TL;DR
Acute Intermittent Porphyria or AIP is an autosomal dominant hepatic disorder causing acute neurovisceral crises without cutaneous symptoms.
- Etiology ▾: HMBS gene mutations triggered by porphyrinogenic drugs, progesterone, or caloric restriction.
- Pathophysiology ▾: ALAS1 induction causes a metabolic bottleneck, leading to the accumulation of neurotoxic ALA and PBG.
- Presentation ▾: Characterized by the “5 Ps”: Pain (abdominal), Polyneuropathy, Psychosis, Port-wine urine, and Precipitants.
- Diagnosis ▾: Massive elevation of PBG in a spot urine sample during an attack is the primary screening test.
- Differential ▾: Distinguished from surgical “acute abdomen” by a lack of peritonitis and from GBS by proximal weakness.
- Treatment ▾: Immediate IV Hemin to repress the heme pathway, supplemented by strictly “safe” symptomatic medications.
- Complications ▾: Requires lifelong surveillance for Hepatocellular Carcinoma (HCC) and progressive Chronic Kidney Disease (CKD).
*Click ▾ for more information
Introduction
Acute Intermittent Porphyria (AIP) is a rare, autosomal dominant metabolic disorder resulting from a partial deficiency of the enzyme hydroxymethylbilane synthase (HMBS), also known as porphobilinogen deaminase (PBGD). It is the most common of the four acute hepatic porphyrias.
Unlike the cutaneous porphyrias, which primarily affect the skin, Acute Intermittent Porphyria (AIP) is characterized by neurovisceral crises. These are discrete, life-threatening episodes of neurological and systemic dysfunction triggered by the massive accumulation of porphyrin precursors, specifically δ-aminolevulinic acid (ALA) and porphobilinogen (PBG), within the liver and nervous system.
The “Intermittent” Nature and Low Penetrance
The term “intermittent” is central to the clinical understanding of the disease. Most individuals who inherit a mutation in the HMBS gene remain asymptomatic throughout their lives; this is known as low clinical penetrance (estimated at only 1% to 10%).
Symptoms only manifest when a secondary “trigger” (such as drugs, hormones, or fasting) drastically increases the demand for hepatic heme, causing the metabolic pathway to “bottleneck” at the site of the enzyme deficiency.
Classification
Acute Intermittent Porphyria (AIP) is categorized under two primary frameworks:
- Hepatic Porphyria: Because the metabolic “explosion” and precursor overproduction occur primarily in the hepatocytes.
- Acute Porphyria: Because it presents with sudden, severe symptomatic “attacks” rather than a chronic, steady-state progression.
Epidemiology and Demographics
While Acute Intermittent Porphyria (AIP) is a pan-ethnic condition, it has a notable prevalence in certain populations due to founder effects (e.g., Northern Sweden, where prevalence is roughly 1 in 1,500).
- Gender Bias: Symptoms are significantly more common in females (approx. 4:1 ratio), typically emerging during the third or fourth decade of life.
- Hormonal Influence: Attacks are rare before puberty and after menopause, highlighting the critical role of cyclical female sex hormones (progesterone) in triggering the disease.
Clinical Significance: The “Great Imitator”
Acute Intermittent Porphyria (AIP) is a chameleon. Because its primary symptoms, for example, severe abdominal pain, psychiatric disturbances, and motor weakness are non-specific, it is frequently misdiagnosed as an acute surgical abdomen (like appendicitis), psychiatric illness, or Guillain-Barré syndrome. A delay in diagnosis can lead to irreversible neurological damage or death, making early clinical suspicion paramount.
Genetic Basis and Pathophysiology
The HMBS Gene
The primary cause of Acute Intermittent Porphyria (AIP) is a mutation in the Hydroxymethylbilane Synthase (HMBS) gene, formerly known as the Porphobilinogen Deaminase (PBGD) gene.
The gene is located on the long arm of chromosome 11 (11q23.3). Over 400 different mutations in the HMBS gene have been identified, including missense, nonsense, and splice-site mutations. This variety explains why the severity can vary between different families, though it does not strictly correlate with the clinical phenotype (genotype-phenotype disparity).
These mutations result in an approximately 50% reduction in the activity of the HMBS enzyme in all tissues, including hepatocytes and erythrocytes.
Inheritance: Autosomal Dominant with Low Penetrance
Acute Intermittent Porphyria (AIP) follows an autosomal dominant inheritance pattern, meaning a mutation in only one of the two alleles is sufficient to cause the enzymatic deficiency. However, the most striking genetic feature of Acute Intermittent Porphyria (AIP) is its low clinical penetrance.
It is estimated that only 10% to 20% of individuals carrying the HMBS mutation will ever experience a clinical attack. The majority of gene carriers remain “latent” where they have the enzyme deficiency but never develop the neurovisceral symptoms.
