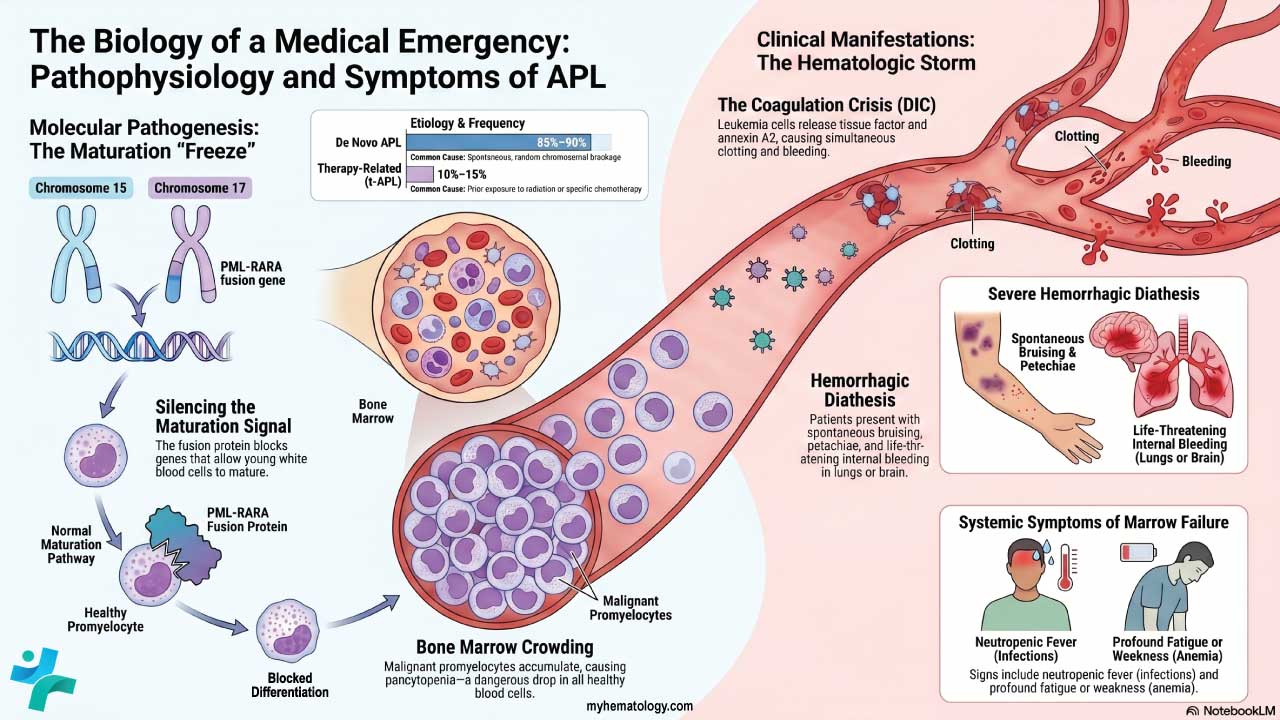
Myeloperoxidase is an enzyme inside the granules of neutrophils and other myeloid cells. It is the most specific cytochemical marker of myeloid lineage [3,4]. The myeloperoxidase stain helps separate acute myeloid leukemia (AML), where blasts are MPO-positive, from acute lymphoblastic leukemia (ALL), where blasts are MPO-negative [10].
- Fix an air-dried peripheral blood or bone marrow smear slide in a fixative (e.g., 10% formal ethanol) at room temperature (typically for 60 seconds).
- Cover the slide with the staining solution and incubate (typically for 1 minute). Rinse the slide again.
- Counterstain with Giemsa stain at a 1:10 dilution (typically for 10 minutes).
- Wash off the counterstain, wipe the back of the slide, and air dry in a tilted position or with a hairdryer on the lowest speed.
- Mount the slide with Depex and a cover slip, making it ready for viewing.
Why This Stain Matters
When a patient arrives with bruising, fatigue, and a high blast count on the blood film, the team needs to know quickly whether they are facing AML or ALL. The two leukemias look almost identical down the microscope but are treated very differently. A myeloperoxidase stain is one of the fastest ways to point the diagnosis in the right direction, often within an hour of receiving the sample.
Introduction
Myeloperoxidase (MPO) is a lysosomal enzyme. It lives inside the azurophilic granules of neutrophils, their precursors, eosinophils, and monocytes [3,5]. The MPO cytochemical stain makes this enzyme visible under the microscope, which is useful for several jobs:
- Diagnosing and classifying leukemia by separating MPO-positive myeloid blasts from MPO-negative lymphoid blasts.
- Supporting urgent decisions in acute promyelocytic leukemia (APL), where strong MPO positivity can support starting treatment before genetic confirmation [11].
- Helping assign lineage in mixed-phenotype acute leukemia (MPAL), where cytoplasmic MPO is now the main criterion for myeloid lineage [1,2].
- Studying inflammation, since tissue MPO levels rise where neutrophils accumulate [7].
Principle of Myeloperoxidase Stain
The principle is a simple enzyme reaction. MPO inside leukocyte granules uses hydrogen peroxide (H₂O₂) to oxidize a colorless chromogen. The product is an insoluble, stable, colored deposit that sticks to the spot where the enzyme is. The result is a brown to greenish-brown granular pattern in the cytoplasm of MPO-containing cells.
The stain separates cell types because of selective enzyme expression. Neutrophils and many other myeloid cells are packed with MPO. Lymphocytes are not. So when you apply the stain to a blood or marrow film, only myeloid cells light up. Lymphoid cells stay clear.
What Myeloperoxidase Does in the Body
MPO is one of the main antimicrobial weapons of the neutrophil. During the respiratory burst, neutrophils combine H₂O₂ with chloride ions, and MPO catalyzes the production of hypochlorous acid (HOCl), a bleach-like oxidant that kills bacteria and some fungi such as Candida species [5,6].
MPO is also important in neutrophil extracellular traps (NETs), web-like structures of DNA and granule proteins that neutrophils throw out to trap microbes in tissue [5]. Beyond infection, plasma MPO is now studied as a biomarker in atherosclerosis and acute coronary syndromes, and MPO-ANCA autoantibodies are central to diagnosing microscopic polyangiitis and eosinophilic granulomatosis with polyangiitis (EGPA) [7,15,16].

Role of Myeloperoxidase in Differentiating AML from ALL
This is the classic use of the stain. Myeloid blasts carry MPO; lymphoid blasts do not [3,4,10].
A positive cytochemistry result is traditionally defined as ≥3% of blasts staining positive. By flow cytometry, ≥10% is the most widely used cutoff, although published thresholds range from 3% to 20% depending on the method and control used [9,10].
A note on classification.
In the WHO 2022 and ICC 2022 systems, AML is defined mainly by genetic abnormalities rather than by morphology and cytochemistry alone. WHO 2022 no longer requires a fixed 20% blast count for AML with defining genetic abnormalities, and ICC 2022 sets a 10% threshold for most cases with recurrent genetic abnormalities [1,2]. MPO is still useful, but it now sits inside a wider workup that includes flow cytometry, cytogenetics, and molecular testing.
Mixed-Phenotype Acute Leukemia (MPAL)
Some leukemias don't fit cleanly into AML or ALL because the blasts show features of both lineages. These are called mixed-phenotype acute leukemias (MPAL). Current WHO and ICC criteria use cytoplasmic MPO (usually detected by flow cytometry or immunohistochemistry) as the primary criterion for assigning myeloid lineage in MPAL [1,2,12]. This is one of the most important modern uses of MPO testing.
Materials
This protocol uses 3,3′-diaminobenzidine (DAB) as the chromogen, which is the standard in modern hematology labs. The older benzidine recipe is no longer used routinely because benzidine is a known human carcinogen [4].
- Air-dried peripheral blood or bone marrow smear (ideally within 24 hours of collection; MPO activity is light-sensitive).