Pathophysiology of AIP
The transition from a latent state to an acute attack (the “second hit”) is driven by factors that increase the demand for hepatic heme.
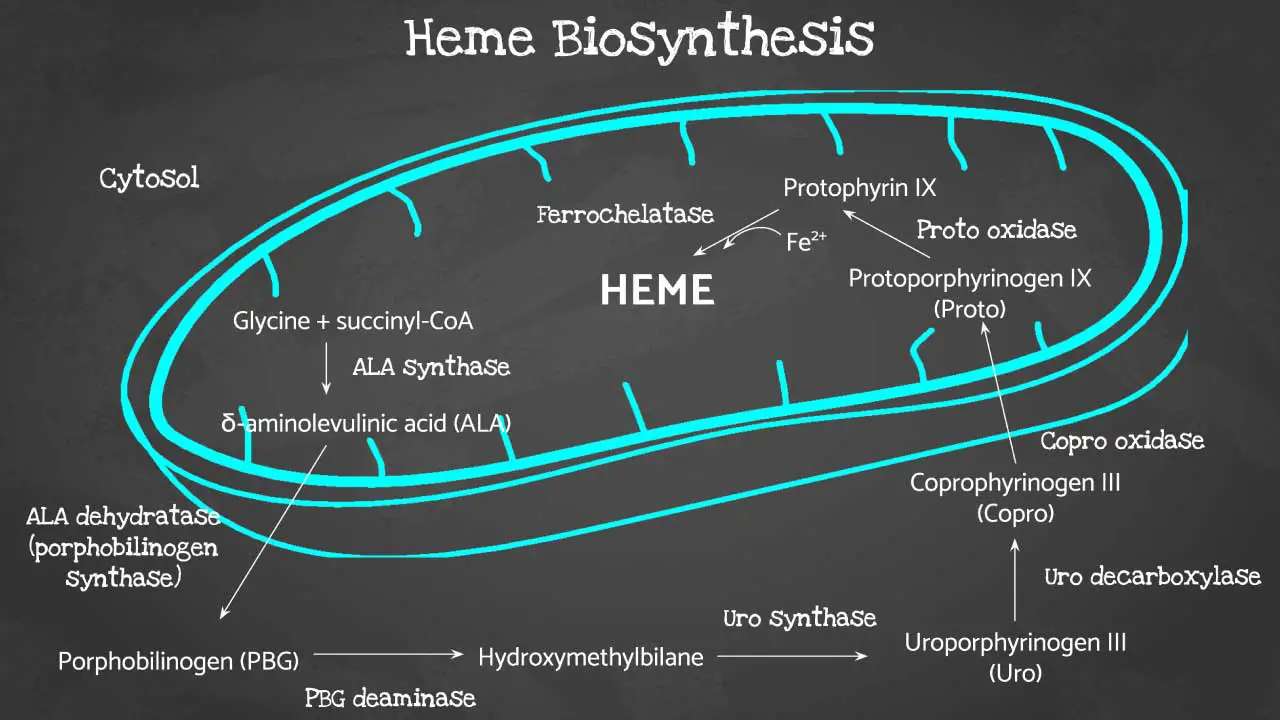
Heme is required for the production of Cytochrome P450 (CYP) enzymes in the liver. When the liver needs more heme (due to drugs, hormones, or stress), it upregulates the first and rate-limiting enzyme of the pathway: δ-aminolevulinic acid synthase 1 (ALAS1).
In a healthy individual, the pathway flows smoothly. In an Acute Intermittent Porphyria (AIP) patient, a trigger induces ALAS1. The pathway produces large amounts of precursors until it hits the HMBS step, which is already operating at 50% capacity. The “bottleneck” causes a massive backup and subsequent overflow of the upstream neurotoxic precursors: Aminolevulinic Acid (ALA) and Porphobilinogen (PBG).