- Fixative: buffered formal acetone, or 10% formal ethanol prepared from one part 37–40% commercial formalin and nine parts absolute ethanol.
- Staining solution (DAB-based):
- DAB: 30 mg
- Phosphate or Tris buffer (pH ~7.4): 60 mL
- 30% hydrogen peroxide: ~120 µL (added just before use)
- Counterstain: Giemsa stain at a 1:10 dilution.
- Mounting medium (e.g., Depex) and coverslips.
Protocol
- Prepare an air-dried film, preferably within 24 hours of collection. Keep it out of direct light.
- Fix the slide in buffered formal acetone or 10% formal ethanol at room temperature for 60 seconds.
- Rinse in slow-running tap water for 15–30 seconds. Air dry.
- Place the slide on a staining rack. Cover the smear with freshly prepared DAB staining solution and incubate for 1 minute.
- Rinse in slow-running tap water for 1 minute.
- Counterstain with Giemsa at 1:10 dilution for 10 minutes.
- Wash off the counterstain.
- Wipe the back and edges of the slide with a lint-free wipe. Do not touch the smear.
- Dry with a hairdryer on the lowest setting (no more than 10 seconds at a time) or air dry tilted.
- Mount with Depex and a coverslip.
- The slide is ready to view.
Enzyme activity is preserved for up to 3 weeks if slides are stored in the dark.
Interpretation
The interpretation has two parts: what the cells look like, and how much positivity counts as a positive case.
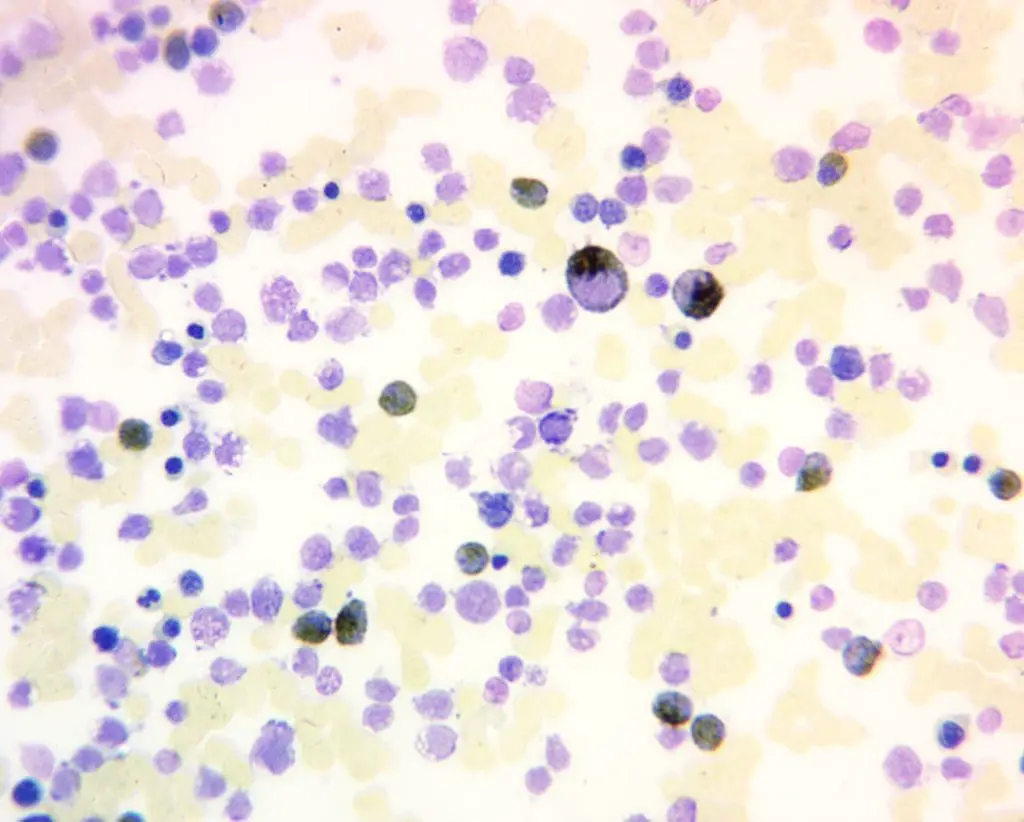
What You See Under the Microscope
A positive MPO reaction shows distinct brown to greenish-brown granules in the cytoplasm. You will see this in:
- Mature neutrophils (use these as your built-in positive control).
- Eosinophils (often a slightly different brownish-red shade).
- Most myeloblasts, promyelocytes, myelocytes, and metamyelocytes.
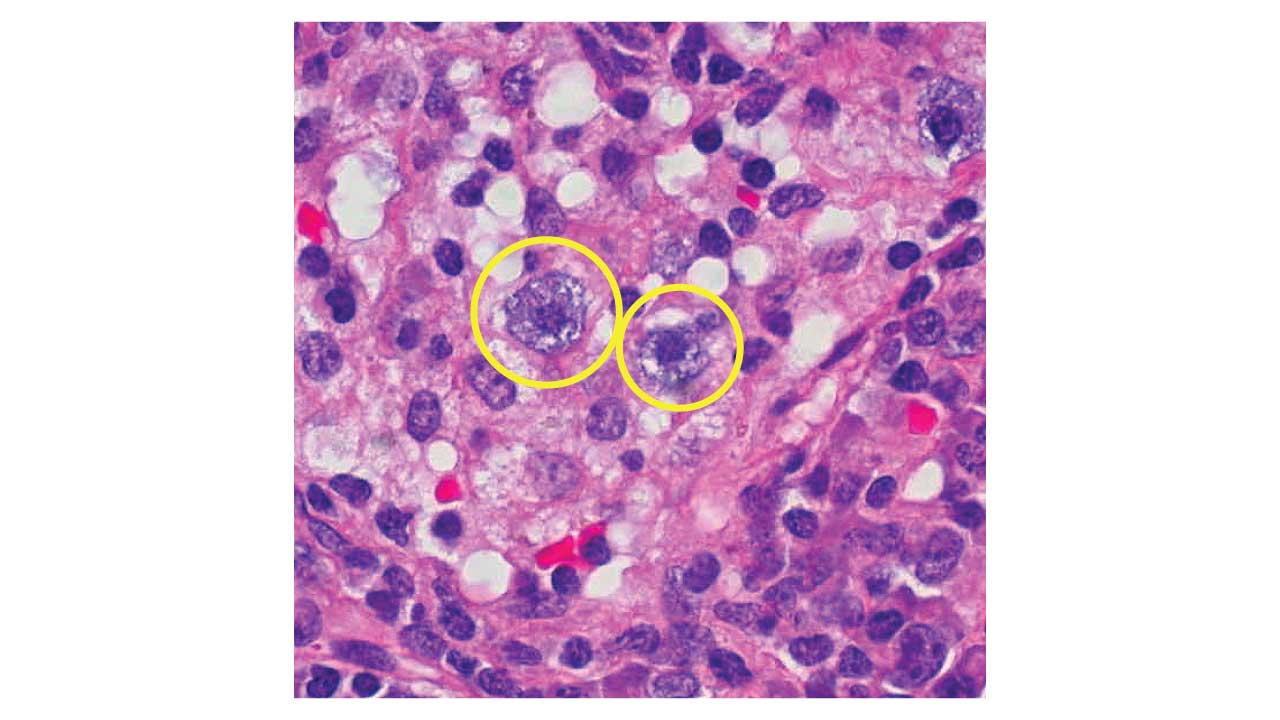
- Auer rods, when present, stain strongly MPO-positive and confirm AML.
A negative reaction means no brown granules. This is expected in:
- Lymphoblasts and lymphocytes (the key teaching point for AML vs ALL).
- The earliest myeloblasts, which may be MPO-negative or only dimly positive.
- Basophils, which are usually negative or only very weakly positive.