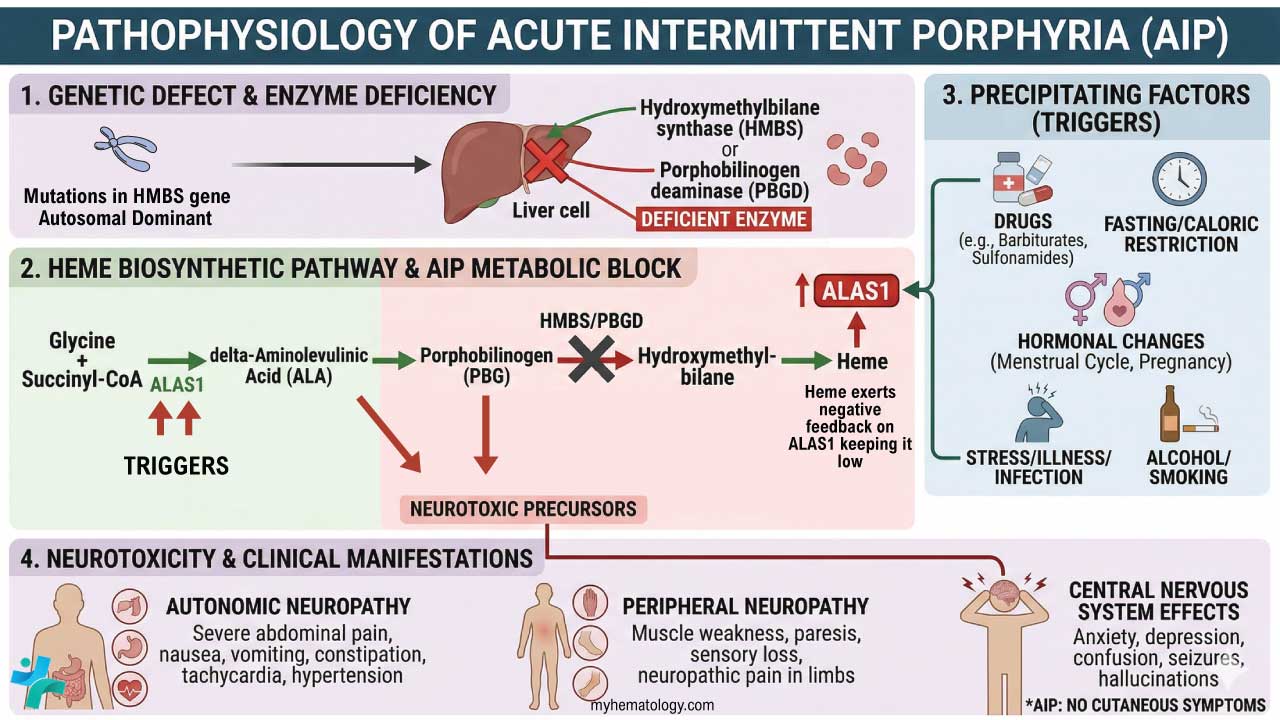
Common Precipitating Factors (Triggers)
The etiology of an attack is almost always multi-factorial.
- Endocrine Factors: Steroid hormones, particularly progesterone, are potent inducers of ALAS1. This explains why attacks are rare before puberty and often occur during the luteal phase of the menstrual cycle.
- Pharmacological Triggers: Many drugs (e.g., barbiturates, sulfonamide antibiotics, rifampin, and certain anticonvulsants) induce hepatic CYP enzymes, thereby depleting the “free heme pool” and triggering ALAS1 via negative feedback.
- Nutritional Status: Low caloric intake or prolonged fasting (common during illness or dieting) removes the “glucose effect,” which normally represses ALAS1.
- Physiological Stress: Infections, surgery, and psychological stress can increase metabolic demands and trigger a crisis.
Mechanisms of Neurotoxicity
The hallmark of an Acute Intermittent Porphyria (AIP) attack is neurological damage. While the exact mechanism is still debated, two primary theories explain why elevated ALA (and possibly PBG) causes symptoms.
A. The GABA-Analog Theory
ALA is structurally very similar to γ-aminobutyric acid (GABA), the primary inhibitory neurotransmitter in the central nervous system. ALA can act as an agonist at GABA receptors or inhibit GABA release. This interference with GABAergic signaling is thought to contribute to the seizures and psychiatric disturbances (anxiety, psychosis) seen in acute attacks.
B. Direct Neurotoxicity and Oxidative Stress
- Oxidative Damage: ALA can undergo auto-oxidation, generating reactive oxygen species (ROS) that cause oxidative damage to nerve cell membranes and DNA.
- Axonal Degeneration: The accumulation of these precursors leads to patchy demyelination and primary axonal degeneration of motor and sensory nerves. This explains the progressive muscle weakness and peripheral neuropathy.
C. Heme Deficiency in Nerve Cells
Some researchers suggest that the deficiency of heme itself within the nerve cells (distal to the metabolic block) impairs the function of essential hemeproteins like mitochondrial cytochromes and nitric oxide synthase, leading to cellular energy failure.
Systemic Consequences
The neurotoxic effects of ALA/PBG manifest in three distinct areas of the nervous system:
- Autonomic Nervous System: Damage to autonomic ganglia leads to sympathetic overactivity (tachycardia, hypertension) and enteric nervous system dysfunction (leading to the classic “abdominal pain” without inflammation).
- Peripheral Nervous System: Typically affects motor neurons first, leading to ascending paralysis that can eventually involve the respiratory muscles.
- Central Nervous System: Hypothalamic involvement can lead to the Syndrome of Inappropriate Antidiuretic Hormone (SIADH), resulting in the life-threatening hyponatremia often seen during crises.
Clinical Presentation and Symptoms
An acute attack of Acute Intermittent Porphyria (AIP) typically develops over hours or days and can last for weeks if untreated. The “classic triad” often taught is abdominal pain, peripheral neuropathy, and psychiatric disturbances, though patients rarely present with all three simultaneously.
Gastrointestinal (Autonomic Neuropathy)
Abdominal pain is the most common presenting symptom, occurring in over 90% of acute attacks. Severe, constant, and poorly localized. It is often described as “boring” or “tearing.” The abdominal exam is usually unremarkable (the abdomen is soft, non-tender, and lacks peritoneal signs). This discrepancy often leads to the mistaken assumption that the patient is drug-seeking or malingering.
Associated GI Symptoms include nausea, vomiting, and profound constipation (due to decreased intestinal motility).
Neurological Manifestations
The neurological symptoms are the most dangerous, as they can lead to permanent disability or death.
A. Autonomic Instability
Beyond the GI tract, the autonomic nervous system is often in a state of “overdrive”.
- Cardiovascular: Tachycardia (present in ~80% of attacks) and hypertension.
- Other: Diaphoresis (excessive sweating) and tremors.
B. Peripheral Neuropathy
Unlike many other neuropathies, Acute Intermittent Porphyria (AIP) typically presents as a proximal motor neuropathy, often beginning in the shoulders or arms rather than the feet.
It can progress to involve all four limbs and, most critically, the respiratory muscles (leading to respiratory failure) and bulbar muscles (leading to dysphagia). Sensory loss can occur but is usually less prominent than motor weakness.
C. Central Nervous System (CNS)
Seizures occur in roughly 10–20% of attacks. Caution: Many common anti-epileptics (like phenytoin or carbamazepine) are porphyrinogenic and will worsen the attack.
Encephalopathy can cause confusion, agitation, and altered consciousness.
Psychiatric Symptoms (“Porphyric Madness”)
Psychiatric manifestations can be the first or only sign of an attack, frequently leading to psychiatric admissions before the metabolic cause is identified.
- Acute Symptoms: Severe anxiety, insomnia, depression, paranoia, and visual/auditory hallucinations.
- Chronic Impact: Some patients may experience chronic low-grade psychiatric instability between acute episodes.
Urinary Changes (The “Port-Wine” Sign)
While not a symptom the patient “feels,” the urine provides a vital diagnostic clue.
Fresh Urine may appear normal or slightly yellow when first voided. When exposed to light and air, the excess porphobilinogen (PBG) polymerizes into porphobilin, turning the urine a dark reddish-brown or “port-wine” color.
Electrolyte Imbalances
Hyponatremia is a classic laboratory finding during an acute attack (occurring in up to 40% of cases). It is primarily caused by:
- SIADH: Due to hypothalamic involvement.
- Gastrointestinal Loss: Through vomiting.
- Renal Loss: Excess sodium excretion.
Severe hyponatremia can exacerbate CNS symptoms and trigger seizures.
No Photosensitivity
Acute Intermittent Porphyria (AIP) does not cause skin lesions. If a patient has acute neurovisceral symptoms PLUS blistering skin lesions on sun-exposed areas, the diagnosis is more likely to be Variegate Porphyria (VP) or Hereditary Coproporphyria (HCP), not Acute Intermittent Porphyria (AIP).