Monoblasts and monocytes sit between the two extremes. They are often negative or show only a faint, finely granular reaction [3,4].
Thresholds for Calling a Case Positive
| Method | Common cutoff for AML | Notes |
| Cytochemistry on slide | ≥3% of blasts MPO-positive | Classical FAB and WHO threshold [10] |
| Flow cytometry (cytoplasmic MPO) | ≥10% (range 3–20%) | More sensitive; cutoff varies by control used [9] |
| Immunohistochemistry | Qualitative | The most sensitive of the three; useful on trephine biopsies and "dry tap" cases [8] |
A 2018 prospective study of 120 AML cases showed that immunohistochemistry detected MPO in cases missed by both flow cytometry and cytochemistry. The authors recommended using more than one technique whenever AML is strongly suspected but the first test is negative [8].
MPO Staining Pattern in Different Cells and Tissues
| Cell Type/Tissue | MPO Pattern | Clinical Significance/Notes |
| Neutrophilic Granulocytes | Intense, dark granular cytoplasm | Maturation increases staining intensity |
| Eosinophils | Positive, often reddish-brown | Distinct from neutrophils |
| Monocytes | Faint, finely granular | Increases with maturation |
| Early Myeloblasts | Negative to weakly positive | Positivity grows with maturation |
| Lymphoblasts | Negative | Key feature for AL |
| Basophils | Usually negative or weakly positive | Variable |
| Spleen (Red Pulp) | Numerous MPO positive granulocytes | NUseful tissue control |
| Auer rods | Strongly positive | Pathognomonic for AML when seen |
Special Situations to Recognize
- Acute promyelocytic leukemia (APL): strong, diffuse MPO positivity with abundant Auer rods (often in bundles called faggot cells). Because APL causes life-threatening coagulopathy, a confident cytochemistry result can support starting all-trans retinoic acid (ATRA) before molecular confirmation arrives [11].
- Isolated MPO positivity in B-ALL: a small subset of adult B-ALL cases show low-level MPO expression. This is associated with poorer outcomes and is a recognized cause of misclassification as AML or MPAL [12].
- Hereditary MPO deficiency: one of the most common inherited neutrophil defects. Most people are healthy because alternative killing pathways compensate. The main clinical risk is severe candidal infection in patients who also have poorly controlled diabetes [5].
Tips for Optimal Result
- Stain fresh slides whenever possible. MPO is light-sensitive and degrades over time.
- Use mature neutrophils on the same slide as your internal positive control. If they don't stain, the run failed.
- Stick to standardized, validated reagents and keep H₂O₂ fresh.
- Have an experienced morphologist or pathologist review borderline cases.
- Always interpret MPO alongside the rest of the workup: flow cytometry, cytogenetics, and molecular testing [1,2,10].
How Myeloperoxidase Compares with Other Common Hematological Stains
Cytochemistry is rarely interpreted in isolation. Here is how MPO sits next to the other workhorse stains used during a leukemia workup.
| Stain Name | Principle | What it shows | Main diagnostic use |
| Myeloperoxidase (MPO) | Enzyme-driven oxidation of a chromogen by MPO in the presence of H₂O₂ | MPO in primary granules of myeloid cells | Most specific myeloid marker. Separates AML from ALL [3,4,10]. |
| Periodic Acid-Schiff (PAS) | Periodic acid oxidizes carbohydrates; Schiff reagent generates color | Glycogen, glycoproteins, mucopolysaccharides | Coarse block positivity in ALL; diffuse positivity in erythroid precursors in erythroleukemia [3]. |
| Perl's Prussian Blue (Iron) | Ferric iron reacts with potassium ferrocyanide | Non-heme iron, primarily hemosiderin | Detects ring sideroblasts in sideroblastic anemia; assesses marrow iron stores [4]. |
| Leukocyte Alkaline Phosphatase (LAP) | Detects alkaline phosphatase enzyme activity in mature neutrophils. | Alkaline phosphatase in secondary granules | Low score in CML; high score in leukemoid reaction and most other myeloproliferative neoplasms [3,4]. |
Conclusion
The myeloperoxidase stain is one of the oldest tools in the hematology lab, and it has aged well. It is cheap, fast, and gives a clear yes-or-no answer about myeloid lineage on the same day a sample arrives. The science around it has moved on; AML is now classified by its genetics, MPAL has its own criteria, and immunohistochemistry has overtaken cytochemistry in sensitivity. But when a team needs to know whether a sick patient has AML or ALL by the end of the morning, a well-run MPO stain still does the job.