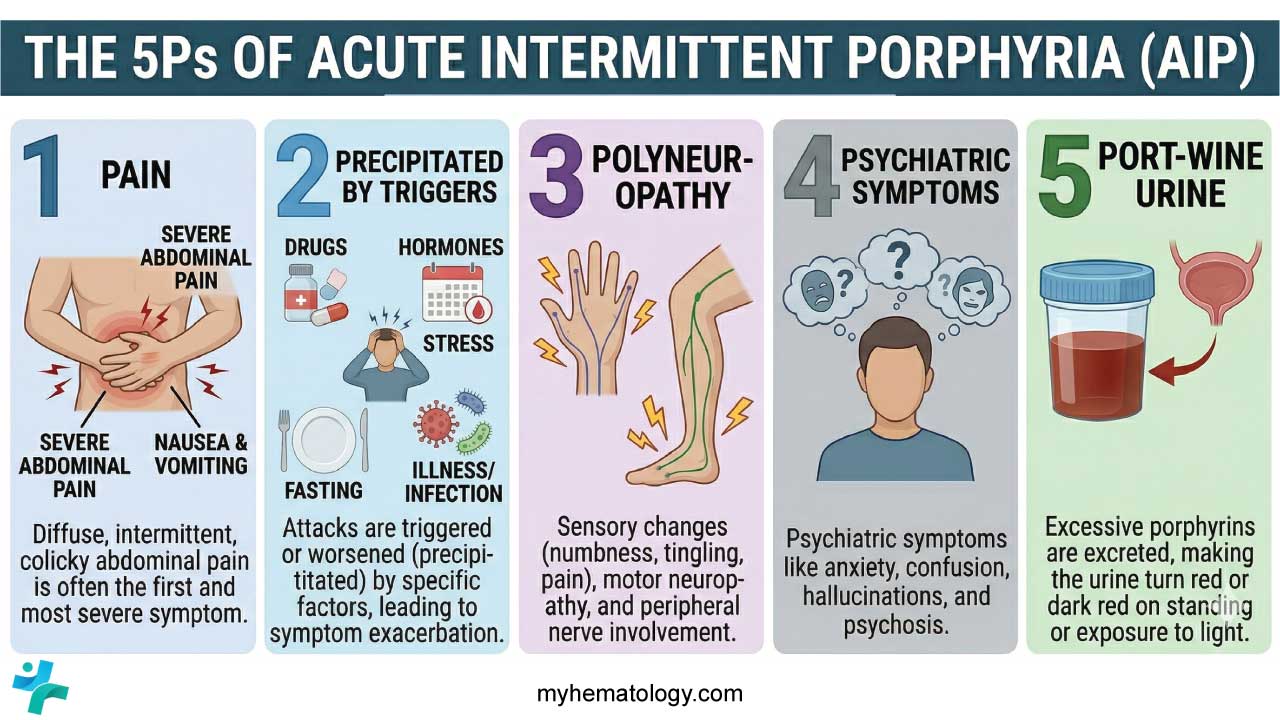
The “5 P’s” of Acute Intermittent Porphyria (AIP)
| P | Manifestation |
| Pain | Severe abdominal pain, often the first sign. |
| Polyneuropathy | Proximal motor weakness, potentially involving respiratory muscles. |
| Psychiatric | Anxiety, psychosis, and confusion. |
| Port-wine Urine | Darkening of urine upon exposure to light. |
| Precipitated | Triggered by Drugs (P450 inducers), Fasting, or Hormones. |
Laboratory Investigations and Diagnosis
The diagnostic process is divided into three phases: rapid screening during an attack, biochemical confirmation, and genetic identification.
First-Line Screening: The “Spot Urine” PBG Test
When a patient presents with a suspected acute attack, the single most important test is a random (spot) urine sample for Porphobilinogen (PBG).
During an acute attack of AIP, urinary PBG levels are typically 5 to 20 times the upper limit of normal. Aminolevulinic Acid (ALA) is also significantly elevated, though PBG is considered more specific for the acute hepatic porphyrias.
Samples must be protected from light, as PBG and ALA are light-sensitive and will degrade, potentially leading to a false-negative result. A normal urinary PBG during a period of active symptoms essentially rules out Acute Intermittent Porphyria (AIP) as the cause of those symptoms.
Qualitative Clues
- The Hoesch/Watson-Schwartz Test: A rapid bedside chemical test where Ehrlich’s reagent is added to urine. A cherry-red color indicates the presence of PBG. While largely replaced by quantitative lab assays, it remains a classic teaching point.
- Visual Inspection: If urine is left standing in light and air, the PBG polymerizes into porphobilin, which gives the urine a characteristic dark reddish-brown or “port-wine” appearance. However, the absence of this color change does not rule out an attack.
Confirmatory Biochemical Testing
Once an acute attack is biochemically suggested by high urinary PBG, further testing is required to differentiate Acute Intermittent Porphyria (AIP) from other porphyrias (like Variegate Porphyria or Hereditary Coproporphyria).
- Erythrocyte HMBS Activity: Measuring the activity of the enzyme Hydroxymethylbilane Synthase in red blood cells. In ~90% of Acute Intermittent Porphyria (AIP) patients, enzyme activity is reduced by 50%. About 5–10% of patients have “Type II” Acute Intermittent Porphyria (AIP), where the enzyme deficiency is limited to the liver, and red blood cell activity remains normal.
- Plasma/Fecal Porphyrins: These are used to distinguish Acute Intermittent Porphyria (AIP) from other acute porphyrias. In Acute Intermittent Porphyria (AIP), fecal porphyrins are typically normal or only slightly elevated, whereas they are high in Variegate Porphyria.
Genetic Testing (The Gold Standard)
Molecular genetic analysis of the HMBS gene is the definitive method for confirming the diagnosis. It is used to identify the specific mutation in the index patient.
Once a mutation is identified in a symptomatic patient, genetic testing is the only reliable way to screen asymptomatic relatives (who may have normal urine PBG levels) to provide counseling on trigger avoidance.
Supportive Laboratory Findings
Several non-specific laboratory abnormalities are frequently seen during an acute crisis.
| Lab Finding | Frequency | Pathophysiology |
| Hyponatremia | ~40% of attacks | Caused by SIADH or gastrointestinal/renal sodium loss. |
| Hypomagnesemia | Common | Associated with the severity of the attack. |
| Leukocytosis | Variable | Often mild, even in the absence of infection. |
| Elevated LFTs | Variable | Mild elevations in AST/ALT are sometimes seen. |
Differential Diagnosis
Acute Intermittent Porphyria (AIP) is often called “The Great Imitator.” Below are the primary conditions that must be ruled out when considering a diagnosis of Acute Intermittent Porphyria (AIP).
The “Acute Abdomen”
Since 90% of AIP patients present with severe abdominal pain, they are often first evaluated by surgical teams.
- Appendicitis, Cholecystitis, or Diverticulitis: These typically present with peritoneal signs (rebound tenderness, guarding) and fever, which are notably absent in Acute Intermittent Porphyria (AIP).
- Bowel Obstruction: While Acute Intermittent Porphyria (AIP) causes decreased motility and constipation, imaging (X-ray/CT) in Acute Intermittent Porphyria (AIP) will show a non-specific ileus rather than a mechanical transition point.
- Renal Colic: The “boring” nature of porphyric pain can mimic kidney stones, but urinalysis in Acute Intermittent Porphyria (AIP) will show elevated PBG rather than hematuria.
Neurological Disorders
When motor weakness or paralysis is the dominant feature, Acute Intermittent Porphyria (AIP) can be mistaken for acute neuromuscular diseases.
- Guillain-Barré Syndrome (GBS): Like Acute Intermittent Porphyria (AIP), GBS presents with progressive weakness. However, GBS is typically an ascending paralysis (starting in the feet), whereas Acute Intermittent Porphyria (AIP) often starts proximally (shoulders/arms). Furthermore, GBS shows “albuminocytologic dissociation” in the CSF (high protein, normal cells), which is not typical of Acute Intermittent Porphyria (AIP).
- Lead Poisoning: This is a critical differential because lead inhibits several enzymes in the heme synthesis pathway (primarily ALA dehydratase).
- Similarities: Abdominal pain and peripheral neuropathy.
- Difference: Lead poisoning causes an elevation in ALA but not PBG. There could be “lead lines” on gingiva or basophilic stippling on a peripheral blood smear.
Other Porphyrias
It is vital to distinguish Acute Intermittent Porphyria (AIP) from its “siblings” in the acute hepatic porphyria family.
- Variegate Porphyria (VP) and Hereditary Coproporphyria (HCP): These can present with the exact same neurovisceral attacks as Acute Intermittent Porphyria (AIP).
- The Key Differentiator: VP and HCP often present with cutaneous photosensitivity (blisters, fragile skin). Acute Intermittent Porphyria (AIP) never involves the skin.
- Porphyria Cutanea Tarda (PCT): This is the most common porphyria globally, but it is purely cutaneous and does not cause acute neurovisceral attacks.
Psychiatric Conditions
Because Acute Intermittent Porphyria (AIP) can present with isolated psychosis or anxiety, patients may be inappropriately triaged to psychiatric units.
Acute Intermittent Porphyria (AIP) should be suspected if the psychiatric symptoms are accompanied by unexplained abdominal pain, hyponatremia, or a history of triggers (like starting a new medication or extreme dieting).
Metabolic and Endocrine Disorders
- Hyponatremia (SIADH): While SIADH is a common complication of Acute Intermittent Porphyria (AIP), it can also be caused by other conditions. If a patient has profound hyponatremia and “weird” neurological symptoms, Acute Intermittent Porphyria (AIP) should be on the differential.
- Diabetic Ketoacidosis (DKA): Can present with abdominal pain and vomiting, but the metabolic acidosis and hyperglycemia of DKA are absent in Acute Intermittent Porphyria (AIP).
Summary of Differential Diagnosis
| Condition | Abdominal Pain | Weakness | Skin Lesions | Key Differentiating Factor |
| AIP | Yes (Severe) | Yes (Proximal) | No | Elevated Urinary PBG |
| Appendicitis | Yes (Localized) | No | No | Peritoneal signs, Leukocytosis |
| Guillain-Barré | Rare | Yes (Distal) | No | CSF Albuminocytologic dissociation |
| Lead Poisoning | Yes | Yes | No | High ALA but Normal PBG |
| Variegate Porphyria | Yes | Yes | Yes | Photosensitivity present |
Treatment and Management
Immediate General Measures
The first priority in a suspected acute crisis is stabilization and the prevention of further metabolic escalation.