Frequently Asked Questions (FAQs)
What cells are myeloperoxidase (MPO) positive?
The strongest MPO positivity is in neutrophils, which use the enzyme as a frontline weapon against microbes. Other MPO-positive cells include eosinophils (often with a distinct reddish-brown tone), monocytes (faint and finely granular), and most maturing myeloid precursors from promyelocytes onward. Myeloid blasts in AML are typically MPO-positive, which is the basis for using the stain to separate AML from ALL [3,10].
What color does an myeloperoxidase (MPO) stain produce?
The color depends on the chromogen. The most widely used reagent is 3,3′-diaminobenzidine (DAB), which gives a brown precipitate. Adding nickel or cobalt to a DAB reaction produces a darker, almost black product with better microscopic contrast. Older benzidine-based methods also produced brown but are no longer routine because benzidine is carcinogenic.
What are the main components of an MPO stain?
A modern MPO cytochemistry stain has three core ingredients:
- A chromogen (usually DAB) that goes from colorless to colored when oxidized by MPO.
- Hydrogen peroxide (H₂O₂) as the substrate the enzyme acts on.
- A buffer (often phosphate or Tris) to keep the pH stable so the enzyme works properly.
A counterstain such as Giemsa is added afterward so the cell nuclei and background show up clearly. Immunohistochemistry-based MPO stains add a fourth component: an anti-MPO antibody linked to an enzyme that triggers the chromogen reaction.
Do monoblasts stain positive for MPO?
Usually no, or only faintly. Monoblasts are at the very start of monocyte differentiation, and MPO expression in the monocyte lineage builds up as the cells mature. A strong positive reaction in a blast points away from a pure monoblastic process. A faint or partial reaction is possible and needs to be interpreted alongside flow cytometry and morphology [3,4].
What are the limitations of the MPO stain?
- Not perfectly specific. A small subset of adult B-ALL cases show isolated MPO positivity, which can cause misclassification [12].
- Not perfectly sensitive. Some AML subtypes have low MPO expression and can be missed by cytochemistry. Immunohistochemistry is the most sensitive of the three methods [8].
- Sample quality matters. Old slides, poor fixation, or stale reagents can all give false negatives.
- Never used alone. MPO must be read alongside flow cytometry, cytogenetics, and molecular testing, especially under the WHO 2022 and ICC 2022 frameworks [1,2].
What are the alternative tests to the MPO stain?
- Flow cytometry with cytoplasmic MPO antibodies: quantitative and very useful for MPAL [9].
- Immunohistochemistry (IHC) on trephine biopsies: the most sensitive method, especially in "dry tap" cases [8].
- Cytogenetics and molecular testing: identify the genetic lesions that now define AML in WHO 2022 and ICC 2022 [1,2].
- Other cytochemical stains: PAS, Sudan Black B (which closely mirrors MPO), and non-specific esterase (for monocytic lineage).
How accurate is MPO staining for diagnosing AML?
MPO is one of the most specific markers for the myeloid lineage, but it is not used alone. Current guidelines combine MPO with flow cytometry, immunohistochemistry, cytogenetics, and molecular testing. Studies show immunohistochemistry detects MPO in some cases missed by cytochemistry or flow cytometry, so labs often use more than one method.
Is MPO deficiency dangerous?
Inherited MPO deficiency is one of the most common neutrophil enzyme defects, and most people who have it stay completely healthy. Their neutrophils still kill most bacteria using other pathways. The main clinical concern is people with both MPO deficiency and poorly controlled diabetes, who have a higher risk of severe Candida (yeast) infections.