- Hospitalization: Patients should be admitted for close monitoring of neurological status, respiratory function, and electrolyte balance.
- Identify and Remove Triggers:
- Discontinue all potentially porphyrinogenic medications immediately (consult databases like https://drugs-porphyria.org/).
- Treat underlying infections or inflammatory stressors.
- End restrictive dieting; ensure adequate caloric intake.
- Hydration and Electrolytes: Correct hyponatremia slowly to avoid central pontine myelinolysis.
Specific Metabolic Therapy
The goal is to provide “Heme” to the liver, which restores negative feedback on ALAS1, thereby stopping the production of neurotoxic ALA and PBG.
A. Intravenous Hemin (The Gold Standard)
- Indication: First-line therapy for severe attacks (neuropathy, seizures, severe pain, or hyponatremia).
- Mechanism: Exogenous hemin enters the hepatic free heme pool and represses the synthesis of ALAS1.
- Administration: Usually given as Heme Arginate or Panhematin via a central line (to avoid thrombophlebitis) for 3 – 4 days.
- Effectiveness: Rapidly reduces urinary ALA and PBG levels and usually aborts the clinical attack within days.
B. Carbohydrate Loading (Glucose Therapy)
- Indication: Only for mild attacks without neurological involvement.
- Mechanism: High glucose intake (300 – 400g/day) downregulates ALAS1 gene expression via the transcription factor PGC-1α.
- Limitation: It is significantly less effective than hemin. If the patient does not improve within 24 – 48 hours, they must be switched to hemin therapy.
Symptomatic Management
Choosing the “right” supportive medications is critical as many standard drugs (like barbiturates or certain anticonvulsants) can be fatal in Acute Intermittent Porphyria (AIP).
| Symptom | Safe Medications | AVOID (Porphyrinogenic) |
| Pain | Opioids (Morphine, Fentanyl) | NSAIDs (some), Pentazocine |
| Nausea | Ondansetron, Prochlorperazine | Metoclopramide (high risk) |
| Tachycardia/HTN | Beta-blockers (Propranolol) | Calcium channel blockers (some) |
| Seizures | Gabapentin, Levetiracetam, Diazepam | Phenytoin, Carbamazepine, Valproate |
Prevention and Long-Term Management
For patients with recurrent attacks, management shifts toward long-term prophylaxis.
- Givosiran (siRNA Therapy): A revolutionary therapy that uses small interfering RNA to “silence” the ALAS1 messenger RNA in the liver. It significantly reduces the frequency of acute attacks and the need for hemin.
- Hormonal Suppression: In women with cyclical premenstrual attacks, GnRH analogues (e.g., Leuprolide) can be used to prevent the progesterone surge.
- Liver Transplantation: A last-resort, curative option for patients with severe, life-threatening, refractory disease.
Prognosis and Long-term Complications
- Hepatocellular Carcinoma (HCC): Patients over age 50 with Acute Intermittent Porphyria (AIP) have a 30-to-70-fold increased risk of HCC, even without cirrhosis. Annual liver ultrasound and AFP monitoring are mandatory.
- Chronic Kidney Disease (CKD): Chronic hypertension and the toxic effects of porphyrin precursors often lead to progressive renal impairment.
- Chronic Neuropathy: Repeated attacks can lead to permanent muscle weakness and chronic neuropathic pain.
Frequently Asked Questions (FAQs)
Why is AIP called “The Great Imitator”?
Acute Intermittent Porphyria (AIP) earned this nickname because its symptoms (abdominal pain, weakness, and psychiatric changes) are non-specific and overlap with dozens of more common conditions. It is frequently misdiagnosed as appendicitis, Guillain-Barré syndrome, or primary acute psychosis, often leading to unnecessary surgeries or harmful drug treatments.
Why are women of childbearing age at higher risk?
Progesterone is a potent inducer of the enzyme ALAS1. The cyclical fluctuations of progesterone during the menstrual cycle, specifically during the luteal phase, can trigger recurrent attacks. This also explains why attacks are exceptionally rare before puberty and after menopause.
Can a patient have Acute Intermittent Porphyria (AIP) if their family history is negative?
Yes. Because of low clinical penetrance, the HMBS mutation can be passed through multiple generations of “latent” carriers who never show symptoms. The absence of a family history does not rule out the diagnosis.
Why is IV Glucose used for mild attacks?
High carbohydrate intake (glucose) inhibits the transcription of the ALAS1 gene in the liver. By “turning off” the production of the first enzyme in the pathway, glucose reduces the buildup of the toxic precursors ALA and PBG. However, it is much less potent than Hemin and is only appropriate for mild cases.
If a spot urine PBG test is negative during pain, does it rule out AIP?