Why is MPO important in acute promyelocytic leukemia (APL)?
APL is a leukemia emergency because patients can bleed severely before any other diagnosis is confirmed. The leukemic cells in APL are very strongly MPO-positive and often contain Auer rods or bundles called faggot cells. A clear MPO result on the same day can support starting all-trans retinoic acid (ATRA) treatment before genetic confirmation arrives, which saves lives.
Can MPO be measured outside of leukemia diagnosis?
Yes. MPO is also studied as a marker of inflammation. Higher blood levels of MPO have been linked to atherosclerosis and acute coronary syndromes. Autoantibodies against MPO (MPO-ANCA) are key to diagnosing certain types of vasculitis, including microscopic polyangiitis and eosinophilic granulomatosis with polyangiitis (EGPA).
Glossary of Related Medical Terms
- Myeloperoxidase (MPO): An enzyme stored in granules inside certain white blood cells, mainly neutrophils. It helps these cells kill bacteria.
- Cytochemistry: A laboratory technique that uses chemical stains on cells to reveal specific enzymes or substances inside them.
- Chromogen: A colorless chemical used in a stain that becomes colored when it reacts with an enzyme, making the enzyme visible under a microscope.
- Azurophilic (primary) granules: Small sacs inside neutrophils that contain MPO and other antimicrobial molecules.
- Blast: An immature, early-stage blood cell. Large numbers of blasts in the marrow or blood suggest acute leukemia.
- Acute myeloid leukemia (AML): A fast-growing cancer of immature myeloid (non-lymphoid) blood cells. Blasts are usually MPO-positive.
- Acute lymphoblastic leukemia (ALL): A fast-growing cancer of immature lymphoid blood cells. Blasts are usually MPO-negative.
- Acute promyelocytic leukemia (APL/APML): A subtype of AML that is a medical emergency because of bleeding risk. Blasts are strongly MPO-positive.
- Mixed-phenotype acute leukemia (MPAL): A rare leukemia whose blasts show features of both myeloid and lymphoid lineages.
- Auer rod: A pink, needle-like structure inside leukemia blasts. When present, it confirms AML.
- Flow cytometry: A laboratory method that uses lasers and fluorescent tags to count cells and detect proteins inside or on them.
- Immunohistochemistry (IHC): A staining method that uses antibodies to find specific proteins in tissue sections.
- Respiratory burst: A rapid release of reactive oxygen molecules by activated neutrophils to kill microbes.
- Hypochlorous acid (HOCl): The bleach-like, microbe-killing chemical produced by MPO inside neutrophils.
- Neutrophil extracellular traps (NETs): Web-like fibers released by neutrophils that trap microbes outside the cell.
- ANCA (antineutrophil cytoplasmic antibody): An autoantibody against neutrophil proteins. MPO-ANCA is linked to certain forms of vasculitis.
- Counterstain: A second stain used to add background color so the main stained cells stand out clearly.
Disclaimer: This protocol is intended for informational purposes only and may need to be modified depending on the specific laboratory procedures and patient circumstances. Always consult with a qualified healthcare professional for guidance. See additional information.
References
- Khoury, J.D., Solary, E., Abla, O. et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia 36, 1703–1719 (2022). https://doi.org/10.1038/s41375-022-01613-1
- Arber, D. A., Orazi, A., Hasserjian, R. P., Borowitz, M. J., Calvo, K. R., Kvasnicka, H. M., Wang, S. A., Bagg, A., Barbui, T., Branford, S., Bueso-Ramos, C. E., Cortes, J. E., Dal Cin, P., DiNardo, C. D., Dombret, H., Duncavage, E. J., Ebert, B. L., Estey, E. H., Facchetti, F., Foucar, K., … Tefferi, A. (2022). International Consensus Classification of Myeloid Neoplasms and Acute Leukemias: integrating morphologic, clinical, and genomic data. Blood, 140(11), 1200–1228. https://doi.org/10.1182/blood.2022015850
- Bain, B. J. and Leach, M. (2025). Blood cells: A practical guide (7th ed.). Wiley-Blackwell.