Yes, for that specific episode. During a true neurovisceral attack of Acute Intermittent Porphyria (AIP), urinary PBG levels are always significantly elevated (typically >5 – 10 times the upper limit of normal). If the PBG is normal while the patient is symptomatic, the symptoms are likely caused by something else.
Is Acute Intermittent Porphyria (AIP) a lifelong condition?
The genetic mutation is lifelong, but the disease itself is episodic. Many patients may have only one or two attacks in their lifetime, while others may suffer from chronic “smoldering” symptoms or frequent recurrent crises. Modern treatments like Givosiran have significantly improved the long-term management of recurrent cases.
Glossary of Related Medical Terms
- ALA (δ-aminolevulinic acid): The first precursor in the heme biosynthetic pathway. It is structurally similar to GABA and is considered a primary neurotoxin in AIP.
- ALAS1 (δ-aminolevulinic acid synthase 1): The rate-limiting enzyme of heme synthesis in the liver. It is induced by drugs, hormones, and fasting, leading to the “overflow” of precursors in AIP.
- Autosomal Dominant with Low Penetrance: A genetic pattern where inheriting one mutated gene is sufficient to cause the condition, yet only a small fraction (approx. 10%) of carriers ever manifest clinical symptoms.
- Heme: An iron-containing porphyrin that serves as a prosthetic group for hemoglobin, myoglobin, and Cytochrome P450 enzymes.
- HMBS (Hydroxymethylbilane Synthase): The third enzyme in the heme pathway (also known as PBG deaminase). A 50% deficiency of this enzyme is the primary defect in AIP.
- Hyponatremia: Low blood sodium levels, frequently seen in Acute Intermittent Porphyria (AIP) due to SIADH or gastrointestinal losses during an attack.
- Neurovisceral: Referring to symptoms that involve both the nervous system (neuropathy, psychosis) and the internal organs (abdominal pain, constipation).
- PBG (Porphobilinogen): The second precursor in the heme pathway. Its massive elevation in the urine is the diagnostic hallmark of an acute porphyric attack.
- Porphyrinogenic: Refers to any factor (drug, chemical, or physiological state) that induces the heme pathway and can precipitate an acute attack.
- SIADH: Syndrome of Inappropriate Antidiuretic Hormone; a condition where the body retains too much water, often complicating Acute Intermittent Porphyria (AIP) crises.
References
- https://drugs-porphyria.org/
- Gonzalez-Mosquera LF, Sonthalia S. Acute Intermittent Porphyria. [Updated 2023 May 1]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2026 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK547665/
- https://porphyriafoundation.org/for-patients/types-of-porphyria/aip/
- Valbuena Valecillos, A., Yatham, P., Alderman, M., Shapiro, L., Tiozzo, E., & Gober, J. (2023). Acute Intermittent Porphyria: A Review and Rehabilitation Perspective. Cureus, 15(8), e44260. https://doi.org/10.7759/cureus.44260
- Kizilaslan, E. Z., Ghadge, N. M., Martinez, A., Bass, M., Winayak, R., Mathew, M., Amin, R., Khan, M., & Kizilbash, N. (2023). Acute Intermittent Porphyria’s Symptoms and Management: A Narrative Review. Cureus, 15(3), e36058. https://doi.org/10.7759/cureus.36058
- Ma, Y., Teng, Q., Zhang, Y., & Zhang, S. (2020). Acute intermittent porphyria: focus on possible mechanisms of acute and chronic manifestations. Intractable & rare diseases research, 9(4), 187–195. https://doi.org/10.5582/irdr.2020.03054
- Lei, J. J., Li, S., Dong, B. X., Yang, J., & Ren, Y. (2024). Acute intermittent porphyria: a disease with low penetrance and high heterogeneity. Frontiers in genetics, 15, 1374965. https://doi.org/10.3389/fgene.2024.1374965
- Zhao, L., Wang, X., Zhang, X., Liu, X., Ma, N., Zhang, Y., & Zhang, S. (2020). Therapeutic strategies for acute intermittent porphyria. Intractable & rare diseases research, 9(4), 205–216. https://doi.org/10.5582/irdr.2020.03089
- Dickey, A. K., & Leaf, R. K. (2024). Givosiran: a targeted treatment for acute intermittent porphyria. Hematology. American Society of Hematology. Education Program, 2024(1), 426–433. https://doi.org/10.1182/hematology.2024000663
- Bustad, H. J., Kallio, J. P., Vorland, M., Fiorentino, V., Sandberg, S., Schmitt, C., Aarsand, A. K., & Martinez, A. (2021). Acute Intermittent Porphyria: An Overview of Therapy Developments and Future Perspectives Focusing on Stabilisation of HMBS and Proteostasis Regulators. International journal of molecular sciences, 22(2), 675. https://doi.org/10.3390/ijms22020675