- Bain BJ, Bates I, Laffan MA. Dacie and Lewis Practical Haematology: Expert Consult: Online and Print 12th Edition (Elsevier). 2016.
- Aratani Y. (2018). Myeloperoxidase: Its role for host defense, inflammation, and neutrophil function. Archives of biochemistry and biophysics, 640, 47–52. https://doi.org/10.1016/j.abb.2018.01.004
- Klebanoff, S. J., Kettle, A. J., Rosen, H., Winterbourn, C. C., & Nauseef, W. M. (2013). Myeloperoxidase: a front-line defender against phagocytosed microorganisms. Journal of leukocyte biology, 93(2), 185–198. https://doi.org/10.1189/jlb.0712349
- Khan, A. A., Alsahli, M. A., & Rahmani, A. H. (2018). Myeloperoxidase as an Active Disease Biomarker: Recent Biochemical and Pathological Perspectives. Medical sciences (Basel, Switzerland), 6(2), 33. https://doi.org/10.3390/medsci6020033
- Ahuja, A., Tyagi, S., Seth, T., Pati, H. P., Gahlot, G., Tripathi, P., Somasundaram, V., & Saxena, R. (2018). Comparison of Immunohistochemistry, Cytochemistry, and Flow Cytometry in AML for Myeloperoxidase Detection. Indian journal of hematology & blood transfusion : an official journal of Indian Society of Hematology and Blood Transfusion, 34(2), 233–239. https://doi.org/10.1007/s12288-017-0849-1
- Peffault de Latour, R., Legrand, O., Moreau, D., Perrot, J. Y., Blanc, C. M., Chaoui, D., Casadevall, N., & Marie, J. P. (2003). Comparison of flow cytometry and enzyme cytochemistry for the detection of myeloperoxydase in acute myeloid leukaemia: interests of a new positivity threshold. British journal of haematology, 122(2), 211–216. https://doi.org/10.1046/j.1365-2141.2003.04384.x
- Arber, D. A., Borowitz, M. J., Cessna, M., Etzell, J., Foucar, K., Hasserjian, R. P., Rizzo, J. D., Theil, K., Wang, S. A., Smith, A. T., Rumble, R. B., Thomas, N. E., & Vardiman, J. W. (2017). Initial Diagnostic Workup of Acute Leukemia: Guideline From the College of American Pathologists and the American Society of Hematology. Archives of pathology & laboratory medicine, 141(10), 1342–1393. https://doi.org/10.5858/arpa.2016-0504-CP
- Saksena, A., Desai, P., Chauhan, V., Gupta, N., & Singh, T. (2017). Revisiting cytochemical myeloperoxidase (MPO) stain for early diagnosis of acute promyelocytic leukemia in areas with limited resources: An experience from a tertiary care center in India. Blood, 130(Supplement 1), 5096.
- Kovach, A. E., Raikar, S. S., Oberley, M. J., Wertheim, G., Rabin, K. R., Punia, J. N., ... & Wood, B. L. (2021). Standardization in the diagnosis of mixed phenotype acute leukemia (MPAL): Semiquantitative, universally applicable flow cytometric criteria for immunophenotypic lineage assignment and isolated MPO. Blood, 138, 4475. https://doi.org/10.1182/blood-2021-154295
- Lambeth J. D. (2007). Nox enzymes, ROS, and chronic disease: an example of antagonistic pleiotropy. Free radical biology & medicine, 43(3), 332–347. https://doi.org/10.1016/j.freeradbiomed.2007.03.027
- Jennette, J. C., & Falk, R. J. (2014). Pathogenesis of antineutrophil cytoplasmic autoantibody-mediated disease. Nature reviews. Rheumatology, 10(8), 463–473. https://doi.org/10.1038/nrrheum.2014.103